
Introduction:
When it comes to GLP-1 Peptide Mapping vs Intact Mass, choosing the right analytical approach depends on your objective—whether it’s detailed structural characterization or rapid molecular confirmation. GLP-1 peptides, widely used in therapeutic applications such as diabetes and obesity treatment, require highly accurate and reliable analytical methods to ensure quality, safety, and regulatory compliance.
At ResolveMass Laboratories Inc., we routinely apply both peptide mapping and intact mass analysis using advanced LC-MS platforms, including LC-MS characterization of GLP-1 peptides, to deliver precise and reproducible results for GLP-1 characterization.
Share via:
Summary:
- GLP-1 Peptide Mapping vs Intact Mass analysis serves different but complementary purposes in peptide characterization.
- Peptide Mapping provides detailed, site-specific information about sequence and modifications.
- Intact Mass Analysis offers fast confirmation of molecular weight and overall purity.
- For regulatory compliance and deep characterization, peptide mapping is essential.
- For rapid identity confirmation and batch consistency, intact mass is more efficient.
- The best approach is often a combination of both techniques for comprehensive GLP-1 analysis.
1: What is GLP-1 Peptide Mapping VS Intact Mass Analysis?
GLP-1 Peptide Mapping vs Intact Mass refers to two complementary LC-MS-based analytical approaches used to characterize glucagon-like peptide-1 (GLP-1) molecules in terms of structure, identity, and molecular integrity. In simple terms, peptide mapping provides deep sequence-level confirmation, while intact mass analysis confirms the overall molecular weight of the full peptide in its undigested form.
For advanced structural workflows, refer to analytical characterization of GLP-1 peptide drugs.
Together, these techniques form a foundational part of GLP-1 peptide characterization in biopharmaceutical development, ensuring both structural accuracy and molecular consistency.
Key points:
- Involves enzymatic digestion (e.g., trypsin or other proteases)
- Produces peptide fragments for LC-MS/MS analysis
- Provides sequence coverage and site-specific information
- Detects modifications such as oxidation, deamidation, or truncation
In essence: Peptide mapping answers the question “Is the GLP-1 peptide sequence correct and structurally intact at every position?”
Intact Mass Analysis (GLP-1 Peptides)
Intact mass analysis is a rapid mass spectrometry technique used to measure the molecular weight of the complete GLP-1 peptide without any prior digestion or fragmentation.
It is mainly used for quick identity confirmation and molecular weight verification.
Key points:
- No enzymatic digestion required
- Direct analysis of the full peptide molecule
- Fast and high-throughput method
- Confirms expected molecular mass and detects major structural changes
In essence: Intact mass analysis answers the question “Does the GLP-1 peptide have the correct overall molecular weight?”
Key Difference in Simple Terms
- Peptide Mapping: Detailed structural fingerprint (sequence-level confirmation)
- Intact Mass Analysis: Overall molecular weight check (quick identity confirmation)
Why Both are used together
In modern GLP-1 characterization workflows, GLP-1 Peptide Mapping vs Intact Mass is not an either-or choice. Instead, both are used together to ensure:
- Fast screening (intact mass)
- Deep structural validation (peptide mapping)
- Regulatory-grade analytical confidence
This combined approach is essential for accurate characterization, quality control, and regulatory compliance in GLP-1 peptide development.
2: What is Peptide Mapping for GLP-1 Peptides?
Peptide mapping for GLP-1 peptides is a high-resolution LC-MS/MS analytical technique used to confirm the primary amino acid sequence and detect structural modifications in GLP-1-based molecules. It is one of the most critical tools in biopharmaceutical characterization because it provides detailed, site-specific information about peptide integrity.
In simple terms, peptide mapping answers: “Is every part of the GLP-1 sequence correct, and are there any structural changes or modifications present?”
For deeper insights, explore:
For sequencing-focused workflows:
It works by enzymatically digesting GLP-1 into smaller peptide fragments, which are then separated and analyzed using LC-MS/MS.
