
Introduction:
GLP-1 impurity peptide mapping is a powerful analytical approach used to detect and characterize impurities and degradation pathways in GLP-1 peptides at a molecular level. Advanced techniques such as peptide mapping of GLP-1 peptides and LC-MS characterization of GLP-1 peptides enable highly accurate impurity profiling.
Glucagon-like peptide-1 (GLP-1) analogs are widely used in the treatment of type 2 diabetes and obesity. Due to their peptide nature, they are highly susceptible to chemical and physical degradation, making impurity profiling critical during development and quality control. Stability considerations are further explained in GLP-1 peptide stability analytical methods.
Regulatory authorities require comprehensive impurity characterization, and peptide mapping via LC-MS/MS has become the gold standard for this purpose, as outlined in regulatory requirements for GLP-1 peptide characterization.
Share via:
Summary:
- GLP-1 impurity peptide mapping is a highly sensitive LC-MS/MS-based technique used to detect, identify, and quantify impurities and degradation products in GLP-1 peptides.
- It enables site-specific characterization of sequence variants, oxidation, deamidation, and truncations, similar to approaches described in GLP-1 sequence variant analysis.
- This case study demonstrates how peptide mapping helps identify root causes of instability in GLP-1 formulations.
- The approach ensures regulatory compliance, product safety, and batch consistency.
- Advanced workflows such as HRMS peptide mapping GLP-1 improve accuracy, reproducibility, and detection limits.
1: What is GLP-1 Impurity Peptide Mapping?
GLP-1 impurity peptide mapping is an LC-MS/MS-based analytical technique that enzymatically digests GLP-1 peptides into smaller fragments and analyzes them to identify impurities and degradation products with high precision.
This method is closely related to GLP-1 impurity characterization and GLP-1 impurity sequencing analysis, providing detailed, site-specific structural insights.
Key Features:
- High-resolution identification of sequence variants
Detects even minor amino acid substitutions or sequence mismatches. - Detection of post-translational modifications (PTMs)
Identifies changes such as oxidation, deamidation, and glycation. - Quantification of low-level impurities
Enables accurate measurement of trace impurities at very low concentrations. - Site-specific degradation analysis
Pinpoints the exact location of chemical or structural degradation within the peptide.
2: Case Study Overview
This case study evaluates impurity formation and degradation pathways in a GLP-1 peptide under stress conditions using peptide mapping to generate detailed, site-specific insights.
The study aligns with workflows described in GLP-1 analog peptide sequencing workflow and supports outsourcing strategies such as CRO for GLP-1 peptide characterization.
Objective:
- Identify and characterize impurities and degradation products
Detect both known and unknown impurities formed during stress exposure. - Understand stability profile under stress conditions
Assess how environmental factors impact peptide integrity and structure. - Support method validation and regulatory submission
Generate reliable data aligned with ICH and FDA expectations.
Sample Details:
- GLP-1 analog formulation
- Tested under the following stress conditions:
- Thermal stress (40°C) → Evaluates heat-induced degradation
- Oxidative conditions → Assesses susceptibility to oxidation (e.g., methionine oxidation)
- pH variation → Examines stability across acidic and basic environments
This structured approach ensures comprehensive impurity profiling and provides critical insights into degradation mechanisms, product stability, and quality control strategies for GLP-1 therapeutics.
3: Analytical Approach
Peptide mapping using LC-MS/MS enables detailed impurity profiling by enzymatically breaking GLP-1 peptides into smaller fragments and analyzing them with high accuracy and sensitivity.
This approach is supported by advanced techniques like:
For comparison with alternative methods, see GLP-1 peptide mapping vs intact mass.
