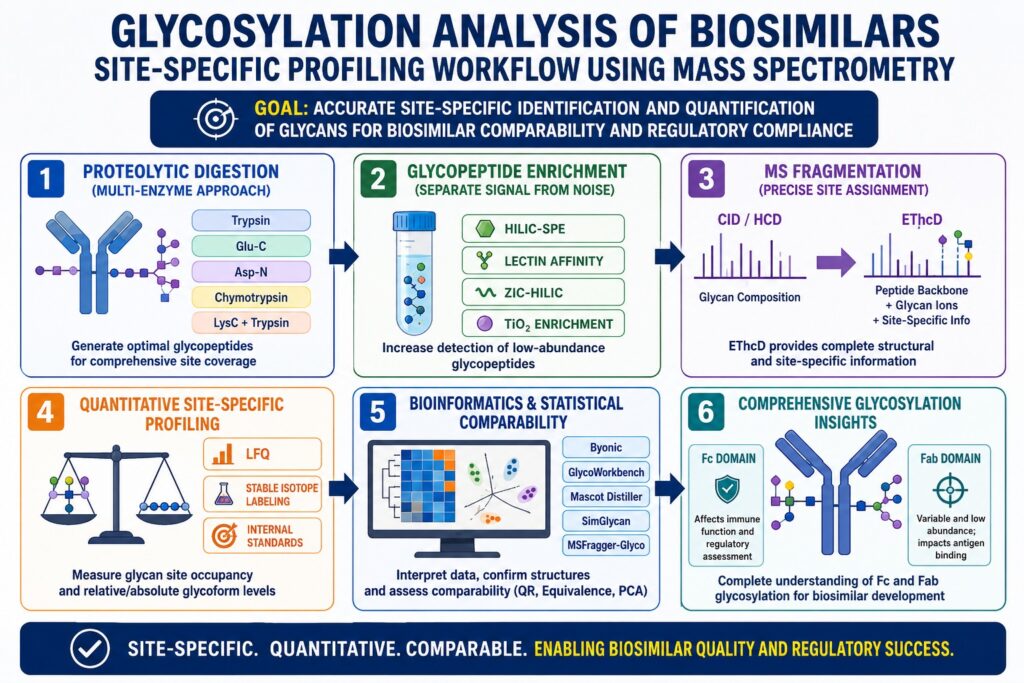
Introduction: Why Site-Specificity Is the Defining Challenge in Glycosylation Analysis of Biosimilars
Glycosylation Analysis of Biosimilars is a critical step in ensuring the safety and effectiveness of biologic drugs. The main difficulty is not just identifying glycans, but understanding exactly which glycan is attached to a specific site on the protein and how much of it is present. For example, a monoclonal antibody like trastuzumab has an N-glycosylation site that can carry many different glycan structures at the same time. This makes the analysis more detailed and demanding.
When developing a biosimilar, manufacturers must show that the glycan pattern at each specific site closely matches the original product. Regulatory authorities expect detailed, site-specific data rather than general averages. Mass spectrometry plays an important role here because it keeps the connection between the glycan and its exact attachment site. This makes Glycosylation Analysis of Biosimilars both complex and highly informative for decision-making.
Explore our full suite of characterization services: Expert Biosimilar Characterization Using Mass Spectrometry
Share via;
Proteolytic Digestion Strategies That Enable Site-Specific Glycopeptide Mapping in Glycosylation Analysis of Biosimilars
The choice of protease directly affects how well glycosylation sites can be analyzed. Trypsin is widely used because it produces peptides of suitable length for LC-MS/MS analysis. However, in regions with many lysine or arginine residues, digestion can create very short peptides. These may overlap with other signals and reduce clarity in results.
