
Introduction:
GLP-1 peptide characterization regulatory requirements are a critical component of modern drug development, especially for incretin-based therapies used in diabetes and obesity management. Regulatory authorities require comprehensive peptide sequencing of GLP-1 peptide data to ensure identity, purity, safety, and efficacy of GLP-1 analogs.
As peptide therapeutics grow in complexity, regulatory expectations have evolved to demand high-resolution, reproducible, and validated analytical approaches. This blog explores those expectations in detail and explains how robust sequencing strategies—such as analytical characterization of GLP-1 peptide drugs—align with global regulatory standards.
Share via:
Summary:
- GLP-1 peptide characterization regulatory requirements demand precise sequence confirmation, impurity profiling, and PTM (post-translational modification) mapping.
- Regulatory agencies expect high-resolution analytical techniques like LC-MS characterization of GLP-1 peptides.
- Sequence integrity, site-specific modifications, and GLP-1 peptide stability analytical methods are critical for approval.
- Compliance with ICH, FDA, and EMA guidelines is essential throughout development.
- Robust documentation, validation, and reproducibility are key to regulatory success.
- Advanced partners offering CRO for GLP-1 peptide characterization ensure regulatory success.
1: What Are the Regulatory Expectations for Peptide Sequencing?
Regulatory agencies expect complete and accurate peptide sequence confirmation along with comprehensive impurity and modification profiling to ensure drug identity, safety, and consistency.
In GLP-1 drug development, this includes advanced workflows such as the GLP-1 analog peptide sequencing workflow to ensure complete molecular characterization.
- Confirming primary structure to verify the exact amino acid sequence
- Identifying post-translational modifications (PTMs) such as oxidation, deamidation, or fatty acid conjugation
- Detecting process-related impurities introduced during synthesis or manufacturing
- Verifying batch-to-batch consistency to ensure reproducibility and product quality
Key Regulatory Expectations
- Full sequence coverage (ideally ≥ 99%) to demonstrate complete structural confirmation
- Identification of all variants and degradation products to ensure product safety
- Accurate mass confirmation using high-resolution analytical techniques
- Site-specific modification mapping to precisely locate and characterize PTMs
For deeper insights, refer to regulatory requirements for GLP-1 peptide characterization.
These expectations are critical for meeting global regulatory standards and ensuring successful approval of GLP-1 peptide therapeutics.
2: Why is Peptide Sequencing Critical for GLP-1 Drugs?
Peptide sequencing is critical because it ensures drug identity, stability, and safety, all of which are mandatory for regulatory approval and clinical reliability.
GLP-1 analogs require advanced strategies like peptide sequencing of GLP-1 drugs due to their structural complexity.
Because even small structural changes impact drug performance, high-precision methods such as de novo GLP-1 peptide sequencing accuracy become essential.
Why GLP-1 Analogs Require Advanced Sequencing
These peptides are complex due to:
- Fatty acid conjugation that improves half-life and bioavailability
- Amino acid substitutions that enhance receptor binding and stability
- Stability-enhancing modifications that reduce enzymatic degradation
Because even minor structural variations can impact efficacy or safety, high-resolution peptide sequencing becomes essential.
Regulatory Importance
- Prevents misidentification or contamination by confirming the exact molecular structure
- Ensures correct pharmacological activity by validating structure–function relationships
- Supports biosimilar comparability studies by demonstrating equivalence to reference products
- Confirms manufacturing consistency across different production batches
In short, peptide sequencing is not just an analytical requirement—it is a regulatory cornerstone that directly impacts approval, quality assurance, and patient safety in GLP-1 drug development.
3: Which Regulatory Guidelines Govern GLP-1 Peptide Characterization?
Global regulatory expectations for GLP-1 peptide characterization are governed by ICH, FDA, and EMA guidelines, all of which require comprehensive analytical data to ensure quality, safety, and efficacy.
These frameworks collectively define how peptide sequencing, impurity profiling, and method validation must be performed and documented for regulatory approval.
To align with these standards, companies rely on comprehensive analytical characterization of GLP-1 peptide drugs supported by validated methods and documentation.
Major Regulatory Frameworks
1. ICH Guidelines (International Council for Harmonisation)
ICH guidelines provide the global foundation for peptide characterization and are widely adopted across regulatory agencies.
- ICH Q6B: Defines specifications for biotechnological products, including peptide-based drugs
- ICH Q3A/Q3B: Focus on impurity identification, qualification, and reporting thresholds
- ICH Q2(R2): Covers analytical method validation, including accuracy, precision, specificity, and robustness
These guidelines ensure standardization and global consistency in analytical expectations.
