
Introduction:
The GLP-1 Peptide Sequencing Analytical Technique plays a critical role in ensuring the structural integrity, purity, and efficacy of GLP-1 analog therapeutics. With the rising demand for peptide-based drugs in diabetes and obesity treatment, analytical precision has become more important than ever. Advanced approaches such as GLP-1 analog peptide sequencing workflow are now essential to achieve accurate characterization.
Among all available techniques, High-Resolution Mass Spectrometry (HRMS) stands out as the gold standard for peptide sequencing. It provides unmatched accuracy in determining molecular weight, identifying sequence variations, and detecting post-translational modifications (PTMs). These capabilities complement LC-MS characterization of GLP-1 peptides and broader analytical characterization of GLP-1 peptide drugs.
This article explores HRMS in detail and explains why it is the preferred analytical approach for peptide sequencing of GLP-1 peptide.
Share via:
Summary:
- HRMS (High-Resolution Mass Spectrometry) is the most powerful GLP-1 Peptide Sequencing Analytical Technique for accurate mass determination and structural confirmation.
- Enables high sensitivity, exact mass accuracy, and detection of modifications in GLP-1 analogs.
- Combines effectively with LC-MS/MS workflows for complete peptide characterization.
- Supports regulatory compliance through robust, reproducible, and traceable data aligned with regulatory requirements for GLP-1 peptide characterization.
- Essential for biopharma development, quality control, and biosimilar verification.
1: Principle of GLP-1 Peptide Sequencing
HRMS (High-Resolution Mass Spectrometry) is an advanced analytical technique that measures the exact mass of peptide ions with extremely high accuracy, enabling precise structural characterization.
In GLP-1 peptide sequencing, HRMS supports peptide sequencing of GLP-1 drugs by helping to:
- Determine exact molecular weight
- Identify amino acid sequence
- Detect modifications and impurities
Unlike conventional mass spectrometry, HRMS delivers mass accuracy in parts per million (ppm), which is essential for analyzing complex molecules such as GLP-1 analogs. This level of precision ensures confident identification of even minor structural variations, supporting both research and regulatory requirements.
2: Why is HRMS the Preferred GLP-1 Peptide Sequencing Analytical Technique?
HRMS is preferred because it provides precise, reliable, and highly detailed structural insights within a single analytical workflow, helping overcome common GLP-1 peptide sequencing challenges.
Key Advantages:
- Ultra-high mass accuracy → Reduces ambiguity in sequence identification
- High sensitivity → Detects low-abundance impurities
- Structural clarity → Identifies modifications like oxidation or deamidation
- Reproducibility → Ensures consistent results for regulatory submissions
Practical Impact:
For GLP-1 analogs and modified peptides, even minor structural changes can significantly impact efficacy and safety. HRMS enables complete molecular confirmation, helping minimize analytical risk and ensuring confidence in peptide characterization.
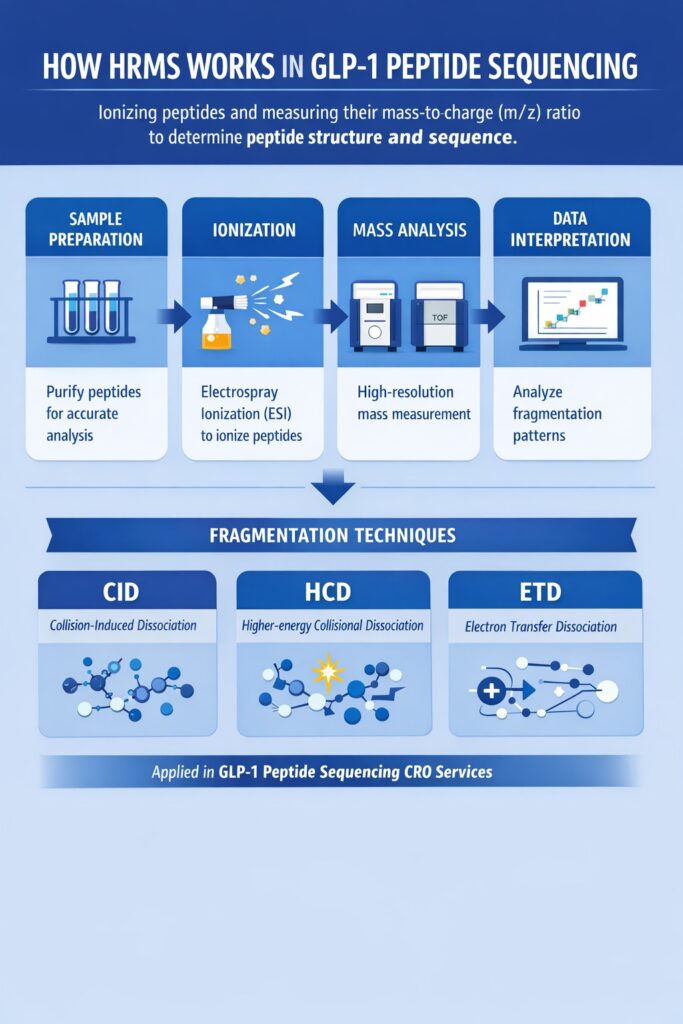
3: How HRMS Works in GLP-1 Peptide Sequencing
HRMS works by ionizing peptide molecules and accurately measuring their mass-to-charge (m/z) ratio, enabling precise determination of peptide structure and sequence.