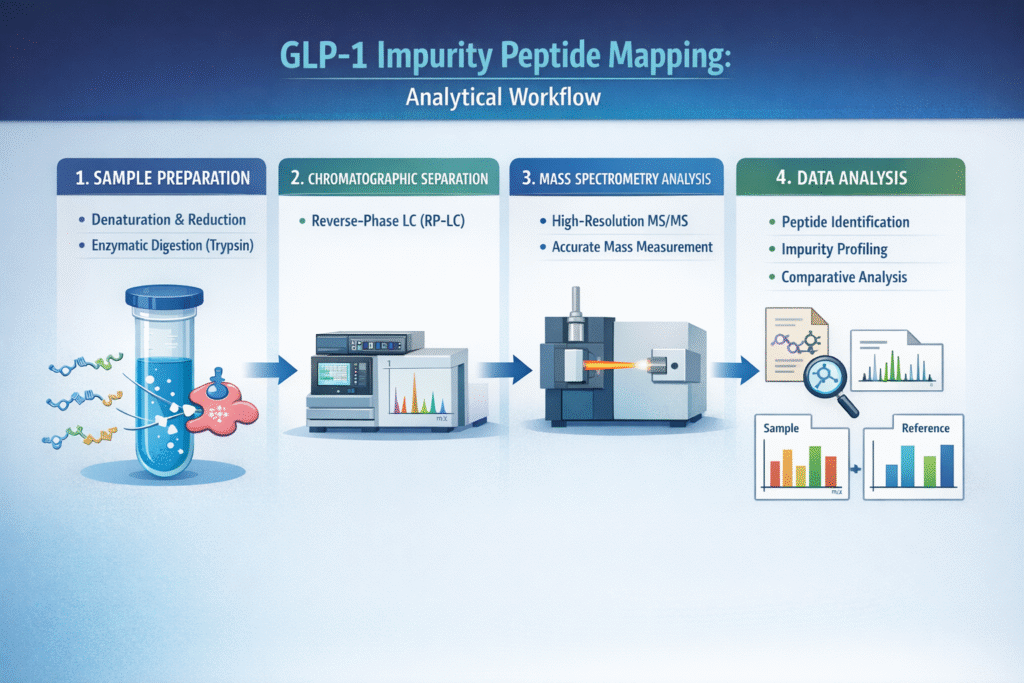
Workflow Overview
1. Sample Preparation
Proper sample preparation ensures accurate and reproducible peptide mapping results.
- Denaturation and reduction
Unfolds the peptide structure and breaks disulfide bonds (if present), improving enzyme accessibility. - Enzymatic digestion (Trypsin)
Cleaves the peptide at specific amino acid residues to generate predictable fragment peptides for analysis.
2. Chromatographic Separation
Reverse-phase liquid chromatography (RP-LC) separates peptide fragments based on hydrophobicity before MS detection.
- High-resolution separation minimizes co-elution
- Improves detection of low-level impurities
- Enhances reproducibility and peak resolution
3. Mass Spectrometry Analysis
High-resolution MS/MS enables precise identification and quantification of peptide fragments and impurities.
- High-resolution MS/MS detection
Provides detailed fragmentation patterns for sequence confirmation - Accurate mass measurement
Ensures high confidence in identifying modifications such as oxidation or deamidation
4. Data Analysis
Advanced data processing tools convert raw MS data into actionable impurity profiles.
- Peptide identification
Matches observed fragments with theoretical peptide sequences - Impurity profiling
Detects and characterizes degradation products and sequence variants - Comparative analysis with reference standard
Highlights deviations from the expected peptide structure
This analytical workflow ensures robust, reproducible, and regulatory-compliant impurity characterization, making it a cornerstone of GLP-1 impurity peptide mapping studies.
4: Results: Impurity and Degradation Identification
GLP-1 impurity peptide mapping successfully identified multiple degradation pathways and impurities at trace levels with high sensitivity and site-specific accuracy.
Detailed sequencing insights can also be explored in:
For higher accuracy, methods such as de novo GLP-1 peptide sequencing accuracy are often applied.
The LC-MS/MS-based peptide mapping approach enabled precise detection of structural modifications, providing critical insights into peptide stability and degradation mechanisms.
Key Findings
1. Oxidation
Methionine oxidation was clearly identified and significantly increased under oxidative stress conditions.
- Oxidation primarily occurred at methionine residues
- Detected as a mass shift (+16 Da) in peptide fragments
- Indicates susceptibility of GLP-1 peptides to oxidative environments
2. Deamidation
Deamidation of asparagine residues was observed, leading to conversion into aspartic acid.
- Site-specific modification detected in target peptide fragments
- Causes structural and charge alterations
- Common under pH stress and prolonged storage conditions
3. Peptide Truncation
Both N-terminal and C-terminal truncations were identified, impacting peptide integrity.
- Results from enzymatic cleavage or chemical instability
- Leads to loss of functional activity
- Detected through missing or shortened peptide fragments
4. Aggregation-Related Modifications
Minor cross-linked and aggregated species were detected at low levels.