How Peptide Mapping Works for GLP-1 Peptides
Peptide mapping works by first breaking the GLP-1 peptide into smaller fragments and then analyzing those fragments using advanced LC-MS/MS systems.
Step-by-step principle:
- Enzymatic digestion of GLP-1 into smaller peptides
- Liquid chromatography (LC) separates the peptide fragments
- Mass spectrometry (MS/MS) detects and identifies each fragment
- Bioinformatics analysis reconstructs the full sequence map
This workflow allows scientists to reconstruct the complete GLP-1 sequence with high confidence.
Key Purpose:
The main purpose of peptide mapping is site-specific structural confirmation of GLP-1 peptides.
It ensures:
- Every amino acid in the GLP-1 sequence is correctly present
- No deletions, substitutions, or truncations exist
- Structural modifications are accurately identified and located
This makes peptide mapping essential for regulatory compliance and biologic drug characterization.
Key Advantages:
- High sequence coverage (often >95%)
- Detection of post-translational modifications (PTMs)
- Identification of impurities and degradation products
- Regulatory-grade structural confirmation
Typical Workflow:
Peptide mapping is widely used because it provides deep structural insight that cannot be achieved through intact mass analysis alone.
Major advantages include:
- High sequence coverage (often >95%)
Ensures near-complete verification of GLP-1 peptide structure - Detection of post-translational modifications (PTMs)
Identifies oxidation, deamidation, glycation, and other modifications - Impurity and degradation profiling
Detects minor variants and breakdown products - Regulatory-grade confidence
Meets expectations for FDA/EMA structural characterization - Site-specific accuracy
Pinpoints exact location of modifications within the sequence
3: What is Intact Mass Analysis for GLP-1 Peptides?
Intact mass analysis for GLP-1 peptides is a rapid LC-MS (liquid chromatography–mass spectrometry) technique used to determine the overall molecular weight of the complete, undigested GLP-1 peptide. It is widely used for quick confirmation of peptide identity and structural integrity during development and quality control.
In simple terms, intact mass analysis answers: “Does the GLP-1 peptide have the correct total molecular weight as expected?”
It provides a quick verification of whether the peptide has the expected molecular formula.
For integrated workflows, see:
Key Purpose:
The main purpose of intact mass analysis is to rapidly confirm the identity and molecular integrity of GLP-1 peptides.
It ensures:
- The peptide has the correct expected molecular weight
- No major truncations or deletions are present
- The batch matches the reference standard at a high level
It is especially valuable for fast screening and routine quality checks.
Key Advantages:
Intact mass analysis is preferred in many workflows due to its speed and simplicity.
Major advantages include:
- Rapid analysis (minutes per sample)
Enables high-throughput testing and fast decision-making - Minimal sample preparation
No enzymatic digestion required - Ideal for batch release testing
Quickly verifies identity during manufacturing QC - Useful for early-stage screening
Helps evaluate multiple GLP-1 variants efficiently - Simple workflow
Reduces complexity compared to peptide mapping
Limitations:
While fast and efficient, intact mass analysis has important limitations when compared to peptide mapping.
