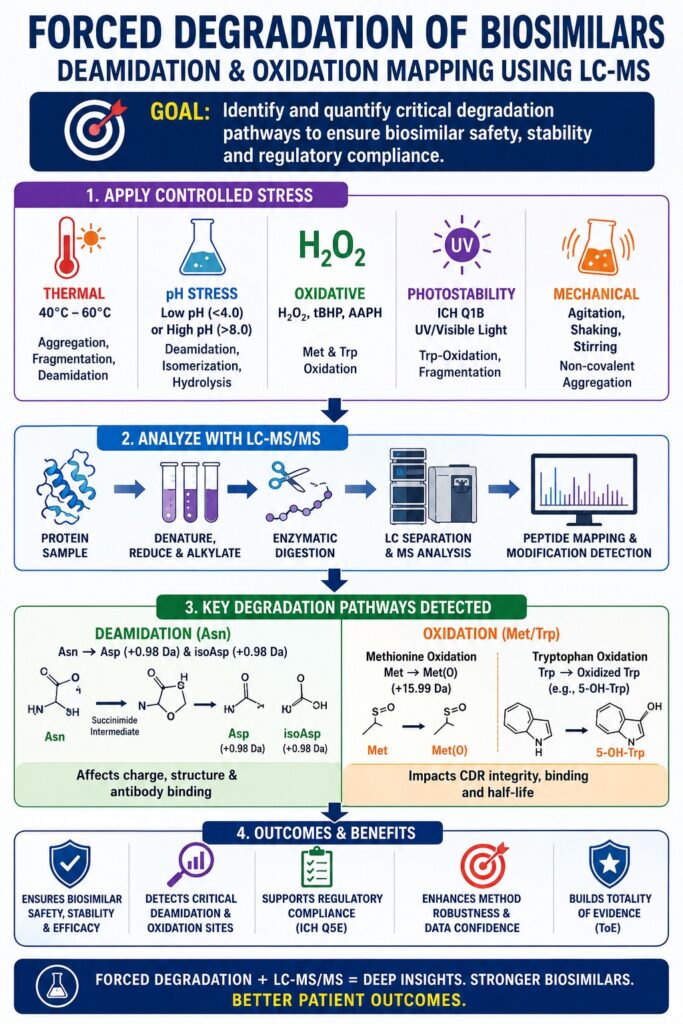
The Strategic Role of Forced Degradation of Biosimilars in Development
Forced Degradation of Biosimilars is an essential analytical approach used to study the stability of therapeutic proteins and to confirm that analytical methods can detect structural changes accurately. In this process, proteins are exposed to controlled stress conditions that go beyond normal storage environments, leading to about 5–20% degradation. This intentional breakdown helps scientists identify critical chemical changes such as deamidation, oxidation, and fragmentation.
In the highly competitive biosimilar industry, these studies are very important. They help show that a biosimilar behaves in a similar way to the original reference product when exposed to stress. This creates a comparable “stress-response fingerprint.” Such detailed analysis reduces the chances of failure in later development stages and ensures that the product remains safe, stable, and effective throughout its lifecycle.
Explore how to accurately verify your molecule’s structure: Learn more about Biosimilar Characterization Using Mass Spectrometry
Share via:
Primary Stressors and Their Analytical Objectives in Forced Degradation of Biosimilars
| Stress Category | Specific Stressor | Targeted Degradation Pathway | Analytical Objective |
|---|---|---|---|
| Thermal Stress | 40°C to 60°C | Aggregation, Fragmentation, Deamidation | Determine conformational stability and simulate long-term storage |
| pH Stress | Low pH (<4.0) or High pH (>8.0) | Deamidation, Isomerization, Hydrolysis | Identify liabilities in purification buffers and elution steps |
| Oxidative Stress | H₂O₂, tBHP, AAPH | Methionine and Tryptophan Oxidation | Evaluate CDR integrity and FcRn binding potential |
| Photostability | ICH Q1B UV/Visible Light | Trp-Oxidation, Fragmentation | Assess degradation risks during drug handling and administration |
| Mechanical Stress | Agitation, Shaking, Stirring | Non-covalent Aggregation | Simulate shipping and processing conditions |
Ensure your analytical methods meet global standards: Access Regulatory Support for Generic Drug Development
Chemical Mechanisms of Asparagine Deamidation Under Forced Degradation of Biosimilars
Asparagine deamidation is a natural chemical process where an asparagine (Asn) residue converts into aspartic acid (Asp) and isoaspartic acid (isoAsp). During Forced Degradation of Biosimilars, this reaction speeds up under alkaline conditions, making it a key factor in studying charge variation in therapeutic antibodies.
