
Introduction:
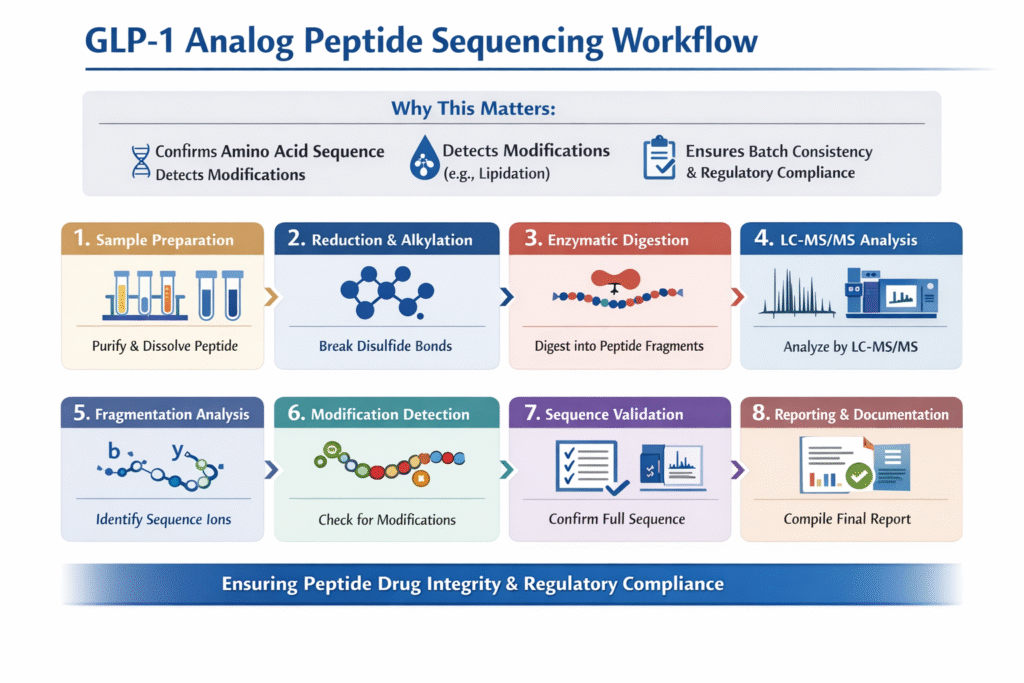
The GLP-1 Analog Peptide Sequencing Workflow is a critical analytical process used to confirm the structural integrity and sequence accuracy of therapeutic peptides such as Semaglutide and Liraglutide. These GLP-1 analogs are widely used in the treatment of type 2 diabetes and obesity, making their precise characterization essential for regulatory approval and product quality.
At ResolveMass Laboratories Inc., advanced mass spectrometry-based workflows—such as peptide sequencing of GLP-1 peptide and LC-MS characterization of GLP-1 peptides—are employed to ensure highly accurate peptide sequencing aligned with global regulatory expectations. This article provides a detailed, step-by-step workflow that reflects industry best practices and scientific rigor.
Share via:
Summary:
- GLP-1 Analog Peptide Sequencing Workflow involves sample preparation, enzymatic digestion, LC-MS/MS analysis, and data interpretation.
- Accurate sequencing ensures identity, purity, and regulatory compliance for peptides like Semaglutide and Liraglutide.
- Key analytical tools include high-resolution LC-MS/MS, peptide mapping, and fragmentation analysis as part of analytical characterization of GLP-1 peptide drugs.
- Challenges include post-translational modifications (PTMs), fatty acid conjugation, and sequence complexity.
- A structured workflow improves reproducibility, regulatory acceptance, and product quality.
1: What is GLP-1 Analog Peptide Sequencing Workflow?
The GLP-1 Analog Peptide Sequencing Workflow is a systematic analytical approach used to determine the amino acid sequence and structural modifications of GLP-1 analogs.
In simple terms: It confirms that the peptide drug is exactly what it is supposed to be—structurally and chemically.
This workflow is a key component of GLP-1 peptide sequencing CRO services and broader CRO for GLP-1 peptide characterization solutions.
Why It Matters
- Confirms primary structure (amino acid sequence)
- Detects modifications (e.g., lipidation in Semaglutide)
- Ensures batch-to-batch consistency
- Supports regulatory submissions (FDA, EMA), aligned with regulatory requirements for GLP-1 peptide characterization
Step 1: Sample Preparation
Proper sample preparation ensures the peptide is clean, stable, and suitable for high-precision analysis.
Key Activities
- Dissolution in appropriate solvents (e.g., water, acetonitrile)
- Desalting to remove buffer salts
- Concentration adjustment
- Filtration to remove particulates
Best Practices
- Maintain low temperature conditions to prevent degradation
- Avoid repeated freeze-thaw cycles
- Use LC-MS grade reagents
Step 2: Reduction and Alkylation (If Required)
This step breaks disulfide bonds and stabilizes cysteine residues for accurate sequencing.
