Introduction to GLP-1 Peptide Characterization Mapping
GLP-1 Peptide Characterization Mapping is an essential scientific approach used to confirm that generic GLP-1 analogs closely match their reference products at the molecular level. In generic drug development, even very small structural differences can affect how safe or effective a drug is. Because of this, peptide mapping is not just supportive data—it is a core requirement for both scientific validation and regulatory approval.
Unlike traditional small-molecule drugs, GLP-1 analogs such as semaglutide and liraglutide are complex peptide-based therapies. Their performance depends on an exact amino acid sequence, correct chemical modifications, and controlled degradation patterns. If any of these elements change, the drug’s behavior in the body may also change. This makes accurate and detailed characterization extremely important.
GLP-1 Peptide Characterization Mapping provides deep structural insights that help confirm safety, effectiveness, and consistency. It also reduces the risk of unwanted immune responses by ensuring that the structure of the generic product remains highly similar to the original drug. This makes it a foundational tool in modern pharmaceutical analysis.
Explore our specialized services for complex biologics: Peptide Characterization Techniques and Applications
Share via:
Why is GLP-1 Peptide Characterization Mapping Critical in Generic Development?
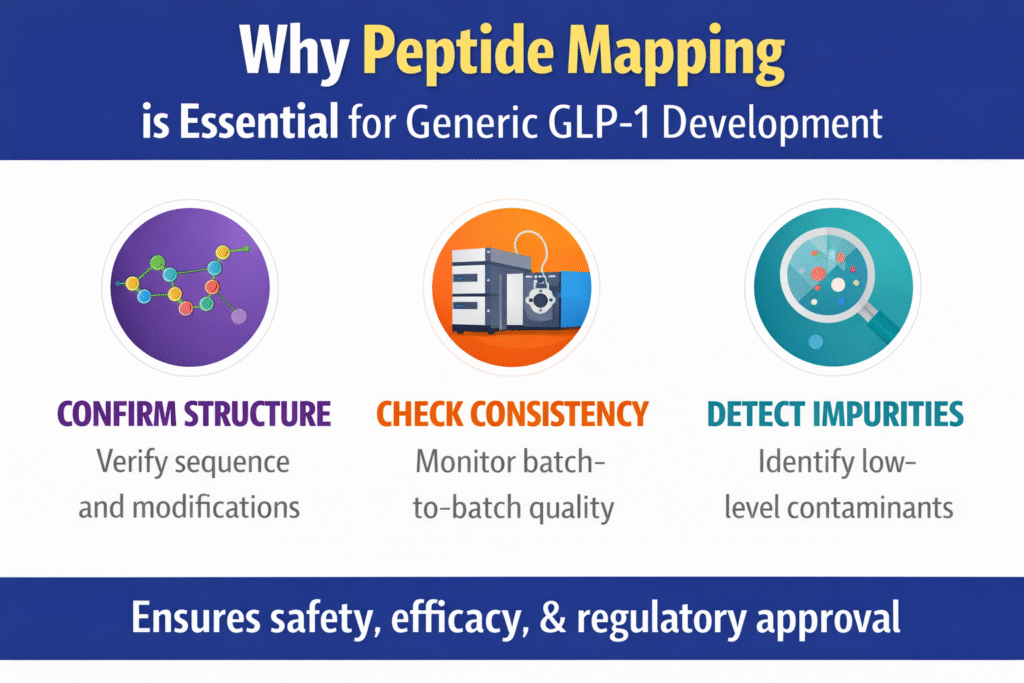
Peptide mapping is critical because it confirms the peptide structure at the amino acid level with high precision. This detailed analysis allows scientists to verify that the generic product matches the reference drug as closely as possible. It also helps detect even very small impurities that could impact how the drug performs clinically.
In GLP-1 analogs, small chemical changes like oxidation, deamidation, or incorrect fatty acid attachment can significantly affect how the drug behaves in the body. These changes may influence absorption, distribution, and metabolism. Identifying such variations early helps ensure consistent therapeutic results and reduces development risks.
LC-MS-based GLP-1 Peptide Characterization Mapping allows detailed comparisons between originator and generic products. It ensures that all critical quality attributes (CQAs) are properly evaluated and controlled. This level of analysis helps prevent unnoticed differences that could affect safety or efficacy.
Key Roles of Peptide Mapping:
- Sequence confirmation of the entire peptide backbone
- Identification of PTMs (e.g., acylation in semaglutide)
- Detection of impurities and truncations
- Monitoring batch-to-batch consistency
- Supporting comparability between originator and generic products
In addition, peptide mapping supports continuous quality monitoring throughout the product lifecycle. It ensures that manufacturing consistency is maintained even after regulatory approval.
