
Introduction:
GLP-1 Sequence Variant Analysis is essential for detecting structural changes in GLP-1 peptides that can impact therapeutic performance and regulatory compliance.
Glucagon-like peptide-1 (GLP-1) analogs are widely used in the treatment of type 2 diabetes and obesity. Due to their complex structure and susceptibility to chemical and enzymatic modifications, even minor sequence variations can significantly affect biological activity, stability, and immunogenicity.
Peptide mapping, particularly when coupled with high-resolution LC-MS/MS, has become the industry-standard analytical technique for identifying sequence variants and modifications in GLP-1 peptides. Learn more about advanced workflows in GLP-1 analog peptide sequencing workflow and LC-MS characterization of GLP-1 peptides.
Share via:
Summary:
- GLP-1 Sequence Variant Analysis is critical for ensuring the safety, efficacy, and consistency of peptide therapeutics.
- Peptide mapping using LC-MS/MS is the gold standard for identifying sequence variants and PTMs—explore peptide mapping of GLP-1 peptides.
- Common variants include amino acid substitutions, truncations, deamidation, oxidation, and glycation.
- Accurate identification requires optimized digestion, high-resolution mass spectrometry, and robust data analysis workflows—see GLP-1 enzymatic digestion mapping.
- Regulatory agencies expect comprehensive characterization—refer to GLP-1 peptide characterization regulatory requirements.
- ResolveMass Laboratories Inc. provides advanced analytical solutions for high-confidence GLP-1 characterization.
1: What is GLP-1 Sequence Variant Analysis?
GLP-1 Sequence Variant Analysis involves identifying deviations in the amino acid sequence or chemical structure of GLP-1 peptides. These variations can directly impact the peptide’s biological activity, stability, and safety profile, making their detection essential in pharmaceutical development and quality control.
For deeper insights into sequencing fundamentals, visit peptide sequencing of GLP-1 peptide and peptide sequencing of GLP-1 drugs.
Sequence variants may arise from:
- Manufacturing inconsistencies
- Degradation during storage
- Chemical modifications
- Biological processing variations
Key Types of Variants
| Variant Type | Description |
|---|---|
| Amino Acid Substitution | Replacement of one amino acid residue with another |
| Truncation | Loss of amino acids from the N- or C-terminus |
| Insertions/Deletions | Addition or removal of residues within the sequence |
| Deamidation | Conversion of Asn or Gln into Asp or Glu |
| Oxidation | Modification of susceptible residues like Met or Trp |
| Glycation | Non-enzymatic attachment of sugar molecules to the peptide |
In GLP-1 Sequence Variant Analysis, identifying and characterizing these variants is critical to ensure product consistency, regulatory compliance, and therapeutic efficacy.
2: Why is Peptide Mapping Important for GLP-1?
Peptide mapping is essential for GLP-1 because it enables precise, site-specific identification of sequence variants and modifications with high sensitivity and accuracy. This ensures that even minor structural changes are detected before they impact drug safety or performance.
It is especially important for impurity detection—learn more in GLP-1 peptide impurity characterization and GLP-1 peptide impurity sequencing analysis.
For real-world applications, explore semaglutide peptide mapping.
Key Benefits
- Confirms primary structure
Verifies that the amino acid sequence matches the intended GLP-1 peptide design. - Detects low-level impurities
Identifies trace variants that may not be visible using less sensitive techniques. - Identifies PTMs at specific residues
Pinpoints exact locations of modifications such as oxidation or deamidation. - Supports regulatory submissions
Provides detailed analytical data required by global regulatory authorities. - Ensures batch-to-batch consistency
Confirms manufacturing reproducibility and product quality across production lots.
Peptide mapping is particularly critical for GLP-1 analogs like semaglutide and liraglutide, where even minor structural variations can significantly affect pharmacokinetics, stability, and therapeutic efficacy.