2. FDA Expectations (U.S. Food and Drug Administration)
The FDA requires comprehensive and scientifically robust analytical characterization for GLP-1 peptides.
- Mandates detailed analytical characterization reports
- Strong emphasis on LC-MS/MS-based sequencing data for structural confirmation
- Supports comparability protocols for generics and biosimilars
- Requires complete impurity and degradation profiling
The FDA prioritizes data integrity, reproducibility, and traceability in all submissions.
3. EMA Requirements (European Medicines Agency)
The EMA places strong emphasis on understanding the relationship between structure and function.
- Focus on structure–function relationships to ensure therapeutic performance
- Requires orthogonal analytical methods (multiple complementary techniques)
- Strong emphasis on stability studies and degradation pathway analysis
- Detailed evaluation of process-related and product-related impurities
EMA expectations often go deeper into mechanistic understanding and long-term stability.
Key Takeaway
To meet GLP-1 peptide characterization regulatory requirements, developers must align with all three frameworks—ICH for global standards, FDA for detailed analytical rigor, and EMA for comprehensive structural and stability evaluation.
This integrated compliance approach ensures faster approvals, reduced regulatory risk, and global market readiness.
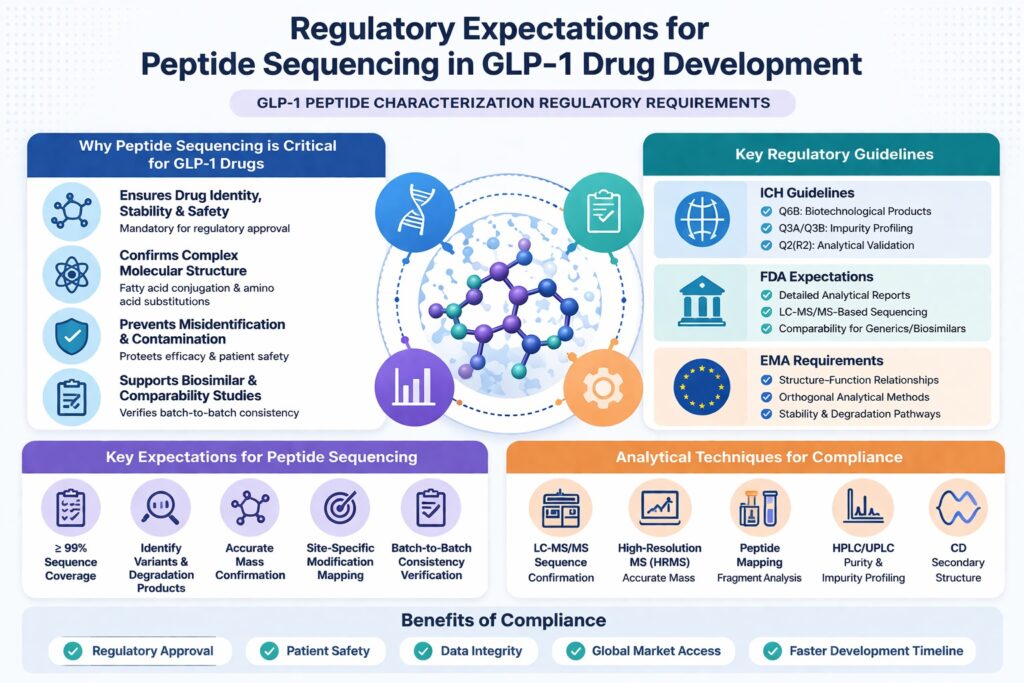
4: What Analytical Techniques Are Required for Compliance?
Regulators expect the use of advanced, high-resolution analytical techniques, with LC-MS/MS as the gold standard for peptide sequencing and characterization.
Regulators require high-resolution analytical tools, especially LC-MS characterization of GLP-1 peptides.
Additionally, advanced GLP-1 peptide sequencing analytical techniques are used to ensure complete structural and impurity profiling.
To meet GLP-1 peptide characterization regulatory requirements, a combination of complementary (orthogonal) techniques is required to fully evaluate structure, purity, and stability.