Workflow Overview:
- Sample Preparation
- Peptides are extracted and purified to remove contaminants and ensure accurate analysis.
- Ionization
- Typically performed using Electrospray Ionization (ESI), which converts peptide molecules into charged ions without significant fragmentation.
- Mass Analysis
- Ions are analyzed using high-resolution instruments such as:
Orbitrap
Time-of-Flight (TOF) - These analyzers provide exact mass measurements with high resolving power.
- Ions are analyzed using high-resolution instruments such as:
- Data Interpretation
- Fragmentation patterns are analyzed to determine the amino acid sequence and identify structural features.
Fragmentation Techniques:
- CID (Collision-Induced Dissociation)
- HCD (Higher-energy Collisional Dissociation)
- ETD (Electron Transfer Dissociation)
These workflows are often integrated into GLP-1 peptide sequencing CRO services for efficient execution.
These fragmentation methods generate specific fragment ions, which are used to reconstruct the peptide sequence with high confidence.
4: Role of HRMS in Identifying Modifications
HRMS excels at detecting subtle chemical modifications in GLP-1 peptides and supports GLP-1 peptide impurity characterization.
Common Modifications Detected:
- Oxidation
- Deamidation
- Glycosylation
- PEGylation (in modified GLP-1 analogs)
Why This Matters:
- Ensures drug safety and stability
- Supports biosimilar comparability studies
- Meets regulatory expectations
HRMS is also critical in GLP-1 peptide stability analytical methods to monitor degradation pathways.
Even minor chemical modifications can significantly impact the pharmacokinetics and efficacy of GLP-1 peptides. HRMS enables precise identification of these changes, making it an essential tool for quality control and regulatory compliance.
5: Comparison: HRMS vs Other Analytical Techniques
| Technique | Sensitivity | Mass Accuracy | Structural Detail |
|---|---|---|---|
| HRMS | Very High | Very High | Excellent |
| LC-MS/MS | High | High | Excellent |
| HPLC | Moderate | NA | NA |
| NMR | Low | NA | NA |
Key Insight:
While techniques like HPLC and NMR are valuable for specific applications, HRMS combined with LC-MS/MS delivers the most comprehensive GLP-1 Peptide Sequencing Analytical Technique, offering superior accuracy, sensitivity, and structural characterization.
6: Applications of HRMS in GLP-1 Analysis
HRMS is widely used across pharmaceutical development and quality assurance for accurate and reliable peptide characterization.
Major Applications:
- Peptide sequencing and confirmation
- Impurity profiling
- Batch-to-batch consistency testing
- Stability studies
- Biosimilar comparison
Industry Use Cases:
- Drug development pipelines
- Regulatory submissions (FDA, Health Canada, EMA)
- Contract research organizations (CROs)
These applications are commonly performed by specialized providers offering CRO for GLP-1 peptide characterization.
HRMS plays a critical role in ensuring data accuracy, product quality, and regulatory compliance throughout the lifecycle of GLP-1 peptide therapeutics.

7: HRMS in Regulatory Compliance
HRMS supports regulatory compliance by delivering accurate and traceable data aligned with regulatory requirements for GLP-1 peptide characterization.
Regulatory Benefits:
- Meets ICH guidelines
- Supports method validation
- Provides audit-ready documentation
Regulatory authorities require precise and well-documented peptide analysis. HRMS ensures data integrity, reliability, and confidence, making it a critical tool for successful regulatory submissions and approvals.
8: Best Practices for HRMS-Based GLP-1 Peptide Sequencing
Following best practices ensures accurate, reproducible, and high-quality HRMS results for GLP-1 peptide analysis.
Recommended Approaches:
- Use high-purity sample preparation methods
- Optimize ionization conditions
- Validate fragmentation methods
- Implement robust data analysis software
Common Challenges:
- Matrix effects
- Signal suppression
- Complex data interpretation
These practices are essential in advanced analytical characterization of GLP-1 peptide drugs.