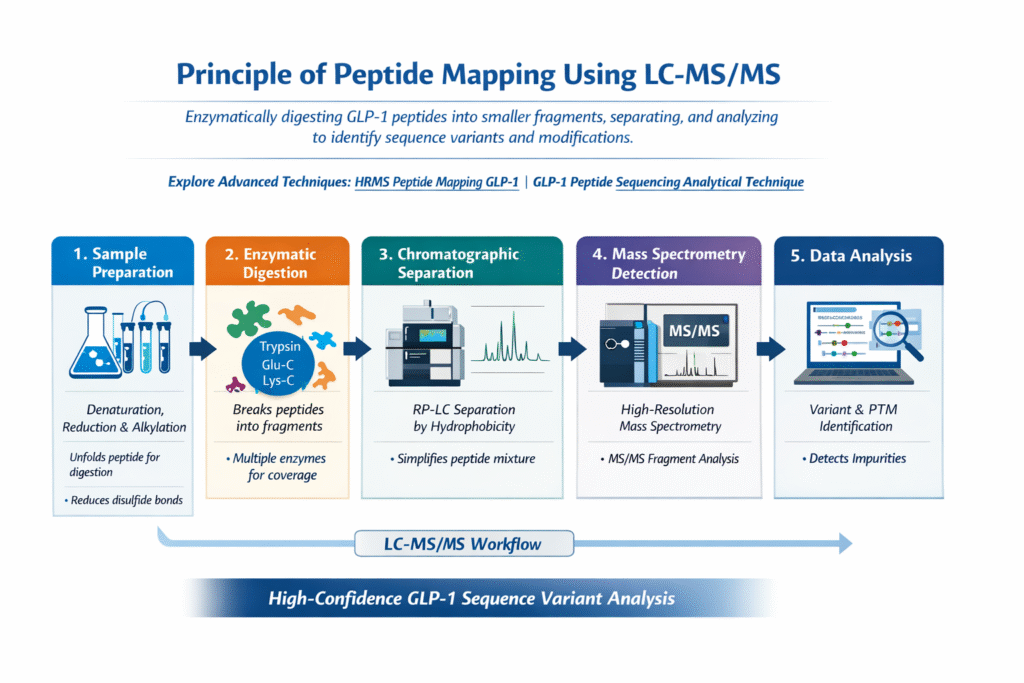
3: Principle of Peptide Mapping using LC-MS/MS
Peptide mapping using LC-MS/MS works by enzymatically digesting GLP-1 peptides into smaller fragments, which are then separated and analyzed to identify their sequence and any modifications. This approach provides high-resolution, site-specific information critical for accurate characterization.
Advanced analytical workflows are discussed in:
Workflow Overview:
1. Sample Preparation
- Involves denaturation, reduction, and alkylation
- Ensures the peptide is unfolded and accessible for enzymatic digestion
- Reduces disulfide bonds and prevents reformation
2. Enzymatic Digestion
- Breaks the peptide into smaller, predictable fragments
- Common enzymes used:
- Trypsin
- Glu-C
- Lys-C
- Multiple enzymes may be used to improve sequence coverage
3. Chromatographic Separation
- Performed using reverse-phase liquid chromatography (RP-LC)
- Separates peptide fragments based on hydrophobicity
- Reduces complexity before MS detection
4. Mass Spectrometry Detection
- Uses high-resolution mass spectrometry (HRMS)
- Measures mass-to-charge ratio (m/z) of peptide ions
- Generates fragmentation patterns (MS/MS spectra) for sequence confirmation
5. Data Analysis
- Specialized software compares observed data with theoretical peptide sequences
- Identifies:
- Sequence variants
- Post-translational modifications (PTMs)
- Unexpected impurities
This LC-MS/MS-based peptide mapping workflow is the foundation of GLP-1 Sequence Variant Analysis, enabling precise identification of structural changes with high confidence.
4: Identifying Sequence Variants in GLP-1 Peptides
Sequence variants in GLP-1 peptides are identified by detecting mass shifts and changes in fragmentation patterns during peptide mapping using LC-MS/MS. These analytical signals allow precise detection and confirmation of even subtle structural differences.
For deeper technical understanding, see:
Key Analytical Strategies
- Accurate Mass Measurement
- Detects very small differences in peptide mass, enabling identification of minor sequence changes or modifications.