Key limitations include:
- No sequence-level information
Cannot confirm amino acid order or full structural details - Cannot localize modifications
Detects mass shifts but not exact modification sites - Limited impurity characterization
Cannot resolve complex or low-level variants effectively - Lower structural resolution
Provides only global molecular weight information
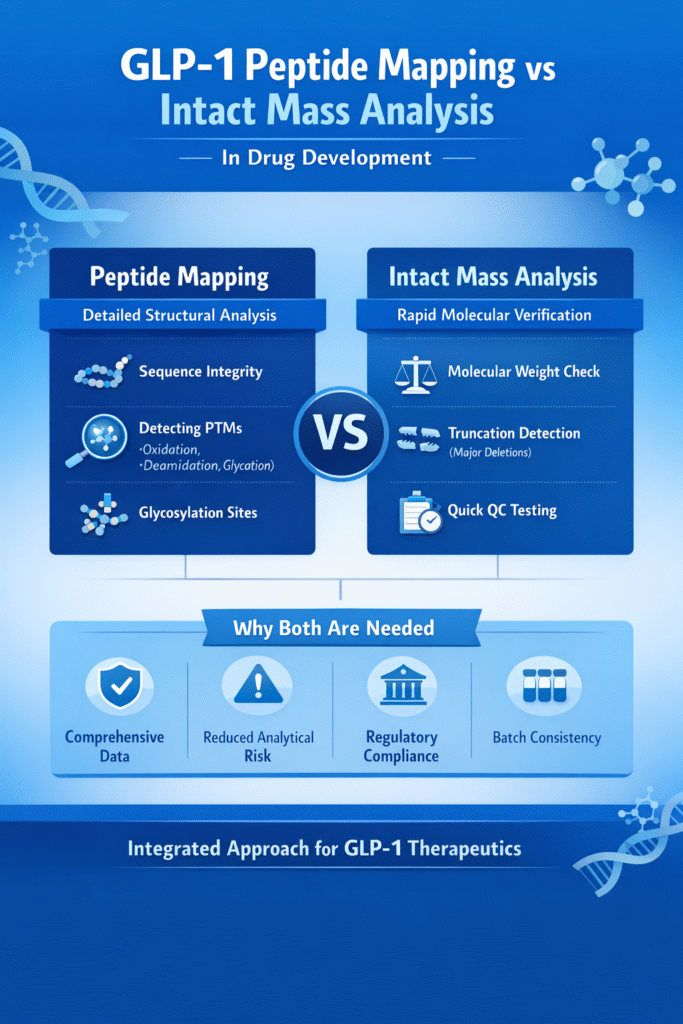
4: GLP-1 Peptide Mapping VS Intact Mass: Key Differences
The core difference in GLP-1 Peptide Mapping vs Intact Mass lies in the depth of structural information each technique provides. Peptide mapping delivers detailed sequence-level insights, while intact mass analysis provides a rapid overview of the peptide’s overall molecular weight and identity.
For complete analytical comparisons, refer to:
In short: peptide mapping = detailed structural confirmation, whereas intact mass = fast molecular weight verification.
| Feature | Peptide Mapping | Intact Mass Analysis |
|---|---|---|
| Level of Detail | High (amino acid resolution) | Low (whole molecule only) |
| Speed | Moderate (multi-step workflow) | Very fast (direct measurement) |
| Structural Information | Sequence + post-translational modifications | Molecular weight only |
| Regulatory Use | Full structural characterization | Identity confirmation |
| Sample Preparation | Complex (requires enzymatic digestion) | Simple (no digestion required) |
| Sensitivity to PTMs | Very high (site-specific detection) | Limited (detects only mass shifts) |
Key Difference in Structural Insight
The most important distinction in GLP-1 Peptide Mapping vs Intact Mass is the level of structural resolution:
- Peptide Mapping:
Provides complete amino acid-level resolution, allowing identification of sequence integrity and exact modification sites. - Intact Mass Analysis:
Provides global molecular weight confirmation, but cannot localize where structural changes occur.
5: Which is better for GLP-1 Peptides?
There is no single “better” method in GLP-1 Peptide Mapping vs Intact Mass analysis. Both techniques are essential, but they serve different analytical purposes depending on the development stage, regulatory requirement, and depth of information needed.
For outsourcing and CRO support:
In practice, the best approach is not choosing one over the other, but using them together as complementary tools for complete GLP-1 peptide characterization.
Intact Mass is Better When:
Intact mass analysis is better when speed, simplicity, and rapid confirmation are the priority.
It is most suitable in situations such as:
- Rapid identity confirmation of GLP-1 peptides
- Screening multiple batches in a short time
- Early-stage formulation or development studies
- Time-sensitive quality control checks during manufacturing
- Quick verification of expected molecular weight
In essence, intact mass is best for fast decision-making and routine monitoring.
Peptide Mapping is Better When:
Peptide mapping is better when detailed structural understanding is required.