Why It’s Important
- Ensures better enzymatic digestion
- Improves peptide fragmentation patterns
Typical Process
- Reduction using DTT (Dithiothreitol)
- Alkylation using Iodoacetamide (IAA)
Outcome
- Linearized peptide structure for better analysis
Step 3: Enzymatic Digestion (Peptide Mapping)
Enzymatic digestion breaks the peptide into smaller fragments for easier sequencing.
This step is essential in peptide sequencing of GLP-1 drugs workflows.
Common Enzymes Used
- Trypsin
- Chymotrypsin
- Glu-C
Why Multiple Enzymes?
GLP-1 analogs like Semaglutide have complex modifications, so multiple enzymes ensure:
- Complete sequence coverage
- Overlapping peptide fragments
Key Output
- Peptide fragments suitable for LC-MS/MS analysis
Step 4: LC-MS/MS Analysis
LC-MS/MS separates and identifies peptide fragments based on mass and fragmentation patterns.
This step is central to LC-MS characterization of GLP-1 peptides.
Workflow Breakdown
- Liquid Chromatography (LC):
- Separates peptide fragments
- Mass Spectrometry (MS):
- Measures molecular mass
- MS/MS (Tandem MS):
- Provides fragmentation data for sequencing
Critical Parameters
- High-resolution instruments (e.g., Orbitrap, Q-TOF)
- Optimized collision energy
- Accurate mass calibration
Step 5: Data Acquisition and Fragmentation Analysis
Fragmentation patterns are analyzed to reconstruct the peptide sequence.
Types of Ions Observed
- b-ions (N-terminal fragments)
- y-ions (C-terminal fragments)
What Analysts Look For
- Complete sequence coverage
- Consistency with theoretical sequence
- Identification of modifications
Step 6: Identification of Modifications
Detecting modifications is essential for GLP-1 analogs due to their structural complexity and is a major part of GLP-1 peptide impurity characterization.
Common Modifications
- Fatty acid conjugation (e.g., Semaglutide)
- PEGylation or linker molecules
- Oxidation or deamidation
Why This Step is Critical
- Impacts drug efficacy and stability
- Required for regulatory documentation
- Supports GLP-1 peptide stability analytical methods
Step 7: Sequence Confirmation and Validation
The final sequence is verified against the reference standard to ensure accuracy.
Validation Criteria
- ≥ 95–100% sequence coverage
- Accurate mass matching
- Reproducible results
Supporting Techniques
- Peptide mapping comparison
- Orthogonal methods (e.g., NMR if needed)
Step 8: Reporting and Documentation
Comprehensive reporting ensures regulatory compliance and data transparency.
Typical Report Includes
- Experimental conditions
- Chromatograms and spectra
- Sequence coverage maps
- Identified modifications
Regulatory Alignment
- ICH Q6B
- FDA peptide characterization guidelines
- Supports analytical characterization of GLP-1 peptide drugs
2: Challenges in GLP-1 Analog Peptide Sequencing Workflow
The GLP-1 Analog Peptide Sequencing Workflow faces challenges primarily due to structural complexity, chemical modifications, and the need for highly sensitive and precise analytical techniques.
Key Challenges
1. Lipidated Side Chains (e.g., Semaglutide)
GLP-1 analogs such as Semaglutide are chemically modified with fatty acid side chains to extend their half-life. While therapeutically beneficial, these lipidations:
- Increase molecular complexity
- Affect ionization efficiency in LC-MS/MS
- Complicate fragmentation patterns, making sequence interpretation difficult
2. Incomplete Digestion
Enzymatic digestion is a critical step in peptide sequencing, but:
- Structural hindrance and modifications can prevent complete cleavage
- Leads to larger, unexpected peptide fragments
- Reduces overall sequence coverage and confidence in results
3. Low-Abundance Impurities
GLP-1 analog samples may contain trace-level impurities that:
- Are difficult to detect due to low concentration
- Require high-resolution and high-sensitivity instruments
- Are crucial for regulatory compliance and safety evaluation
4. Co-eluting Fragments
During chromatographic separation:
- Some peptide fragments may elute at the same time
- This causes overlapping peaks in LC-MS spectra
- Makes accurate identification and sequence reconstruction more complex
Advanced High-Resolution MS Platforms
- Use of cutting-edge instruments like Orbitrap and Q-TOF
- Enables precise mass detection and improved impurity profiling
- Helps resolve co-eluting and low-abundance species
3: Best Practices for Accurate Sequencing
In the GLP-1 Analog Peptide Sequencing Workflow, following optimized protocols and leveraging advanced instrumentation are essential to achieve accurate, reproducible, and regulatory-compliant results.
