
Introduction:
The Generic pharmaceutical CDMO ecosystem plays a critical role in helping pharmaceutical companies develop and commercialize high-quality generic medicines. Contract Development and Manufacturing Organizations (CDMOs) provide technical expertise, regulatory guidance, and manufacturing capabilities that allow companies to bring affordable medicines to the market faster.
In Canada, generic drug development requires compliance with strict regulatory frameworks governed by Health Canada. Working with an experienced CDMO ensures that pharmaceutical companies can navigate these requirements efficiently while maintaining product quality and regulatory compliance.
This article explores how contract development and manufacturing for generic pharmaceuticals works in Canada, the services provided by CDMOs, and why partnering with the right organization is essential for success.
Share via:
Summary:
- Generic pharmaceutical CDMO Canada services help pharmaceutical companies develop, test, and manufacture generic drugs efficiently while meeting regulatory requirements.
- Canada has a strong regulatory framework through Health Canada, making it a reliable market for generic drug development.
- Partnering with an experienced CDMO reduces development timelines, ensures quality compliance, and supports regulatory submissions.
- Advanced capabilities such as analytical testing, bioanalysis, method validation, and regulatory support are critical for successful generic drug commercialization.
- Companies like ResolveMass Laboratories Inc. provide specialized analytical and bioanalytical services that support CDMO workflows for generic pharmaceutical development.
1: What is a Generic Pharmaceutical CDMO in Canada?
A Generic pharmaceutical CDMO Canada is a specialized organization that supports pharmaceutical companies in developing, testing, and manufacturing generic drugs under contract.
In simple terms, CDMOs allow pharmaceutical companies to outsource complex development and manufacturing processes while focusing on their core business strategies.
Key Services Provided by Generic Pharmaceutical CDMOs
Typical CDMO services for generic drug development include:
- Drug formulation development
- Analytical method development
- Bioanalytical testing
- Stability studies
- Scale-up manufacturing
- Regulatory support and documentation
- Quality control and validation
These services ensure that the generic product meets regulatory standards and is therapeutically equivalent to the reference listed drug.
2: Why the Generic Pharmaceutical CDMO Canada Market is Growing
The Generic pharmaceutical CDMO market is expanding due to increasing demand for cost-effective medicines and the growing number of drug patent expirations.
Major Drivers of Growth
| Driver | Impact |
|---|---|
| Patent Expirations | Creates opportunities for generic drug development |
| Rising Healthcare Costs | Drives demand for affordable medicines |
| Regulatory Requirements | Requires specialized expertise |
| Outsourcing Trends | Pharmaceutical companies prefer specialized partners |
Canada’s strong pharmaceutical ecosystem and regulatory framework make it an attractive destination for generic drug development partnerships. Learn more about CDMO services for generic projects in Canada.
3: Regulatory Framework for Generic Pharmaceuticals in Canada
In Canada, generic drugs must meet the regulatory standards set by Health Canada to ensure safety, quality, and efficacy.
Companies seeking approval must submit an Abbreviated New Drug Submission (ANDS) demonstrating that their product is bioequivalent to the brand-name reference drug.
Key Regulatory Requirements
- Demonstration of bioequivalence
- Validated analytical methods
- Stability studies
- Manufacturing quality compliance
- Good Manufacturing Practice (GMP) certification
A reliable Generic pharmaceutical CDMO Canada partner helps ensure that all regulatory documentation and testing requirements are met before submission.
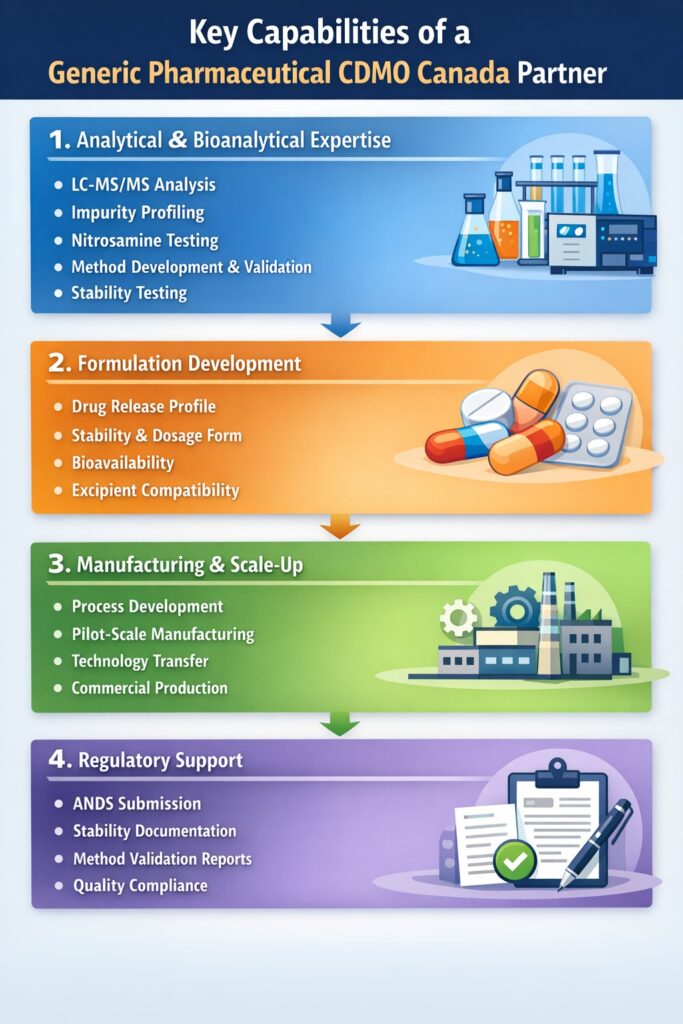
4: Key Capabilities of a Generic Pharmaceutical CDMO Canada Partner
A successful Generic pharmaceutical CDMO Canada partnership depends on strong scientific expertise, advanced analytical capabilities, and deep regulatory understanding. These capabilities ensure that generic drugs are developed efficiently, meet quality standards, and receive regulatory approval in Canada.