Core Analytical Techniques
| Technique | Purpose |
|---|---|
| LC-MS/MS | Sequence confirmation and post-translational modification (PTM) mapping |
| High-Resolution Mass Spectrometry (HRMS) | Accurate mass determination and molecular weight confirmation |
| Peptide Mapping | Fragment-level sequence validation and structural verification |
| HPLC/UPLC | Purity assessment and impurity profiling |
| Circular Dichroism (CD) | Secondary structure analysis |
Why LC-MS/MS Is Essential
LC-MS/MS is the most critical analytical tool for regulatory compliance because it provides detailed structural insights at the molecular level:
- Generates fragment ion spectra (b/y ions) for precise sequence confirmation
- Enables de novo sequencing, even without reference standards
- Detects low-level impurities and modifications with high sensitivity
- Supports site-specific PTM mapping, essential for GLP-1 analogs
Key Takeaway
No single technique is sufficient on its own. Regulatory agencies require a multi-technique (orthogonal) approach, where LC-MS/MS serves as the backbone, supported by HRMS, chromatography, and structural analysis methods to ensure comprehensive, accurate, and regulatory-compliant peptide characterization.
5: What Are the Key Components of GLP-1 Peptide Characterization Regulatory Requirements?
Regulatory expectations for GLP-1 peptides focus on identity, purity, structure, and stability, ensuring the drug is safe, effective, and consistently manufactured.
To meet GLP-1 peptide characterization regulatory requirements, developers must address the following critical components:
- Primary structure confirmation
- PTM mapping
- GLP-1 peptide impurity characterization
- Stability studies using GLP-1 peptide stability analytical methods
- Method validation
These collectively ensure full compliance with global regulatory expectations.
1. Primary Structure Confirmation
This ensures the correct amino acid sequence of the peptide.
- Full amino acid sequence validation
- High sequence coverage (typically ≥99%)
- Confirmation using techniques like LC-MS/MS
2. Post-Translational Modifications (PTMs)
Regulators require detailed identification and localization of all modifications.
- Common PTMs: oxidation, deamidation, glycation
- Fatty acid conjugation site confirmation (critical for GLP-1 analogs)
- Site-specific mapping to ensure structural integrity
3. Impurity Profiling
A complete impurity profile is essential for safety assessment.
- Process-related impurities from synthesis or manufacturing
- Degradation products formed during storage or handling
- Isomeric variants that may impact efficacy
4. Stability Studies
Stability data demonstrates how the peptide behaves under different conditions.
- Forced degradation studies to identify degradation pathways
- Long-term and accelerated stability studies
- Evaluation of changes in purity, potency, and structure over time
5. Method Validation
All analytical methods must be validated according to regulatory standards.
- Accuracy, precision, and specificity
- Limit of Detection (LOD) and Limit of Quantification (LOQ)
- Robustness and reproducibility of analytical methods
Key Takeaway
Together, these components form the foundation of GLP-1 peptide characterization regulatory requirements, ensuring comprehensive analytical understanding and regulatory compliance. A well-structured approach covering all five areas significantly improves the chances of successful regulatory approval and market readiness.

6: How Do Regulatory Agencies Evaluate Sequence Data?
Regulatory agencies evaluate sequence data based on accuracy, reproducibility, and completeness to ensure the peptide is correctly characterized and consistently produced.
For GLP-1 drugs, this evaluation is highly data-driven and requires both quantitative metrics and comprehensive documentation.
Evaluation Criteria:
- Regulators assess the quality of peptide sequencing data using the following parameters:
- Sequence Coverage (%)
Demonstrates how much of the peptide sequence has been confidently identified (typically ≥99% expected) - Mass Accuracy (ppm level)
Ensures precise molecular weight confirmation using high-resolution mass spectrometry - Fragment Ion Matching
Validates sequence through consistent b/y ion fragmentation patterns - Reproducibility Across Batches
Confirms that analytical results are consistent for different manufacturing batches - Data Integrity and Traceability
Ensures all data is reliable, attributable, and compliant with regulatory standards (e.g., ALCOA+ principles)
Documentation Requirements:
In addition to analytical performance, regulators require complete and transparent documentation:
- Raw and processed data for full traceability
- Method validation reports demonstrating reliability and robustness
- Chromatograms and spectra for visual and technical verification
- Detailed data interpretation explaining sequence confirmation and impurity identification
Accurate results depend on robust workflows like GLP-1 peptide sequencing CRO services that ensure reproducibility and compliance.
7: What Challenges Exist in Meeting Regulatory Expectations?
The biggest challenge is accurately characterizing structurally complex peptides like GLP-1 analogs, where even minor variations can impact safety, efficacy, and regulatory approval.
For a deeper understanding, refer to GLP-1 peptide sequencing challenges.