It is essential in situations such as:
- Full structural characterization of GLP-1 peptides
- Regulatory submissions (FDA/EMA documentation)
- Identification and localization of post-translational modifications (PTMs)
- Detailed impurity and degradation profiling
- Confirmation of sequence integrity at amino acid level
In essence, peptide mapping is best for deep scientific and regulatory-grade analysis.
Best Practice in Industry:
In modern biopharmaceutical workflows, especially for GLP-1-based therapeutics, laboratories do not rely on a single method.
Instead, the industry standard is a complementary analytical strategy:
- Intact Mass Analysis → Fast screening and identity check
- Peptide Mapping → Deep structural validation and regulatory confirmation
This combined approach ensures:
- Strong regulatory compliance support
- High confidence in molecular identity
- Comprehensive structural characterization
- Reduced analytical risk
6: Why Regulators expect both Methods
Regulatory agencies such as the FDA and EMA expect the use of orthogonal analytical techniques because no single method can fully confirm both the identity and structural integrity of GLP-1 peptides. In GLP-1 Peptide Mapping vs Intact Mass workflows, this requirement ensures that peptide characterization is both scientifically robust and regulatory compliant.
In simple terms, regulators require multiple layers of evidence to confirm that a GLP-1 peptide is correctly structured, pure, and consistent across batches.
Learn more:
- Regulatory requirements for GLP-1 peptide characterization
- GLP-1 peptide characterization regulatory requirements
Why a Single Method is not Enough
Each technique has inherent limitations, which is why regulators discourage reliance on only one method:
- Intact Mass Analysis alone is insufficient
It confirms molecular weight but does not provide sequence-level confirmation or precise structural details. - Peptide Mapping alone is not sufficient for rapid verification
It provides deep structural insight but is more time-consuming and less suitable for quick identity checks.
Because of these limitations, a single technique cannot fully address all regulatory expectations for biologics such as GLP-1 peptides.
Regulatory Requirement: Orthogonal Analytical Approach
Regulators emphasize orthogonal methods, meaning different techniques that validate the same result from different scientific principles.
In GLP-1 characterization, this means:
- Intact Mass Analysis → Confirms overall molecular weight
- Peptide Mapping → Confirms sequence and structural integrity
Together, they provide complementary evidence that strengthens analytical certainty.
Why Combining Both Methods is Required
Using both methods in GLP-1 Peptide Mapping vs Intact Mass workflows ensures a complete analytical package that satisfies regulatory expectations.
Key benefits include:
- Higher data reliability
Cross-validation between two independent analytical techniques improves confidence in results. - Reduced analytical risk
Minimizes the chances of missing structural deviations or identity mismatches. - Stronger regulatory submissions
Provides comprehensive evidence required for IND, NDA, and BLA filings. - Better batch consistency assurance
Ensures reproducibility and comparability across manufacturing batches.
7: Application in GLP-1 Drug Development
In GLP-1 Peptide Mapping vs Intact Mass workflows, both techniques play a crucial role in the development of GLP-1-based therapeutics such as semaglutide-like molecules. These peptides are highly sensitive to even minor structural changes, which can significantly impact their biological activity, stability, and therapeutic efficacy.
For stability and impurity insights:
- GLP-1 peptide impurity sequencing analysis
- GLP-1 peptide stability analytical methods
- GLP-1 peptide impurity characterization
Because of this sensitivity, robust analytical characterization using both peptide mapping and intact mass analysis is essential throughout the drug development lifecycle.
For deeper technical insights:
- GLP-1 peptide sequencing challenges
- De novo GLP-1 peptide sequencing accuracy
- GLP-1 analog peptide sequencing workflow
Applications of Peptide Mapping:
Peptide mapping is primarily used for deep structural and sequence-level characterization.
Key applications include:
- Confirming amino acid sequence integrity
Ensures the GLP-1 peptide sequence matches the intended design without substitutions or deletions. - Detecting post-translational modifications (PTMs)
Identifies structural changes such as:- Oxidation
- Deamidation
- Glycation
- Mapping glycosylation or PEGylation sites
Determines exact modification locations in modified GLP-1 analogs used for improved stability or half-life extension.