Protease Selection and Multi-Enzyme Approaches
| Protease | Cleavage Specificity | Advantage for Glycopeptide Analysis |
|---|---|---|
| Trypsin | C-terminal Lys, Arg | Broad coverage; predictable peptide lengths |
| Glu-C | C-terminal Glu (pH 4) or Glu/Asp (pH 8) | Ideal for regions dense in Glu; alternative Fc coverage |
| Asp-N | N-terminal Asp, Cys | Complements trypsin at hydrophilic glycosylation sites |
| Chymotrypsin | C-terminal Phe, Trp, Tyr, Leu | Useful for Fab domain glycosylation in IgG4 variants |
| LysC + Trypsin | Sequential digestion | Reduces missed cleavages at glycosylated Lys-adjacent sites |
Using multiple enzymes together is now a common practice in Glycosylation Analysis of Biosimilars. It improves accuracy and ensures that both N-linked and O-linked glycosylation sites are properly studied.
Optimize your mapping strategy: Advanced Peptide Mapping for Biosimilars
Glycopeptide Enrichment: Separating Signal from Noise
Glycopeptides are often present in very low amounts, making enrichment necessary. Without it, signals from non-glycosylated peptides can dominate the results and hide important information.
Common enrichment techniques include:
- HILIC-based SPE: High recovery and compatible with MS workflows
- Lectin affinity enrichment: Targets specific glycan types but may introduce bias
- ZIC-HILIC chromatography: Allows online enrichment with minimal sample loss
- TiO₂ enrichment: Useful for capturing sialylated glycopeptides
These methods improve detection and are essential for reliable Glycosylation Analysis of Biosimilars.
Ensure analytical precision for your molecules: Analytical Services for Generic Drug Development
MS Fragmentation Strategies for Site-Specific Glycan Assignment in Glycosylation Analysis of Biosimilars
Fragmentation techniques in mass spectrometry determine how well glycan structures and attachment sites are identified. Methods like CID and HCD mainly break down glycans, giving composition data but limited peptide information.
CID/HCD vs. ETD/EThcD: When to Use Each
CID and HCD produce diagnostic ions that confirm the presence of glycopeptides. However, they do not provide enough detail to locate the exact glycosylation site in complex peptides.
ETD solves this problem by breaking peptide bonds while keeping glycans intact. This allows precise identification of glycosylation sites. However, ETD can be less efficient for larger molecules.
EThcD combines both methods and provides a more complete dataset, including:
- Peptide backbone fragments
- Glycan-specific ions
- Structural and sequence information
Because of its balanced performance, EThcD is widely used in advanced Glycosylation Analysis of Biosimilars.
Leverage high-resolution data for your projects: Prove Biosimilarity Using State-of-the-Art LC-MS
Quantitative Site-Specific Profiling in Glycosylation Analysis of Biosimilars
Regulatory bodies require quantitative data that shows how much of each glycoform is present at each site. Simply detecting glycans is not enough for biosimilar approval.
Approaches to Glycopeptide Quantification
- Label-Free Quantification (LFQ): Simple and useful for early comparisons, but less precise
- Stable Isotope Labeling: Provides higher accuracy and allows direct comparison
- Internal Standards: Enables absolute quantification and supports regulatory compliance
Critical Metric: Glycan Site Occupancy
Site occupancy measures how many potential glycosylation sites are actually occupied. This cannot be determined from general glycan analysis alone. Even small differences can affect how a drug behaves in the body.
Mass spectrometry enables accurate measurement by comparing glycosylated and non-glycosylated peptides, making it essential in Glycosylation Analysis of Biosimilars.
Navigate complex regulatory requirements: Comprehensive Regulatory Support for Generic Drug Development
Fc Glycosylation vs. Fab Glycosylation in Glycosylation Analysis of Biosimilars
Fc and Fab regions present different analytical challenges due to their structure and function.
Fc Domain Analysis
The Fc region is less accessible and requires careful preparation, including:
- Reduction and alkylation
- Heat denaturation
- Long digestion times
Glycans in this region affect immune functions, making them highly important for regulatory evaluation.