The Succinimide Intermediate and Backbone Rearrangement
Deamidation occurs through the formation of a cyclic intermediate called succinimide. This happens when the peptide backbone reacts with the side chain of asparagine. The reaction depends on how flexible the protein structure is and the surrounding amino acids.
Once formed, the succinimide intermediate is unstable and breaks down in two ways:
- Hydrolysis at the \alpha-carbonyl produces aspartic acid.
- Hydrolysis at the \beta-carbonyl produces isoaspartic acid.
Isoaspartic acid formation is especially important because it slightly changes the protein backbone. This small structural shift can affect how the antibody binds to its target, especially in sensitive regions like CDRs.
Sequence Motifs and Environmental Influence
The rate of deamidation depends on the amino acid sequence. The Asn-Gly (NG) sequence is highly prone to this reaction due to low steric hindrance. Other sequences like Asn-Ser (NS) and Asn-Ala (NA) are also susceptible.
Protein structure also plays a role. Asparagine residues located on the surface or in flexible regions are more likely to undergo deamidation than those buried deep inside the protein.
Quantify deamidation and other modifications with precision: Discover our Peptide Mapping Services for Biosimilars
Molecular Drivers of Methionine and Tryptophan Oxidation in Forced Degradation of Biosimilars
Oxidation is one of the most common changes observed during Forced Degradation of Biosimilars. It can be triggered by light exposure, metal impurities, or oxidizing agents present in formulations. Methionine and tryptophan residues are particularly sensitive, and their modification can affect both the function and stability of the protein.
Methionine to Methionine Sulfoxide Conversion
Methionine contains sulfur, which reacts easily with reactive oxygen species. This reaction forms methionine sulfoxide and increases the mass of the peptide by +15.99 Da.
- Fc Region Sensitivity: In IgG1 antibodies, residues like M252 and M428 are highly prone to oxidation. Changes here can reduce binding to the FcRn receptor, which may shorten the drug’s half-life.
- Impact on CDRs: If oxidation occurs in the antigen-binding region, it may reduce the ability of the antibody to bind effectively, lowering its potency.
Tryptophan Oxidation and the Aggregation Nexus
Tryptophan oxidation leads to several products such as 5-hydroxytryptophan and kynurenine. Under stress conditions like UV exposure, this can increase protein aggregation.
This happens because oxidation changes the polarity of the amino acid, which may cause partial unfolding of the protein. As a result, hidden hydrophobic regions become exposed, promoting unwanted protein interactions.
Deep dive into your molecule’s specific modifications: Learn about Post-Translational Modifications (PTMs) Analysis
LC-MS/MS Peptide Mapping in Forced Degradation of Biosimilars
High-resolution LC-MS/MS peptide mapping is a key technique used during Forced Degradation of Biosimilars to detect and measure structural changes. It allows precise identification of amino acid sequences and modifications by analyzing mass-to-charge ratios.
The Bottom-Up Workflow and Sample Preparation
The bottom-up method includes the following steps:
- Denaturation and Reduction: Proteins are unfolded and disulfide bonds are broken.
- Alkylation: Prevents reformation of disulfide bonds.
- Enzymatic Digestion: Proteins are cut into smaller peptides using enzymes like trypsin.
- LC-MS Analysis: Peptides are separated and analyzed using advanced instruments such as Orbitrap or Q-TOF.