Overall, peptide mapping provides high-resolution structural confirmation essential for regulatory submission and detailed characterization.
Applications of Intact Mass Analysis:
Intact mass analysis is used for rapid, high-level molecular verification.
Key applications include:
- Verifying correct molecular weight
Confirms that the GLP-1 peptide matches its theoretical mass. - Detecting major truncations or deletions
Identifies large structural changes or incomplete synthesis products. - Quick quality control during manufacturing
Enables fast batch-to-batch comparison in production environments.
Overall, intact mass analysis provides fast identity confirmation and early-stage quality assurance.
8: Challenges in Analysis of GLP-1 Peptides
GLP-1 peptides present significant analytical challenges because of their structural sensitivity, chemical modifications, and complex behavior in LC-MS-based workflows. In GLP-1 Peptide Mapping vs Intact Mass analysis, these challenges directly impact data accuracy, method selection, and overall confidence in peptide characterization.
In simple terms, GLP-1 peptides are difficult to analyze because they are small, chemically modified, and highly prone to degradation, requiring highly optimized analytical strategies.
For specialized sequencing services:
GLP-1 peptides present unique analytical challenges due to their:
- Short chain length
- High susceptibility to degradation
- Chemical modifications used for stability enhancement
Why GLP-1 Peprides are Analytically Challenging
GLP-1 peptides are designed for therapeutic stability, but this same complexity creates analytical difficulties.
Key structural reasons include:
- Short chain length → limits fragment diversity and spectral resolution
- High susceptibility to degradation → increases variability in results
- Chemical modifications for stability enhancement (e.g., lipidation, PEGylation) → complicate mass interpretation
These factors make both peptide mapping and intact mass analysis more demanding compared to standard peptides.
Common Challenges in Intact Mass Analysis
In GLP-1 Peptide Mapping vs Intact Mass, intact mass analysis is fast but faces important limitations:
- Overlapping isotopic patterns
Closely spaced mass signals can complicate accurate deconvolution of GLP-1 molecular weight. - Limited structural resolution
Cannot differentiate between similar variants or pinpoint modifications. - Sensitivity to minor modifications
Small chemical changes may not always be clearly resolved.
As a result, intact mass is powerful for speed but limited in detailed structural interpretation.
Common Challenges in Peptide Mapping
Peptide mapping provides deep structural insight but introduces its own technical challenges:
- Incomplete digestion
Enzymatic cleavage may not fully process GLP-1 peptides, leading to missing or partial fragments. - Low abundance impurities
Minor variants may be difficult to detect without optimized sensitivity settings. - Complex data interpretation
Large datasets require advanced bioinformatics tools for accurate sequence reconstruction.
Despite these challenges, peptide mapping remains essential for high-resolution structural confirmation.
LC-MS Specific Challenges
Both techniques share common analytical challenges when using LC-MS systems:
- Matrix interference
Co-eluting impurities can suppress or distort ion signals. - Ion suppression effects
Reduce sensitivity and affect quantitative accuracy. - Instrument resolution limits
High-resolution MS is required to distinguish closely related peptide species.
Why Advanced Systems are Required
Due to these complexities, GLP-1 peptide analysis requires advanced LC-MS instrumentation and optimized workflows.
Effective strategies include:
- High-resolution mass spectrometry (HRMS)
- Optimized digestion protocols
- Improved chromatographic separation
- Sophisticated data deconvolution tools
These enhancements ensure accurate interpretation in both peptide mapping and intact mass workflows.

9: Expert Insight from Analytical Practice
In real-world biopharmaceutical environments, analysts rarely choose between peptide mapping and intact mass analysis. Instead, they design integrated workflows:
- Intact mass → Fast confirmation of identity
- Peptide mapping → Deep structural validation
- Additional LC-MS assays → Stability and impurity profiling
This layered approach ensures both speed and accuracy in GLP-1 peptide characterization.