- MS/MS Fragmentation
- Confirms the amino acid sequence by analyzing fragment ions (b/y ions), ensuring correct variant identification.
- Retention Time Comparison
- Identifies structural differences based on shifts in chromatographic behavior between variant and reference peptides.
- Database Matching
- Compares experimentally observed peptide data with theoretical or reference sequences to confirm known and unknown variants.
Example
| Variant Type | Mass Shift | Detection Method |
|---|---|---|
| Deamidation | +0.984 Da | MS/MS fragmentation + retention time shift |
| Oxidation | +15.995 Da | MS/MS fragmentation |
| Truncation | −X Da | Absence of expected peptide peaks |
By combining these strategies, GLP-1 Sequence Variant Analysis enables highly confident identification, localization, and characterization of peptide variants, ensuring product quality and regulatory compliance.
5: Identification of Post-Translational Modifications (PTMs)
PTMs in GLP-1 peptides are identified by detecting characteristic mass shifts and fragmentation signatures using LC-MS/MS. These modifications can influence peptide stability, potency, and immunogenicity, making their accurate identification essential in GLP-1 Sequence Variant Analysis.
Stability-related modifications are covered in GLP-1 peptide stability analytical methods.
Common PTMs in GLP-1
- Deamidation (Asn → Asp)
A common degradation pathway that can affect peptide charge and stability. - Oxidation (Met, Trp)
Occurs due to exposure to oxidative conditions, potentially impacting biological activity. - Acylation (in modified analogs)
Intentional modification in GLP-1 analogs (e.g., fatty acid conjugation) to enhance half-life. - Cyclization (N-terminal pyroglutamate formation)
Structural modification that may occur naturally or during processing.
PTM Detection Approach
- Monitor Mass Shifts
Identify specific changes in molecular weight corresponding to known PTMs. - Analyze Fragment Ions (b/y ions)
Use MS/MS fragmentation patterns to confirm the presence and type of modification. - Confirm Site-Specific Localization
Precisely determine the exact amino acid residue where the modification occurs.
Accurate identification of PTMs through peptide mapping is a critical component of GLP-1 Sequence Variant Analysis, ensuring comprehensive structural characterization and regulatory compliance.
6: Challenges in GLP-1 Sequence Variant Analysis
GLP-1 Sequence Variant Analysis presents several analytical challenges due to peptide complexity, low-level impurities, and limitations in detection and data interpretation. Addressing these challenges is critical to ensure accurate and reliable characterization.
To overcome these, many organizations rely on expert CRO support such as CRO for GLP-1 peptide characterization and GLP-1 peptide sequencing CRO services.
Key Challenges
- Low-abundance variants
Trace-level sequence variants can be difficult to detect against dominant peptide signals. - Co-eluting peptides
Similar peptides may overlap during chromatographic separation, complicating identification. - Incomplete digestion
Inefficient enzymatic cleavage can result in unexpected or missing peptide fragments. - Instrument sensitivity limitations
Some variants may fall below the detection limits of standard mass spectrometers. - Data interpretation complexity
Large datasets and overlapping signals require expert analysis and advanced tools.
Mitigation Strategies
- Use high-resolution mass spectrometers
Enhances sensitivity and mass accuracy for detecting minor variants. - Optimize enzymatic digestion protocols
Ensures consistent and complete peptide cleavage for better sequence coverage. - Apply orthogonal analytical methods
Combine techniques (e.g., LC-MS, HPLC) to improve confidence in results. - Implement advanced data processing software
Enables automated, accurate identification and characterization of variants.
By addressing these challenges with robust analytical strategies, GLP-1 Sequence Variant Analysis can achieve high accuracy, reproducibility, and regulatory compliance.
7: Regulatory Expectations for GLP-1 Characterization
Regulatory agencies require comprehensive identification, characterization, and quantification of sequence variants in GLP-1 peptides to ensure product safety, quality, and efficacy. Robust analytical data is a critical component of successful regulatory submissions and lifecycle management.