Minimizing Artifacts with Optimized Digestion Parameters
Artificial deamidation can occur during sample preparation, especially at higher pH levels. Optimized conditions help reduce these unwanted changes.
| Digestion Parameter | Conventional Protocol | ResolveMass Optimized Protocol | Impact on Data Quality |
|---|---|---|---|
| Digestion Time | 16–24 Hours (Overnight) | 1–4 Hours | Dramatically reduces artifactual deamidation |
| Digestion pH | 8.0 – 8.5 | 6.0 – 6.5 | Stabilizes asparagine residues during the procedure |
| Enzyme Ratio | 1:50 | 1:10 to 1:20 | Ensures complete cleavage in shorter timeframes |
| Cleanup | Manual Desalting | Automated (AssayMAP) | Increases reproducibility and peptide recovery |
Validate your biosimilarity with industry-leading mass spectrometry: See how to Prove Biosimilarity Using LC-MS
Advanced LC-MS Fragmentation Techniques in Forced Degradation of Biosimilars
Detecting small mass changes, such as the 0.98 Da shift from deamidation, requires high precision tools.
Collision-Induced Dissociation (CID) and HCD
CID and HCD break peptides into smaller fragments, producing ions that help identify modification sites. HCD is especially useful for generating clear and detailed spectra.
Electron Transfer Dissociation (ETD) for Isomer Differentiation
ETD preserves delicate modifications and helps distinguish between similar structures like aspartic acid and isoaspartic acid. It is particularly useful when standard methods are not sufficient.
Get a high-level view of your protein’s integrity: Explore Intact Mass Analysis for Biosimilars
Subunit and Middle-Up Analysis in Forced Degradation of Biosimilars
While peptide mapping provides detailed insights, subunit analysis offers a faster way to compare biosimilars.
IdeS Digestion and Subunit Reduction
The IdeS enzyme cuts antibodies into smaller parts, which are then reduced into:
- Light chain (LC)
- $Fd’$ fragment
- $Fc/2$ subunit
Each part can be directly analyzed using LC-MS, making the process faster and efficient.
This method is useful for:
- Comparing oxidation levels in different regions
- Monitoring structural changes
- Evaluating glycosylation patterns
Ensure your glycosylation profiles match the reference product: Request a Glycosylation Analysis for Biosimilars
Case Study: Infliximab and Bevacizumab Biosimilarity
Studies comparing infliximab and bevacizumab show:
- Similar oxidation patterns in both biosimilars and reference products
- Noticeable oxidation at specific residues affecting binding
- Slightly higher sensitivity to stress in some biosimilar versions
These findings show how Forced Degradation of Biosimilars can reveal subtle differences that may not appear under normal conditions.
Accelerate your generic peptide development: View our Generic Peptide and Semaglutide Project Services
Software and Informatics in Forced Degradation of Biosimilars
Modern data analysis relies on advanced software tools to automate workflows.
Platforms like Waters UNIFI and SCIEX BioPharmaView support:
- Automated sequence verification
- Accurate quantification
- Easy comparison between samples
- Custom quality checks
Data Independent Acquisition ($MS^E$) for Comprehensive Coverage
$MS^E$ captures data for all ions, ensuring that even low-level changes are detected. This makes it highly effective for complete degradation analysis.
Regulatory Alignment: ICH Q5E and Forced Degradation of Biosimilars
The analysis of degradation patterns supports regulatory requirements under ICH Q5E guidelines.
The Role of Tiered Testing
Regulatory evaluation includes:
- Tier 1: Critical attributes like potency
- Tier 2: Modifications such as oxidation and deamidation
- Tier 3: General quality trends
Totality of Evidence (ToE)
Forced Degradation of Biosimilars contributes to the Totality of Evidence by demonstrating that biosimilars behave similarly to their reference products under stress conditions.
Conclusion: Mastering the Complexity of Biosimilar Stability with Forced Degradation of Biosimilars
Developing a biosimilar requires a deep understanding of both structure and function. Forced Degradation of Biosimilars provides a reliable framework to study how proteins respond to stress.