- Indicates early-stage aggregation pathways
- May impact safety, immunogenicity, and efficacy
- Requires monitoring during formulation and storage

Table: Identified Impurities and their Characteristics
| Impurity Type | Modification Site | Detection Method | Impact on Stability |
|---|---|---|---|
| Oxidation | Methionine | LC-MS/MS | Reduces potency |
| Deamidation | Asparagine | Peptide mapping | Alters structure |
| Truncation | N/C terminal | HRMS | Affects efficacy |
| Aggregation | Multiple sites | MS/MS | Impacts safety |
5: Why Peptide Mapping is Critical for GLP-1 Analysis
Peptide mapping is critical for GLP-1 analysis because it provides precise, site-specific identification of impurities and degradation products that cannot be achieved using intact mass analysis alone.
Unlike bulk mass measurements, peptide mapping breaks the molecule into defined fragments, enabling detailed structural characterization at the amino acid level, which is essential for ensuring drug quality and compliance.
Advantages:
- High sensitivity for low-level impurities
- Structural insights at amino acid level
- Regulatory acceptance (ICH, FDA)
- Reproducibility and robustness
For deeper technical understanding, refer to:
Comparison: Peptide Mapping VS Traditional Methods
| Parameter | Peptide Mapping | Traditional Methods |
|---|---|---|
| Resolution | High | Moderate |
| Specificity | Site-specific | Limited |
| Sensitivity | Very high | Moderate |
| Impurity Identification | Detailed | Partial |
| Regulatory Acceptance | High | Moderate |
6: Regulatory Significance
GLP-1 impurity peptide mapping is critical for regulatory submissions because it ensures comprehensive impurity characterization, method validation, and compliance with global guidelines.
Regulatory agencies require detailed analytical evidence demonstrating the identity, purity, and stability of peptide therapeutics, and peptide mapping provides the level of detail needed to meet these expectations.
Key Guidelines
- ICH Q6B (Biotechnology Products)
Defines specifications for identity, purity, and impurities in peptide and biologic products - ICH Q2 (Analytical Validation)
Establishes validation parameters such as accuracy, precision, specificity, and robustness - FDA Peptide Characterization Requirements
Emphasize comprehensive structural analysis and impurity profiling for drug approval
Compliance Benefits
1. Ensures Product Quality and Safety
- Detects and characterizes critical impurities and degradation products
- Minimizes risks related to efficacy and immunogenicity
2. Supports Batch Release and Stability Studies
- Enables consistent quality assessment across batches
- Provides reliable data for stability testing and shelf-life determination
3. Meets Global Regulatory Expectations
- Aligns with international regulatory frameworks
- Facilitates smooth approval processes across multiple regions
More details can be found in:
7: Challenges and Solutions
Although GLP-1 impurity peptide mapping is highly powerful, it requires careful optimization to ensure accurate, reproducible, and sensitive impurity detection.
Due to the complexity of peptide structures and LC-MS/MS datasets, several analytical and operational challenges can arise during method development and routine analysis.
Outsourcing to expert providers such as GLP-1 peptide sequencing CRO services or outsource GLP-1 peptide sequencing services can help overcome these limitations.
Common Challenges:
1. Complex Data Interpretation
- Large datasets generated from LC-MS/MS analysis
- Overlapping peptide signals and fragmentation patterns
- Difficulty in distinguishing closely related impurities and modifications
2. Sample Preparation Variability
- Inconsistent digestion efficiency (e.g., trypsin activity)
- Variations in denaturation and reduction steps
- Risk of introducing artificial modifications during preparation
3. Low-Abundance Impurity Detection
- Trace-level impurities may be below detection limits
- Signal suppression due to complex sample matrices
- Challenges in achieving high sensitivity and accuracy
Solutions:
1. Use of High-Resolution Mass Spectrometry (HRMS)
- Provides accurate mass measurements and high sensitivity
- Improves detection of low-level impurities and minor variants
- Enhances confidence in impurity identification
2. Advanced Bioinformatics Tools
- Automated peptide identification and mapping software
- Improved data processing and interpretation
- Enables rapid and reliable impurity profiling
3. Robust Method Validation Protocols
- Standardization of sample preparation procedures
- Validation of parameters such as precision, accuracy, and reproducibility
- Ensures consistent performance across different batches and labs
8: Key Takeaways from the Case Study
GLP-1 impurity peptide mapping provides critical insights into degradation pathways, enabling precise impurity characterization and supporting regulatory compliance.