Below are the most critical capabilities that pharmaceutical companies should expect from a reliable CDMO partner.
1. Analytical and Bioanalytical Expertise
Analytical and bioanalytical testing ensures that a generic drug meets quality standards and demonstrates equivalence to the reference product. It is one of the most important capabilities of a Generic pharmaceutical CDMO Canada provider.
Analytical testing supports every stage of drug development—from early formulation work to regulatory submissions.
Typical analytical capabilities include:
- LC-MS/MS analysis
- Impurity profiling and characterization
- Nitrosamine impurity testing
- Analytical method development
- Method validation
- Stability-indicating analytical methods
Advanced analytical laboratories such as ResolveMass Laboratories Inc. specialize in mass spectrometry-based analytical services that support pharmaceutical CDMO programs and regulatory submissions.
Advanced labs like ResolveMass Laboratories Inc. provide bioanalytical strategy services to support CDMO programs.
These services generate the reliable data required for demonstrating drug safety, quality, and regulatory compliance.
2. Formulation Development
Formulation development focuses on designing a generic drug product that matches the reference listed drug in performance and therapeutic effect.
In a Generic pharmaceutical CDMO Canada program, formulation scientists work to replicate the original drug’s performance characteristics while maintaining stability and manufacturability.
Key formulation considerations include:
- Drug release profile
- Physical and chemical stability
- Dosage form design
- Bioavailability
- Excipient compatibility
The goal is to ensure therapeutic equivalence, meaning the generic drug delivers the same clinical effect as the original product.
They may also leverage peptide testing and characterization services and peptide characterization for complex molecules.
Proper formulation development also helps minimize regulatory risks during the approval process.
3. Manufacturing and Scale-Up
Manufacturing and scale-up capabilities allow a generic drug to move from laboratory development to commercial production.
An experienced Generic pharmaceutical CDMO Canada partner provides expertise in scaling manufacturing processes while maintaining product quality and consistency.
Typical manufacturing support includes:
- Process development and optimization
- Pilot-scale manufacturing
- Technology transfer
- Commercial-scale production
These processes must comply with Good Manufacturing Practices (GMP) as required by Health Canada and international regulatory agencies.
Compliance with GMP ensures consistent, high-quality batches. Outsourced chemistry in drug discovery can also support scale-up activities for specialized compounds.
A strong CDMO ensures that manufacturing processes are scalable, efficient, and capable of producing consistent product batches.
4. Regulatory Support
Regulatory support is essential for navigating the complex approval pathway for generic drugs in Canada.
A reliable Generic pharmaceutical CDMO provider assists pharmaceutical companies in preparing the technical and regulatory documentation required for approval.
Key regulatory support activities include:
- Abbreviated New Drug Submission (ANDS) preparation
- Analytical method validation reports
- Stability study documentation
- Quality and compliance documentation
Regulatory expertise significantly reduces the risk of submission delays and helps companies meet the requirements of Health Canada.
By combining scientific expertise with regulatory knowledge, CDMOs enable pharmaceutical companies to bring high-quality generic medicines to the Canadian market more efficiently.
5: Benefits of Working with a Generic Pharmaceutical CDMO Canada Partner
Partnering with an experienced CDMO offers several strategic advantages for pharmaceutical companies.
Faster Time to Market
Outsourcing development activities allows companies to accelerate drug development timelines.
Access to Specialized Expertise
CDMOs provide multidisciplinary teams including:
- Analytical chemists
- Formulation scientists
- Regulatory experts
- Quality assurance specialists
Cost Efficiency
Developing internal capabilities can be expensive. CDMOs provide cost-effective access to specialized infrastructure.
Regulatory Compliance
Experienced CDMOs ensure that all development and manufacturing processes comply with Canadian and international regulations.
6: How ResolveMass Laboratories Supports Generic Pharmaceutical CDMO Canada Projects
ResolveMass Laboratories Inc. provides advanced analytical and bioanalytical solutions that support contract development and manufacturing programs for generic pharmaceuticals.