Learn more about compliance in:
- Regulatory requirements for GLP-1 peptide characterization
- Analytical characterization of GLP-1 peptide drugs
Key Guidelines
- ICH Q6B – Specifications for Biotechnological Products
Defines acceptance criteria and characterization requirements for biologics, including peptide therapeutics. - ICH Q2 (R1) – Analytical Method Validation
Establishes validation parameters such as accuracy, precision, specificity, and robustness. - USP <1047> – Biologics Characterization
Provides guidance on analytical approaches for structural characterization of peptides and proteins.
Regulatory Requirements
- Identification of all detectable variants
Comprehensive profiling of sequence variants and post-translational modifications. - Quantification of impurities
Accurate measurement of variant levels, especially those impacting safety and efficacy. - Validation of analytical methods
Demonstration that methods are reliable, reproducible, and fit for purpose. - Demonstration of method robustness and reproducibility
Ensures consistent performance across different conditions, instruments, and analysts.
Meeting these regulatory expectations through rigorous GLP-1 Sequence Variant Analysis ensures compliance, supports product approval, and builds confidence in therapeutic quality.
8: Best Practices for Accurate GLP-1 Sequence Variant Analysis
Accurate GLP-1 Sequence Variant Analysis requires well-optimized workflows, validated analytical methods, and expert data interpretation to ensure reliable and reproducible results. Implementing best practices minimizes analytical variability and improves confidence in variant identification.
Organizations can also outsource GLP-1 peptide sequencing services for improved efficiency and expertise.
Recommended Practices
- Use multiple enzymes for better sequence coverage
Combining enzymes like Trypsin, Glu-C, and Lys-C enhances peptide fragmentation and ensures comprehensive mapping. - Employ high-resolution LC-MS/MS systems
High mass accuracy and sensitivity are essential for detecting low-level variants and subtle modifications. - Validate method sensitivity and specificity
Ensure the method can accurately detect and differentiate variants at required concentration levels. - Perform replicate analyses for reproducibility
Multiple runs help confirm consistency and reliability of results across experiments. - Maintain strict sample handling protocols
Proper storage, preparation, and handling prevent artificial modifications and degradation.
By following these best practices, GLP-1 Sequence Variant Analysis achieves high precision, regulatory compliance, and robust characterization of peptide therapeutics.
9: Why choose ResolveMass Laboratories Inc.?
ResolveMass Laboratories Inc. offers industry-leading expertise in GLP-1 Sequence Variant Analysis using advanced peptide mapping technologies.
From sequencing to full characterization, explore:
Our Capabilities
- High-resolution LC-MS/MS platforms
- Expertise in complex peptide characterization
- Regulatory-compliant method development and validation
- Comprehensive data analysis and reporting
- Support for GLP-1 drug development and quality control
Our team combines deep scientific expertise with cutting-edge technology to deliver accurate, reliable, and regulatory-ready results.
Conclusion:
GLP-1 Sequence Variant Analysis is a critical component of peptide drug development, ensuring structural integrity, safety, and regulatory compliance.
Peptide mapping using LC-MS/MS provides unmatched precision in identifying sequence variants and modifications, enabling pharmaceutical companies to maintain high-quality standards. By adopting advanced analytical strategies and best practices, organizations can confidently characterize GLP-1 peptides and meet global regulatory expectations.
ResolveMass Laboratories Inc. stands as a trusted partner in delivering high-quality GLP-1 Sequence Variant Analysis, supporting innovation and excellence in peptide therapeutics.
Frequently Asked Questions:
GLP-1 Sequence Variant Analysis is the process of identifying any changes in the amino acid sequence or chemical structure of GLP-1 peptides. These variations can occur during manufacturing, storage, or due to chemical degradation. Detecting them is critical because even small changes can affect drug safety, efficacy, and stability. Advanced analytical techniques like LC-MS/MS are typically used for this purpose. This analysis ensures consistency and compliance with regulatory standards. It is a key step in peptide drug development and quality control.
Peptide mapping is important because it provides detailed, site-specific information about the peptide structure. It allows scientists to confirm the primary sequence and detect even low-level impurities or modifications. This technique is highly sensitive and accurate, making it ideal for complex peptide drugs like GLP-1 analogs. It also helps ensure batch-to-batch consistency during manufacturing. Additionally, peptide mapping supports regulatory submissions by providing comprehensive characterization data. Overall, it is essential for maintaining product quality and safety.