Conclusion:
In the debate of GLP-1 Peptide Mapping vs Intact Mass, the answer is not about choosing one over the other, but about understanding their complementary roles.
Intact mass analysis provides speed and simplicity, while peptide mapping delivers depth and structural precision. Together, they form a complete analytical strategy essential for modern GLP-1 peptide development and regulatory compliance.
For complete analytical support:
For biopharmaceutical companies, adopting both techniques ensures robust quality control, regulatory readiness, and confidence in molecular integrity.
At ResolveMass Laboratories Inc., advanced LC-MS workflows are designed to integrate both peptide mapping and intact mass analysis for comprehensive GLP-1 characterization, ensuring data reliability at every stage of development.
Frequently Asked Questions:
The main difference lies in the level of detail provided. Peptide mapping offers sequence-level structural information and identifies modifications, while intact mass analysis confirms the overall molecular weight of the GLP-1 peptide. In practice, peptide mapping is used for deep characterization, whereas intact mass is used for rapid identity verification.
Peptide mapping is essential because it confirms the exact amino acid sequence and detects structural modifications such as oxidation or deamidation. It provides site-specific insights that are critical for regulatory submissions and Peptide mapping is essential because it confirms the exact amino acid sequence and detects structural modifications such as oxidation or deamidation. It provides site-specific insights that are critical for regulatory submissions and ensures the structural integrity of GLP-1 peptides used in therapeutics.ensures the structural integrity of GLP-1 peptides used in therapeutics.
Intact mass analysis is best used when quick confirmation of molecular weight and identity is required. It is widely applied in early-stage development, batch screening, and routine quality control because it delivers fast results with minimal sample preparation.
No, intact mass analysis cannot replace peptide mapping. While it is useful for rapid verification, it does not provide sequence-level or site-specific structural information. Regulatory guidelines require detailed characterization, which only peptide mapping can deliver.
Regulatory agencies require both techniques because they provide orthogonal analytical evidence. Intact mass confirms the overall molecular weight, while peptide mapping confirms sequence and structural integrity. Using both ensures higher data reliability and stronger regulatory compliance.
GLP-1 peptides are challenging due to their short structure, susceptibility to degradation, and chemical modifications. Common issues include overlapping isotopic patterns in intact mass, incomplete digestion in peptide mapping, and matrix interference in LC-MS, all of which require advanced analytical methods to resolve.
Reference
- Zhang H, Xin B, Caporuscio C, Olah TV. Bioanalytical strategies for developing highly sensitive liquid chromatography/tandem mass spectrometry based methods for the peptide GLP‐1 agonists in support of discovery PK/PD studies. Rapid Communications in Mass Spectrometry. 2011 Nov 30;25(22):3427-35.https://analyticalsciencejournals.onlinelibrary.wiley.com/doi/abs/10.1002/rcm.5241
- Esposito S, de Leonibus ML, Ingenito R, Bianchi E, Orsatti L, Monteagudo E. A liquid chromatography high-resolution mass spectrometry in vitro assay to assess metabolism at the injection site of subcutaneously administered therapeutic peptides. Journal of Pharmaceutical and Biomedical Analysis. 2018 Sep 10;159:449-58.https://www.sciencedirect.com/science/article/pii/S0731708518312457
- Kim SH, Kim SS, Kim HJ, Park EJ, Na DH. Peptide mapping analysis of synthetic semaglutide and liraglutide for generic development of drugs originating from recombinant DNA technology. Journal of Pharmaceutical and Biomedical Analysis. 2025 Apr 15;256:116682.https://www.sciencedirect.com/science/article/pii/S0731708525000238
- Petersson P, Buckenmaier S, Euerby MR, Stoll DR. A strategy for assessing peak purity of pharmaceutical peptides in reversed-phase chromatography methods using two-dimensional liquid chromatography coupled to mass spectrometry. Part I: Selection of columns and mobile phases. Journal of Chromatography A. 2023 Mar 29;1693:463874.https://www.sciencedirect.com/science/article/pii/S0021967323001024