These challenges can be effectively mitigated through method optimization, proper instrument calibration, and expert analytical interpretation, ensuring reliable and regulatory-compliant results.
9: Why Choose Expert Analytical Partners for HRMS?
Partnering with experienced analytical laboratories ensures high-quality, accurate, and regulatory-compliant GLP-1 peptide sequencing results.
What to Look For:
- Proven expertise in peptide analysis
- Advanced HRMS instrumentation
- Regulatory compliance experience
- Strong data interpretation capabilities
An expert partner ensures:
- Faster turnaround
- Accurate results
- Reduced regulatory risk
Working with experts offering GLP-1 peptide sequencing CRO services ensures faster turnaround and reduced risk.
Choosing the right analytical partner helps ensure data confidence, streamlined development, and successful regulatory outcomes for GLP-1 peptide therapeutics.
Conclusion:
The GLP-1 Peptide Sequencing Analytical Technique is a cornerstone of modern peptide drug development, and HRMS stands at the forefront of this field. Its unmatched accuracy, sensitivity, and ability to detect structural nuances make it indispensable for GLP-1 analysis.
From sequencing and impurity profiling to regulatory compliance, HRMS—supported by workflows like GLP-1 analog peptide sequencing workflow—ensures both quality and confidence in peptide therapeutics.
Frequently Asked Questions:
The GLP-1 Peptide Sequencing Analytical Technique refers to advanced methods used to determine the amino acid sequence and structure of GLP-1 peptides. It ensures the peptide drug is correctly synthesized, pure, and structurally accurate for therapeutic use.
HRMS is crucial because it provides ultra-high mass accuracy and sensitivity, allowing precise identification of peptide sequences and modifications. It ensures reliable characterization, which is essential for drug safety, efficacy, and regulatory approval.
HRMS focuses on high-resolution mass accuracy, while LC-MS/MS combines separation and fragmentation for detailed sequencing. Together, they provide comprehensive peptide characterization, with HRMS enhancing confidence in mass identification and structural analysis.
HRMS can detect various modifications such as oxidation, deamidation, glycosylation, and PEGylation. These modifications can affect drug stability and performance, making their identification critical in pharmaceutical development.
HRMS is used for peptide sequencing, impurity profiling, stability studies, biosimilar comparison, and batch consistency testing. It plays a vital role throughout drug development and quality control processes.
Reference
- Maheshwari D, Badgujar D, Kumar G, Sharma N. Peptide Mapping Using Multienzyme Digestion Strategies Integrated with LC‐HRMS Workflow: A Case Study. Journal of Peptide Science. 2026 Mar;32(3):e70089.https://onlinelibrary.wiley.com/doi/abs/10.1002/psc.70089
- Arbouche N, Ameline A, Gheddar L, Raul JS, Kintz P. Glucagon like peptide-1 analogues analysis in whole blood samples by the use of LC-ESI-HRMS method. Toxicologie Analytique et Clinique. 2025 Mar 1;37(1):S61.https://www.sciencedirect.com/science/article/pii/S2352007824002920
- Arbouche N, Blanchot A, Raul JS, Kintz P. Semaglutide and health risk: development and validation of a LC-HRMS method for testing semaglutide in whole blood and application to real cases. Journal of Chromatography B. 2024 Jul 15;1242:124187.https://www.sciencedirect.com/science/article/pii/S157002322400196X
- Badgujar D, Bawake S, Chawathe A, Sharma N. Size‐exclusion LC‐UV/HRMS based method for the analysis of aggregates in synthetic GLP‐1 analog liraglutide and evaluation of excipient impact on aggregation. Biomedical Chromatography. 2024 Oct;38(10):e5983.https://analyticalsciencejournals.onlinelibrary.wiley.com/doi/abs/10.1002/bmc.5983
- Badgujar D, Bawake S, Sharma N. A comprehensive study on the identification and characterization of major degradation products of synthetic liraglutide using liquid chromatography‐high resolution mass spectrometry. Journal of Peptide Science. 2025 Jan;31(1):e3652.https://onlinelibrary.wiley.com/doi/abs/10.1002/psc.3652
- Zhang H, Xin B, Caporuscio C, Olah TV. Bioanalytical strategies for developing highly sensitive liquid chromatography/tandem mass spectrometry based methods for the peptide GLP‐1 agonists in support of discovery PK/PD studies. Rapid Communications in Mass Spectrometry. 2011 Nov 30;25(22):3427-35.https://analyticalsciencejournals.onlinelibrary.wiley.com/doi/abs/10.1002/rcm.5241