Common Challenges:
- Low-abundance impurity detection
Trace-level impurities are difficult to detect but critical for safety evaluation - PTM heterogeneity
Variability in post-translational modifications complicates consistent characterization - Isomer differentiation
Structural isomers (same mass, different structure) are challenging to distinguish - Complex fragmentation patterns
Peptides generate intricate MS/MS spectra, making sequence confirmation difficult - Data interpretation complexity
Large datasets require deep expertise to accurately interpret and validate results
Effective Solutions
- Expert data interpretation
Skilled scientists ensure accurate conclusions and regulatory-ready reporting - Use of high-resolution MS platforms
Enhances sensitivity, accuracy, and detection of minor variants - Advanced bioinformatics tools
Automates data analysis and improves confidence in sequence assignment - Orthogonal analytical techniques
Combines multiple methods (e.g., LC-MS/MS, HPLC, peptide mapping) for comprehensive validation
8: How Does ResolveMass Ensure Regulatory Compliance?
ResolveMass provides end-to-end support through GLP-1 peptide sequencing CRO services and advanced analytical platforms.
Their expertise in CRO for GLP-1 peptide characterization ensures:
Key Capabilities:
- Advanced LC-MS/MS and HRMS platforms
- Expertise in GLP-1 peptide sequencing
- Comprehensive impurity and PTM analysis
- Fully compliant with ICH, FDA, and EMA guidelines
What Sets ResolveMass Apart:
- Deep experience in complex peptide analysis
- Strong regulatory understanding
- End-to-end support from development to submission
- High data integrity and reproducibility
9: Best Practices for Meeting GLP-1 Peptide Characterization Regulatory Requirements
Following best practices ensures smoother regulatory approval, reduced risk, and higher data reliability in GLP-1 drug development.
To successfully meet GLP-1 peptide characterization regulatory requirements, organizations should adopt a structured and compliance-focused approach.
Recommended Approach
- Use validated, high-resolution analytical methods
Apply techniques like LC-MS/MS and HRMS with full method validation to ensure accuracy and reproducibility - Ensure ≥99% sequence coverage
Achieve near-complete sequence confirmation to meet regulatory expectations for identity verification - Perform comprehensive PTM mapping
Identify and localize all post-translational modifications, including oxidation, deamidation, and conjugation sites - Conduct forced degradation studies
Understand degradation pathways and assess peptide stability under stress conditions - Maintain complete documentation and traceability
Ensure all raw data, processed results, and reports are well-documented and audit-ready
Following proven workflows like GLP-1 analog peptide sequencing workflow significantly improves compliance success.
Conclusion:
GLP-1 peptide characterization regulatory requirements are rigorous and continuously evolving, reflecting the complexity of modern peptide therapeutics. Regulatory agencies demand comprehensive sequencing data, impurity profiling, and stability analysis to ensure drug safety and efficacy.
By leveraging technologies like LC-MS/MS and partnering with experts in GLP-1 peptide sequencing CRO services, pharmaceutical companies can ensure successful regulatory submissions and faster market entry.
Meeting these expectations requires not only advanced analytical technologies like LC-MS/MS but also deep expertise in peptide chemistry and regulatory science. Partnering with experienced organizations like ResolveMass Laboratories Inc. ensures that your GLP-1 drug development program is aligned with global regulatory standards and positioned for success.
Frequently Asked Questions:
Reference
- Kaspar AA, Reichert JM. Future directions for peptide therapeutics development. Drug discovery today. 2013 Sep 1;18(17-18):807-17.https://www.sciencedirect.com/science/article/pii/S1359644613001578
- Madny MA, Murthy A. From Venom‐to‐Vial‐to‐Pill: The Translational Journey of GLP‐1 Peptides and the Evolving Landscape of Biopharmaceutics Modeling. Journal of peptide science. 2026;32(4):e70092.https://onlinelibrary.wiley.com/doi/abs/10.1002/psc.70092
- Otvos L. The latest trends in peptide drug discovery and future challenges. Expert Opinion on Drug Discovery. 2024 Aug 2;19(8):869-72.https://www.tandfonline.com/doi/abs/10.1080/17460441.2024.2365969
- Hach M, Engelund DK, Mysling S, Mogensen JE, Schelde O, Haselmann KF, Lamberth K, Vilhelmsen TK, Malmstrøm J, Højlys-Larsen KB, Rasmussen TS. Impact of manufacturing process and compounding on properties and quality of follow-On GLP-1 polypeptide drugs. Pharmaceutical Research. 2024 Oct;41(10):1991-2014.https://link.springer.com/article/10.1007/s11095-024-03771-6