Key Takeaways
- GLP-1 peptides are highly prone to oxidation, deamidation, and truncation
These degradation pathways significantly impact stability, potency, and safety. - GLP-1 impurity peptide mapping provides unmatched detail in impurity profiling
Enables site-specific identification of even low-level impurities. - The method helps identify root causes of degradation
Supports formulation optimization and improved product stability strategies. - Essential for drug development, quality control, and regulatory approval
Ensures compliance with ICH and FDA guidelines while maintaining product consistency.
Specialized applications such as semaglutide peptide mapping further demonstrate the importance of this technique.
Conclusion:
GLP-1 impurity peptide mapping is the most reliable and comprehensive approach for analyzing impurities and degradation products in GLP-1 peptides.
This case study highlights how advanced LC-MS/MS-based peptide mapping enables precise identification, quantification, and characterization of impurities, ensuring product quality and regulatory compliance.
As GLP-1 therapies continue to grow in importance, adopting robust analytical strategies like peptide mapping is essential for ensuring safety, efficacy, and consistency.
Frequently Asked Questions:
GLP-1 impurity peptide mapping is an LC-MS/MS-based analytical technique used to identify and characterize impurities and degradation products in GLP-1 peptides. It works by enzymatically digesting the peptide into smaller fragments and analyzing them with high precision. This method provides site-specific information about structural changes. It is widely used in pharmaceutical development and quality control. It also supports regulatory compliance by offering detailed impurity profiling.
Peptide mapping is important because GLP-1 peptides are highly susceptible to degradation such as oxidation and deamidation. This technique provides detailed insights at the amino acid level, which cannot be achieved with intact mass analysis alone. It helps identify impurities accurately and supports stability studies. Additionally, it ensures product safety, efficacy, and consistency. Regulatory agencies also expect such detailed characterization.
GLP-1 impurity peptide mapping can detect various impurities including oxidation (e.g., methionine oxidation), deamidation (asparagine to aspartic acid), peptide truncations, and aggregation-related modifications. It can also identify sequence variants and unknown degradation products. The method is highly sensitive and capable of detecting impurities at trace levels. This makes it ideal for comprehensive impurity profiling.
LC-MS/MS combines chromatographic separation with mass detection to analyze peptide fragments. LC separates components based on their properties, while MS/MS provides structural information through fragmentation patterns. This allows accurate identification and quantification of impurities. High-resolution MS improves sensitivity and accuracy. It is considered a gold standard for peptide characterization.
The most common degradation pathways include oxidation, deamidation, and peptide truncation. Oxidation typically affects methionine residues, while deamidation occurs at asparagine sites. Truncation can occur at the N- or C-terminal ends due to chemical or enzymatic processes. These changes can impact the stability and efficacy of the peptide. Monitoring these pathways is essential for product quality.
Reference
- Staby A, Steensgaard DB, Haselmann KF, Marino JS, Bartholdy C, Videbæk N, Schelde O, Bosch-Traberg H, Spang LT, Asgreen DJ. Influence of Production Process and Scale on Quality of Polypeptide Drugs: a Case Study on GLP-1 Analogs: Staby et al. Pharmaceutical Research. 2020 Jul;37(7):120.https://link.springer.com/article/10.1007/s11095-020-02817-9
- Hach M, Engelund DK, Mysling S, Mogensen JE, Schelde O, Haselmann KF, Lamberth K, Vilhelmsen TK, Malmstrøm J, Højlys-Larsen KB, Rasmussen TS. Impact of manufacturing process and compounding on properties and quality of follow-On GLP-1 polypeptide drugs. Pharmaceutical Research. 2024 Oct;41(10):1991-2014.https://link.springer.com/article/10.1007/s11095-024-03771-6
- Konduru N, Badam SK, Thamira R, Battula SR, Gundla R. Development and Validation of a Robust Reversed-Phase LC Method for Liraglutide (GLP-1 analogue) and Its Impurities in Injectable Formulations Using a Quality by Design Approach. Available at SSRN 5511143.https://papers.ssrn.com/sol3/papers.cfm?abstract_id=5511143
- Badgujar D, Bawake S, Sharma N. A comprehensive study on the identification and characterization of major degradation products of synthetic liraglutide using liquid chromatography‐high resolution mass spectrometry. Journal of Peptide Science. 2025 Jan;31(1):e3652.https://onlinelibrary.wiley.com/doi/abs/10.1002/psc.3652