By combining LC-MS/MS analysis, optimized workflows, and subunit strategies, scientists can accurately evaluate key modifications like deamidation and oxidation. These insights support strong regulatory submissions and ensure that biosimilars meet safety and efficacy standards.
As biologics continue to advance, high-quality analytical techniques will remain critical for delivering consistent and reliable therapies.
Ready to start your development journey? Inquire about Formulation Development for Generic Drugs
For more information on advanced characterization services or to discuss a biosimilar program, please contact the technical team:
https://resolvemass.ca/contact/
FAQs: Forced Degradation and LC-MS Mapping
The asparagine-glycine (NG) sequence is very sensitive because glycine is small and does not block the reaction. This allows the protein chain to easily form the intermediate structure needed for deamidation. During Forced Degradation of Biosimilars, especially at higher pH, this site becomes one of the fastest to undergo change and form charge variants.
Methionine residues in key regions of the antibody are important for binding to the FcRn receptor. When these residues are oxidized, their chemical nature changes, which weakens this interaction. As a result, the antibody may be cleared from the body more quickly, reducing its overall effectiveness and duration of action.
An ideal degradation range is usually between 5% and 20% of the main protein peak. This range provides enough breakdown products for proper detection without damaging the molecule too much. Excessive stress can create unrealistic degradation patterns that are not relevant to real storage conditions.
HCD is often chosen because it produces more detailed fragment information across the full mass range. This makes it easier to identify exactly where oxidation has occurred in the peptide. The improved data quality helps accurately locate small mass changes linked to modified amino acids.
Photostability testing exposes proteins to strong light to check for light-sensitive changes. Tryptophan residues in CDR regions can absorb this energy and become oxidized. This may affect how the antibody binds to its target, making it important to confirm stability under light exposure.
The middle-up method breaks antibodies into smaller subunits using specific enzymes and analyzes them directly. It is faster than full peptide mapping and requires less preparation. This approach is useful for quickly checking major changes like oxidation or glycation during Forced Degradation of Biosimilars.
The succinimide intermediate is a key step in the deamidation process. Under high pH conditions, its formation becomes much faster, making it easier to identify sensitive sites. This helps scientists understand which regions of the biosimilar are more likely to change over time.
Reference:
- Yamaci, M. C., et al. (2025). Comparative forced degradation study of anticomplement therapeutic proteins. Pharmaceuticals, 18(4), 579. https://doi.org/10.3390/ph18040579
- Nowak, C., Cheung, J. K., Dellatore, S. M., Katiyar, A., Bhat, R., Sun, J., Ponniah, G., Neill, A., Mason, B., Beck, A., Liu, H., & others. (2017). Forced degradation of recombinant monoclonal antibodies: A practical guide. mAbs, 9(8), 1217–1230. https://doi.org/10.1080/19420862.2017.1368595
- Wang, Y., Li, X., Liu, Y.-H., Richardson, D., Li, H., Shameem, M., & Yang, X. (2016). Simultaneous monitoring of oxidation, deamidation, isomerization, and glycosylation of monoclonal antibodies by liquid chromatography–mass spectrometry method with ultrafast tryptic digestion. mAbs, 8(8), 1477–1486. https://doi.org/10.1080/19420862.2016.1226715
- Sankar, K., Krishnan, S., & Nowak, C. (2018). Prediction of methionine oxidation risk in monoclonal antibodies using structure-based modeling and LC–MS peptide mapping. mAbs, 10(8), 1281–1290. https://doi.org/10.1080/19420862.2018.1518853
- Dyck, Y. F. K., Ståhl, S., & Solà, R. J. (2019). Forced degradation testing as a complementary tool for biosimilar development. Pharmaceuticals, 12(4), 163. https://doi.org/10.3390/ph12040163
- International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH). (1998). ICH Q1B: Photostability testing of new active substances and medicinal products (Step 5). European Medicines Agency. https://www.ema.europa.eu/en/documents/scientific-guideline/ich-q-1-b-photostability-testing-new-active-substances-and-medicinal-products-step-5_en.pdf