Understand your molecule’s modifications: Detailed Analysis of Post-Translational Modifications (PTMs)
Fab Domain Glycosylation Analysis
Fab glycosylation is more variable and harder to detect due to:
- Lower abundance
- Higher structural diversity
- Impact on antigen binding
Accurate mapping of these regions is essential for complete Glycosylation Analysis of Biosimilars.
Regulatory and Bioinformatics Considerations in Glycosylation Analysis of Biosimilars
Regulatory agencies now expect detailed, site-specific glycan data in biosimilar submissions. This has increased the need for advanced data analysis tools.
Bioinformatics Tools for Glycopeptide Data Interpretation
The complexity of site-specific glycan data — hundreds of glycoform combinations across multiple sites — demands purpose-built bioinformatics:
| Software Tool | Primary Function | Key Feature for Biosimilar Work |
|---|---|---|
| Byonic (Protein Metrics) | Glycopeptide database search | Handles large glycan databases; score-based glycoform assignment |
| GlycoWorkbench | Glycan structure drawing and annotation | Visual verification of proposed glycan structures |
| Mascot Distiller + Glycan DB | MS/MS interpretation | Integration with existing proteomics pipelines |
| SimGlycan | Glycan fragmentation prediction | Validates oxonium ion pattern assignments |
| MSFragger-Glyco | Open-search glycopeptide ID | Discovers unexpected glycoforms without pre-defined database |
Statistical Comparability Framework
To compare biosimilars with reference products, statistical methods are used:
- Quality Range (QR): Defines acceptable variation
- Equivalence Testing: Confirms similarity within limits
- PCA: Provides a visual comparison of datasets
These methods ensure that Glycosylation Analysis of Biosimilars meets regulatory standards.
Intact Glycoprotein and Middle-Up Approaches in Glycosylation Analysis of Biosimilars
Intact and middle-up mass spectrometry approaches provide a broader view of glycosylation patterns. They help detect variations that may not appear in peptide-level analysis.
Intact mass analysis gives an overview of the whole protein, while middle-up approaches analyze large fragments that retain glycan structures. These methods improve detection sensitivity and provide additional insights.
However, they cannot always assign glycans to exact sites when multiple sites are present. This is why they are used alongside peptide-level analysis in Glycosylation Analysis of Biosimilars.
Get the big picture of your biologic: Precision Intact Mass Analysis for Biosimilars
Conclusion: The Future of Glycosylation Analysis of Biosimilars
Glycosylation Analysis of Biosimilars has evolved into a precise and data-driven process. It now focuses on detailed, site-specific, and quantitative characterization of glycan structures. This level of detail is necessary for regulatory approval and product consistency.
Modern workflows combine advanced fragmentation techniques, multiple enzyme digestion, enrichment strategies, and powerful software tools. Together, they provide a complete understanding of glycosylation patterns.
As biosimilars become more complex, the importance of accurate glycosylation analysis will continue to grow. At ResolveMass Laboratories Inc., advanced LC-MS/MS workflows are used to deliver reliable, site-specific glycan data that meet global regulatory expectations.
📩 Ready to discuss your biosimilar glycan characterization needs?
Contact ResolveMass Laboratories Inc.
Frequently Asked Questions (FAQs)
Released glycan profiling separates glycans from the protein and studies them independently, which removes critical site-specific information. This means it cannot show which glycan was attached to which position on the protein. In Glycosylation Analysis of Biosimilars, this detail is essential because the same glycan can behave differently depending on its location. Regulatory agencies such as the FDA and EMA expect site-specific data as part of biosimilar evaluation. Without this level of detail, the analysis is considered incomplete for regulatory purposes.
Advanced mass spectrometry methods can detect a wide range of glycoforms at a single Fc glycosylation site. In most cases, around 30 to 80 different glycoforms may be identified at the Asn-297 position in IgG1 antibodies. These include high-mannose, hybrid, and complex glycans with different levels of fucosylation and sialylation. In Glycosylation Analysis of Biosimilars, the main challenge is not just identifying these forms but measuring their relative amounts accurately. This helps confirm that the biosimilar matches the reference product within accepted variability.