Key Expertise Areas
ResolveMass offers specialized services such as:
- LC-MS/MS bioanalysis
- Impurity identification
- Nitrosamine impurity testing
- Bioanalytical method validation
- Pharmacokinetic bioanalysis
These capabilities play a critical role in ensuring that generic drugs meet regulatory requirements for quality, safety, and bioequivalence.
These services ensure compliance and support regulatory submissions. Learn more about bioanalysis in Canada.
The laboratory’s scientific expertise and advanced instrumentation help pharmaceutical companies generate high-quality analytical data required for regulatory submissions.
7: Choosing the Right Generic Pharmaceutical CDMO Canada Partner
Selecting the right CDMO is one of the most important decisions in generic drug development.
Key Factors to Consider
| Factor | Why It Matters |
|---|---|
| Regulatory experience | Ensures smooth ANDS approval |
| Analytical capabilities | Supports bioequivalence studies |
| Manufacturing expertise | Enables scalable production |
| Quality systems | Ensures regulatory compliance |
| Scientific expertise | Improves development efficiency |
Check out CDMO for generic drug development in Canada for examples of expert partners.
A reliable Generic pharmaceutical CDMO Canada partner will demonstrate strong technical expertise, regulatory knowledge, and a proven track record in pharmaceutical development.
8: Future Trends in Generic Pharmaceutical CDMO Canada Services
The Canadian CDMO sector is evolving as pharmaceutical development becomes more complex.
Emerging Trends
- Increased demand for advanced analytical services
- Growth in complex generics
- Expansion of biologics and biosimilars
- Integration of AI-driven drug development tools
These trends highlight the importance of partnering with scientifically advanced laboratories and CDMOs capable of supporting modern pharmaceutical development.
Conclusion:
The Generic pharmaceutical CDMO Canada sector enables pharmaceutical companies to develop and manufacture high-quality generic medicines efficiently. Partners like ResolveMass Laboratories Inc. provide advanced analytical, bioanalytical, and regulatory support, helping companies meet Health Canada requirements and bring affordable medicines to market.
By partnering with experienced organizations, companies gain access to advanced scientific expertise, regulatory guidance, and manufacturing capabilities that accelerate drug development and ensure compliance with Health Canada standards.
Organizations such as ResolveMass Laboratories Inc. contribute to this ecosystem by providing specialized analytical and bioanalytical services that support generic drug development and regulatory submissions.
As demand for affordable medicines continues to grow, the role of a reliable Generic pharmaceutical CDMO Canada partner will become even more critical for pharmaceutical companies aiming to successfully launch generic drugs in the Canadian market.
Frequently Asked Questions:
A Generic pharmaceutical CDMO Canada is a contract development and manufacturing organization that helps pharmaceutical companies develop, test, and manufacture generic drugs while meeting Canadian regulatory requirements. These organizations provide services such as formulation development, analytical testing, manufacturing, and regulatory support.
A Generic pharmaceutical CDMO Canada typically provides a wide range of services including:
-Drug formulation development
-Analytical method development and validation
-Bioanalytical testing
-Stability studies
-Process development and scale-up
-Commercial manufacturing
-Regulatory documentation and submissions
These services support the entire lifecycle of generic drug development.
A Generic pharmaceutical CDMO Canada assists pharmaceutical companies by preparing regulatory documentation such as:
-Analytical method validation reports
-Stability study reports
-Quality documentation
-Bioequivalence data
-ANDS submission materials
These services help streamline the regulatory approval process.
The timeline for generic drug development can vary depending on product complexity, regulatory requirements, and analytical testing needs. With an experienced Generic pharmaceutical CDMO Canada partner, development timelines can typically range from 18 to 36 months before regulatory approval.
When selecting a Generic pharmaceutical CDMO Canada provider, companies should evaluate:
-Regulatory experience with Health Canada
-Analytical and bioanalytical capabilities
-Manufacturing expertise
-Quality systems and GMP compliance
-Proven track record in generic drug development
Choosing the right partner can significantly impact development success and approval timelines.
Reference
- Reed F. Beall , Lorian Hardcastle , Fiona Clement , Aidan Hollis. How will recent trade agreements that extend market protections for brand-name prescription pharmaceuticals impact expenditures and generic access in Canada?https://www.sciencedirect.com/science/article/abs/pii/S0168851019302258
- Regulation of Pharmaceuticals in Canada.https://papers.ssrn.com/sol3/papers.cfm?abstract_id=958929
- Steven G. Morgan , Paige A. Thomson , Jamie R. Daw , Melissa K. Friesen. Canadian policy makers’ views on pharmaceutical reimbursement contracts involving confidential discounts from drug manufacturers.https://www.sciencedirect.com/science/article/pii/S0168851013001607
- Pharmaceutical security for Canada. https://www.cmaj.ca/content/194/32/E1113.short
- Managing pharmaceutical expenditure: an overview and options for Canada.https://utoronto.scholaris.ca/items/7d8b1541-4818-45b5-b5c5-4baad8195dce