Learn more about the differences between mapping and sequencing: Peptide Mapping vs. Peptide Sequencing: Key Differences
The Impact of Manufacturing Variability on GLP-1 Peptide Characterization Mapping
Manufacturing variability can introduce structural differences in peptide drugs, making GLP-1 Peptide Characterization Mapping essential for detecting and controlling these changes. Even small variations in the production process can lead to measurable differences in the final product. This is why detailed analytical testing is required at every stage.
GLP-1 analogs are highly sensitive to factors such as peptide synthesis conditions, reagent quality, and purification methods. Any change in these parameters can introduce unintended modifications. These changes are often not visible without advanced tools like peptide mapping.
Research has shown that even minor process changes can create variants detectable only through LC-MS-based mapping. These include misincorporated amino acids, oxidized residues, and isomerization products. Detecting these early helps avoid performance issues later.
Discover our specific study on Semaglutide: Peptide Mapping Study of Semaglutide
Examples of Structural Variations:
- Misincorporated amino acids
- Oxidized residues
- Isomerization products
These variations highlight the need for strict process control. Peptide mapping provides a reliable method to ensure that manufacturing changes do not affect product quality.
Table: Manufacturing Impact vs Analytical Detection
| Manufacturing Variation | Potential Structural Impact | Detected by Peptide Mapping |
|---|---|---|
| Oxidative stress | Methionine oxidation | ✔ Yes |
| pH variation | Deamidation (Asn/Gln) | ✔ Yes |
| Synthesis errors | Sequence mismatch | ✔ Yes |
| Lipidation inconsistency | Altered PK profile | ✔ Yes |
Regulatory Expectations for GLP-1 Peptide Characterization Mapping
Regulatory agencies consider GLP-1 Peptide Characterization Mapping a mandatory part of the analytical strategy for generic drug approval. It is one of the most reliable methods for confirming the molecular structure of peptide-based drugs. Without it, regulatory submissions would lack critical supporting data.
Organizations like the FDA and EMA require detailed structural characterization using multiple analytical tools. Peptide mapping plays a central role by providing direct evidence of the primary structure and identifying structural variants. While other techniques support the analysis, peptide mapping cannot be replaced.
Ensure your submission meets all guidelines: FDA Requirements for Peptide Characterization
In GLP-1 generic development, peptide mapping supports comparability studies and ensures manufacturing consistency. It helps confirm that there are no clinically meaningful differences between the generic and reference products. This is essential for gaining approval and maintaining patient confidence.
It also supports impurity profiling and risk assessment. By identifying potential safety concerns early, it helps reduce delays during regulatory review.
Role of LC-MS in GLP-1 Peptide Characterization Mapping
LC-MS-based GLP-1 Peptide Characterization Mapping is widely used because of its high sensitivity and accuracy. It combines liquid chromatography separation with mass spectrometry detection to provide a detailed view of peptide structures. This makes it especially useful for complex biologics.
The process usually involves enzymatic digestion using proteases like trypsin or Glu-C. The resulting fragments are separated and analyzed using tandem mass spectrometry (MS/MS). This allows scientists to study both the full molecule and its smaller components.
See our workflow for GLP-1 analogs: GLP-1 Analog Peptide Sequencing Workflow
Key capabilities include accurate mass measurement, sequence identification, and impurity detection. These features allow multiple variants to be analyzed in a single run, improving efficiency without compromising accuracy. As technology improves, LC-MS continues to deliver more reliable and reproducible results.
Detecting Critical Quality Attributes (CQAs) in GLP-1 Analogs
GLP-1 Peptide Characterization Mapping plays a direct role in identifying and monitoring critical quality attributes (CQAs). These attributes must be carefully controlled to ensure consistent safety and effectiveness. Poor control can lead to reduced drug performance or safety concerns.
Common CQAs include amino acid sequence integrity, correct acylation sites, and levels of oxidation or deamidation. Regions prone to aggregation are also important, as they can affect stability and immune response. Monitoring these ensures the product remains stable throughout its lifecycle.
Access comprehensive testing for your API: Peptide Purity Testing Services
If these attributes are not properly managed, the drug may show reduced potency or altered pharmacokinetics. In some cases, it may even trigger immune reactions. Peptide mapping provides the detailed information needed to prevent such issues.
Immunogenicity Risk and Impurity Profiling
Peptide mapping is essential for identifying impurities that could cause immune responses. Even very small amounts of impurities can have significant effects, which is why sensitive analytical methods are required.
Generic GLP-1 products must show low levels of impurities and no new immunogenic structures. GLP-1 Peptide Characterization Mapping helps achieve this by providing detailed impurity profiles and identifying potential risks early.
Partner with an expert CRO for your sequencing needs: GLP-1 Peptide Sequencing CRO Services
Types of Impurities Detected:
- Truncated peptides
- Misfolded variants
- Chemical degradation products
Understanding these impurities helps improve manufacturing processes and supports regulatory submissions.
Comparative Analytical Strategy for GLP-1 Generics
Peptide mapping is a key part of the overall analytical strategy used in generic drug development. It provides direct structural evidence that complements other analytical methods. Together, these approaches ensure a complete understanding of the product.