Core fucosylation plays a major role in how therapeutic antibodies interact with the immune system. When fucose is absent, antibodies can bind more strongly to Fc receptors, leading to increased immune activity such as ADCC. Even small differences in fucosylation levels between a biosimilar and its reference product can affect biological performance. In Glycosylation Analysis of Biosimilars, measuring the level of fucosylated and afucosylated glycans is therefore very important. This ensures consistent therapeutic activity and regulatory compliance.
Standard mass spectrometry methods like CID and HCD cannot clearly differentiate between α2,3 and α2,6 sialic acid linkages because they produce similar fragment ions. To overcome this, additional techniques are required. These may include enzyme treatments that selectively remove specific linkages or chemical labeling methods that create detectable differences. Ion mobility mass spectrometry can also help by separating molecules based on their shape. In Glycosylation Analysis of Biosimilars, these advanced approaches improve structural accuracy.
Small differences in glycan structures at specific sites can significantly influence how a drug behaves in the body. For example, high-mannose glycans can lead to faster clearance, while sialylated glycans may extend circulation time. These variations can affect both drug efficacy and safety. In Glycosylation Analysis of Biosimilars, understanding site-specific microheterogeneity helps predict pharmacokinetic behavior. This is why detailed glycan profiling is essential during biosimilar development.
Sample preparation must be carefully controlled to avoid errors in glycan analysis. High temperatures can damage sensitive sialic acids, while long storage times may cause unwanted chemical changes in peptides. Changes in pH can also lead to loss of certain glycans or incorrect identification of modification sites. In Glycosylation Analysis of Biosimilars, such artifacts can affect data accuracy and lead to incorrect conclusions. Following validated protocols helps maintain consistency and reliability in results.
O-glycosylation is more difficult to analyze because it does not follow a clear sequence pattern like N-glycosylation. Specialized fragmentation methods such as ETD or EThcD are used to preserve glycan structures while breaking the peptide backbone. This allows accurate identification of glycosylation sites within the protein. Additional enzyme treatments can confirm these findings by showing expected mass changes. In Glycosylation Analysis of Biosimilars, combining these techniques ensures precise mapping of O-glycosylation sites.
Ion mobility mass spectrometry adds another layer of separation by analyzing the shape and size of molecules. This helps distinguish glycopeptides that have the same mass but different structures. It is especially useful for identifying isomers and resolving overlapping signals in complex samples. In Glycosylation Analysis of Biosimilars, IM-MS improves confidence in glycan identification. It also supports more detailed and accurate characterization of biosimilar products.
Regulatory agencies review glycan data as part of a larger analytical comparison between the biosimilar and the reference product. They expect validated methods that provide accurate and reproducible results. Statistical analysis is used to confirm that any differences fall within acceptable limits. Agencies also require confirmation using more than one analytical approach. In Glycosylation Analysis of Biosimilars, this ensures that conclusions are reliable and supported by strong scientific evidence.
Reference:
- Nupur, N., Joshi, S., Guillarme, D., & Rathore, A. S. (2022). Analytical similarity assessment of biosimilars: Global regulatory landscape, recent studies and major advancements in orthogonal platforms. Frontiers in Bioengineering and Biotechnology, 10, 832059. https://pmc.ncbi.nlm.nih.gov/articles/PMC8865741/
- D’Atri, V., Guillarme, D., & Beck, A. (2025). Biopharmaceutical analysis—current analytical challenges, limitations, and perspectives. Analytical and Bioanalytical Chemistry. https://pmc.ncbi.nlm.nih.gov/articles/PMC12783247/
- Berkowitz, S. A., Engen, J. R., Mazzeo, J. R., & Jones, G. B. (2012). Analytical tools for characterizing biopharmaceuticals and the implications for biosimilars. Nature Reviews Drug Discovery, 11(7), 527–540. https://pmc.ncbi.nlm.nih.gov/articles/PMC3714370/