LC-MS/MS (Liquid Chromatography–Mass Spectrometry) is the most commonly used technique for GLP-1 Sequence Variant Analysis. It combines separation of peptide fragments with highly sensitive mass detection. This allows accurate identification of sequence variants and post-translational modifications. The technique provides high resolution, mass accuracy, and structural insights. It is widely accepted by regulatory agencies for peptide characterization. LC-MS/MS also enables detection of low-abundance impurities in complex samples.
Common sequence variants in GLP-1 peptides include amino acid substitutions, truncations, and insertions or deletions. Chemical modifications such as deamidation, oxidation, and glycation are also frequently observed. These variants can arise during synthesis, storage, or handling of the peptide. Even minor variations can alter the peptide’s biological activity or stability. Identifying these variants is essential to ensure therapeutic effectiveness. Proper characterization helps maintain product consistency and regulatory compliance.
Post-translational modifications (PTMs) are chemical changes that occur after the peptide is synthesized. In GLP-1 peptides, common PTMs include deamidation, oxidation, acylation, and cyclization. These modifications can impact the peptide’s stability, half-life, and biological activity. Some PTMs are intentional in drug design, while others occur due to degradation. Identifying and controlling PTMs is essential for ensuring product quality. Analytical techniques like LC-MS/MS are used to detect and localize these modifications.
PTMs are identified using LC-MS/MS by detecting specific mass shifts associated with each modification. The peptide is fragmented, and the resulting ions are analyzed to determine the exact location of the modification. This process allows precise site-specific identification of PTMs. Retention time shifts in chromatography also help confirm structural changes. Advanced software tools are used to match experimental data with theoretical models. This ensures accurate and reliable characterization of modifications.
LC-MS/MS offers high sensitivity and accuracy for detecting peptide variants and modifications. It provides detailed structural information through fragmentation patterns. The technique allows precise identification of amino acid sequences and PTMs. It is capable of detecting low-level impurities in complex samples. LC-MS/MS is widely accepted by regulatory agencies for analytical characterization. Its reliability makes it the preferred method for peptide mapping in pharmaceutical analysis.
Reference
- Dods RL, Donnelly D. The peptide agonist-binding site of the glucagon-like peptide-1 (GLP-1) receptor based on site-directed mutagenesis and knowledge-based modelling. Bioscience reports. 2016 Jan 15;36(1):e00285.https://portlandpress.com/bioscirep/article-abstract/36/1/e00285/56401
- Kim SH, Kim SS, Kim HJ, Park EJ, Na DH. Peptide mapping analysis of synthetic semaglutide and liraglutide for generic development of drugs originating from recombinant DNA technology. Journal of Pharmaceutical and Biomedical Analysis. 2025 Apr 15;256:116682.https://www.sciencedirect.com/science/article/pii/S0731708525000238
- Knihs VM, Filippin-Monteiro FB. GLP1R (glucagon-like-peptide-1 incretin receptor), diabetes and obesity phenotypes: An in silico approach revealed new pathogenic variants. Diabetes & Metabolic Syndrome: Clinical Research & Reviews. 2024 Feb 1;18(2):102956.https://www.sciencedirect.com/science/article/pii/S1871402124000171
- Longwell CK, Hanna S, Hartrampf N, Sperberg RA, Huang PS, Pentelute BL, Cochran JR. Identification of N-terminally diversified GLP-1R agonists using saturation mutagenesis and chemical design. ACS chemical biology. 2020 Dec 14;16(1):58-66.https://pubs.acs.org/doi/abs/10.1021/acschembio.0c00722
- Jazayeri A, Rappas M, Brown AJ, Kean J, Errey JC, Robertson NJ, Fiez-Vandal C, Andrews SP, Congreve M, Bortolato A, Mason JS. Crystal structure of the GLP-1 receptor bound to a peptide agonist. Nature. 2017 Jun 8;546(7657):254-8.https://www.nature.com/articles/nature22800