A typical strategy includes peptide mapping, intact mass analysis, bioassays, and stability testing. Each method offers unique insights, but peptide mapping stands out for its detailed, residue-level information.
Get support for your regulatory filings: Peptide Characterization for IND and NDA
This level of detail allows detection of even very small differences between products. It supports high-quality comparisons that are necessary for regulatory approval and product success.
Challenges in GLP-1 Peptide Characterization Mapping (and How to Overcome Them)
Despite its advantages, GLP-1 Peptide Characterization Mapping comes with challenges. These include handling complex modifications, detecting very low-level impurities, and ensuring consistent enzymatic digestion.
Complex changes like lipidation require specialized analytical approaches. Low-level impurities demand highly sensitive instruments. Variability in digestion can also affect results if not properly controlled.
Understand the hurdles in modern peptide analysis: GLP-1 Peptide Sequencing Challenges
Solutions include using multiple enzymes, high-resolution mass spectrometry, and strong method validation practices. These approaches improve accuracy and reproducibility. With modern tools, many of these challenges are now easier to manage.
Future Trends in GLP-1 Peptide Characterization Mapping
The future of GLP-1 Peptide Characterization Mapping is being shaped by automation and artificial intelligence. These technologies are improving both speed and accuracy in data analysis.
New tools include automated peptide identification and AI-based impurity prediction. These reduce manual work and improve consistency. High-throughput LC-MS systems also allow faster data generation.
These advancements are expected to shorten development timelines and improve confidence in results. As technology continues to grow, peptide mapping will become even more powerful and widely used.
Conclusion
GLP-1 Peptide Characterization Mapping is a critical component in the development of generic GLP-1 analogs. It ensures structural accuracy, supports safety evaluation, and meets strict regulatory requirements. Without it, proving equivalence would be extremely difficult.
As GLP-1 therapies become more advanced, the need for precise analytical methods continues to increase. Peptide mapping provides the level of detail required to detect even the smallest structural differences. This makes it essential for both development and quality control.
For companies working on generic GLP-1 products, investing in strong peptide mapping capabilities is not optional—it is necessary for long-term success, regulatory approval, and patient safety.
👉 For expert support in GLP-1 characterization and analytical services, connect us.
Frequently Asked Questions (FAQs)
Peptide mapping works by breaking the protein into smaller fragments and analyzing each one carefully. This allows scientists to find even very small or hidden impurities. These may include truncated sequences or chemical changes. Such detection is critical for maintaining product quality.
GLP-1 analogs are complex because they contain specific sequences and chemical modifications. They are also sensitive to environmental conditions like heat and pH. Even small changes can affect their behavior in the body. This makes advanced techniques necessary for proper analysis.
LC-MS helps separate peptide fragments and measure their mass very precisely. It allows scientists to identify sequences and detect modifications in a single workflow. This combination improves both speed and accuracy. It is one of the most trusted tools in peptide analysis.
Peptide mapping helps detect structural changes or impurities that might trigger an immune response. By identifying these risks early, it supports safer drug development. It also helps ensure the final product does not introduce new safety concerns. This is important for patient protection.
Peptide mapping provides detailed structural information, but it is not enough on its own. Other tests like biological assays and stability studies are also needed. Together, these methods give a complete understanding of the drug. This combined approach ensures better quality and compliance.
Common modifications include oxidation, deamidation, and lipid attachment. These changes can affect how the drug works and how stable it is. Detecting them early helps maintain product consistency. It also ensures better control over manufacturing quality.
Changes in the manufacturing process can introduce small structural differences in the peptide. These may not be visible without detailed analysis. Peptide mapping helps detect such variations quickly. This ensures that product quality remains consistent across batches.
Reference:
- Gillespie, M. A., Ranganathan, S., & Gallagher, E. S. (2024). Peptide mapping for sequence confirmation of therapeutic proteins and recombinant vaccine antigens by high-resolution mass spectrometry: Software limitations, pitfalls, and lessons learned. Journal of the American Society for Mass Spectrometry. https://pmc.ncbi.nlm.nih.gov/articles/PMC12563827/
- Degueldre, M., Wielant, A., Girot, E., Burkitt, W., O’Hara, J., Debauve, G., Gervais, A., & Jone, C. (2019). Native peptide mapping – A simple method to routinely monitor higher order structure changes and relation to functional activity. mAbs, 11(8), 1391–1401. https://pmc.ncbi.nlm.nih.gov/articles/PMC6816347/
- Ivleva, V. B., Gowetski, D. B., & Lei, Q. P. (2021). Streamlining peptide mapping LC-MS approach for studying fusion peptide-conjugated vaccine immunogens. Journal of the American Society for Mass Spectrometry, 32(12), 2777–2790. https://pmc.ncbi.nlm.nih.gov/articles/PMC6438762/