Key Recommendations
1. Use Multiple Enzymes for Better Coverage
- Employ enzymes such as Trypsin, Glu-C, and Chymotrypsin
- Generates overlapping peptide fragments, improving sequence confidence
- Helps overcome challenges from structural modifications and steric hindrance
2. Validate Results with Orthogonal Techniques
- Complement LC-MS/MS data with additional analytical approaches
- Techniques may include:
- NMR spectroscopy
- Amino acid analysis
- Peptide mapping comparisons
- Ensures independent confirmation of sequence and structure
3. Maintain Strict Quality Control Procedures
- Use LC-MS grade reagents and calibrated instruments
- Implement system suitability tests and method validation
- Monitor parameters like:
- Mass accuracy
- Retention time reproducibility
- Signal sensitivity
- Ensures data integrity and regulatory acceptance
4. Use Experienced Analytical Teams
- Skilled scientists can:
- Interpret complex fragmentation patterns
- Identify subtle modifications (e.g., lipidation, oxidation)
- Troubleshoot issues like co-elution or incomplete digestion
- Expertise is critical for high-confidence sequencing and reporting
Why These Best Practices Matter
Adopting these best practices in the GLP-1 Analog Peptide Sequencing Workflow leads to:
- Higher sequence accuracy and coverage
- Improved reproducibility across batches
- Reduced analytical errors and rework
- Stronger regulatory submissions (FDA, ICH compliance)
4: Why Choose ResolveMass Laboratories Inc.?
ResolveMass Laboratories Inc. brings deep expertise in peptide characterization and GLP-1 analog analysis.
What Sets Us Apart
- Specialized in complex peptide sequencing
- Advanced LC-MS/MS platforms
- Strong regulatory compliance expertise
- Proven experience with Semaglutide & Liraglutide workflows
- Comprehensive services including GLP-1 peptide sequencing CRO services
Conclusion:
The GLP-1 Analog Peptide Sequencing Workflow is a multi-step, highly specialized process essential for ensuring the safety, efficacy, and regulatory compliance of peptide therapeutics like Semaglutide and Liraglutide.
From sample preparation to advanced LC-MS/MS analysis and final validation, each step plays a crucial role in delivering accurate and reliable sequencing results. By integrating approaches such as peptide sequencing of GLP-1 peptide and analytical characterization of GLP-1 peptide drugs, organizations can confidently meet global regulatory expectations.
ResolveMass Laboratories Inc. stands as a trusted partner in delivering precise, reproducible, and regulatory-ready peptide sequencing solutions.
Frequently Asked Questions:
GLP-1 Analog Peptide Sequencing Workflow is an analytical process used to determine the amino acid sequence and structural modifications of GLP-1 peptides. It ensures that drugs like Semaglutide and Liraglutide match their intended structure. The workflow typically includes sample preparation, enzymatic digestion, LC-MS/MS analysis, and data interpretation. It is essential for confirming identity, purity, and consistency. This process plays a critical role in regulatory submissions and quality control.
LC-MS/MS is the core analytical technique used for peptide sequencing due to its high sensitivity and accuracy. It separates peptide fragments and provides detailed mass and fragmentation data. This allows scientists to reconstruct the full amino acid sequence. It also helps identify impurities and structural modifications. Without LC-MS/MS, achieving high-confidence sequencing would be difficult.
GLP-1 peptide sequencing involves challenges such as lipidation, complex structures, and incomplete enzymatic digestion. Modifications like fatty acid conjugation can affect ionization and fragmentation patterns. Low-level impurities may be difficult to detect without high-resolution instruments. Co-eluting peaks can complicate data interpretation. These factors require advanced techniques and experienced analysts.
Common modifications include fatty acid conjugation, PEGylation, oxidation, and deamidation. These modifications are often intentionally introduced to improve drug stability and half-life. However, they increase analytical complexity during sequencing. Identifying and confirming these changes is essential for regulatory compliance. Accurate detection ensures drug safety and efficacy.
Enzymatic digestion breaks large peptides into smaller, manageable fragments for analysis. Enzymes like Trypsin, Glu-C, and Chymotrypsin are commonly used. Using multiple enzymes helps generate overlapping fragments for better sequence coverage. This improves confidence in sequence reconstruction. It is a critical step in peptide mapping and LC-MS/MS workflows.
Reference
- Martin ME. Anesthetic Guidelines for Patients on Glucagon-Like Peptide Receptor Agonists (Doctoral dissertation, The University of Arizona).https://search.proquest.com/openview/c530368f00bd99716ab4294cd7e533a7/1?pq-origsite=gscholar&cbl=18750&diss=y
- Apostolov A, Pathare AD, Lavogina D, Zhao C, Kask K, Blanco Rodriguez L, Ruiz-Duran S, Risal S, Rooda I, Damdimopoulou P, Saare M. Semaglutide alters the human embryo-endometrium interface. medRxiv. 2026:2026-03.https://www.medrxiv.org/content/10.64898/2026.03.03.26347354.abstract
- Zhou J, Zheng Y, Xu B, Long S, Zhu LE, Liu Y, Li C, Zhang Y, Liu M, Wu X. Exploration of the potential association between GLP-1 receptor agonists and suicidal or self-injurious behaviors: a pharmacovigilance study based on the FDA Adverse Event Reporting System database. BMC medicine. 2024 Feb 14;22(1):65.https://link.springer.com/article/10.1186/s12916-024-03274-6


