
Introduction:
HRMS Peptide Mapping GLP-1 is a powerful analytical approach used to characterize the structure, purity, and sequence integrity of GLP-1 peptides and their analogs. By combining enzymatic digestion with high-resolution mass spectrometry, laboratories can obtain highly accurate molecular insights critical for drug development, quality control, and regulatory submissions.
For deeper insights into sequence-level analysis, explore GLP-1 peptide sequencing and peptide sequencing of GLP-1 drugs.
GLP-1 peptides are widely used in the treatment of type 2 diabetes and obesity, making their structural verification essential. High-resolution mass spectrometry (HRMS) provides the sensitivity and accuracy required to detect even minor modifications, degradants, and sequence variants. Learn more about LC-MS characterization of GLP-1 peptides and analytical characterization of GLP-1 peptide drugs.
At ResolveMass Laboratories Inc., advanced HRMS workflows are designed to ensure reliable, reproducible, and regulatory-compliant peptide mapping for GLP-1 therapeutics.
Share via:
Summary:
- HRMS Peptide Mapping GLP-1 enables precise structural characterization of GLP-1 and its analogs.
- High-resolution mass spectrometry ensures accurate mass detection, sequence confirmation, and impurity profiling.
- Proper sample preparation, enzymatic digestion, and chromatographic separation are critical for reliable results.
- Optimized instrument parameters and data analysis workflows significantly improve sensitivity and reproducibility.
- Following best practices ensures regulatory compliance, data integrity, and robust peptide characterization.
1: What is HRMS Peptide Mapping for GLP-1?
HRMS Peptide Mapping GLP-1 is an advanced analytical technique used to identify, confirm, and characterize GLP-1 peptide sequences and their modifications using high-resolution mass spectrometry after enzymatic digestion.
For workflow-specific insights, refer to GLP-1 analog peptide sequencing workflow and GLP-1 enzymatic digestion mapping.
This approach combines precise enzymatic cleavage with high-resolution detection to generate a detailed molecular fingerprint of GLP-1 and its analogs, ensuring structural integrity and product quality.
How the Process Works:
- Enzymatic Digestion:
GLP-1 peptides are cleaved into smaller, predictable fragments using specific enzymes (e.g., trypsin). - Liquid Chromatography (LC) Separation:
The resulting peptide fragments are separated based on their physicochemical properties such as hydrophobicity. - High-Resolution Mass Spectrometry (HRMS) Detection:
The separated peptides are analyzed with high mass accuracy to determine their molecular weights and structural features.
Key Objectives:
- Confirm Amino Acid Sequence:
Ensures the primary structure of the GLP-1 peptide is correct. - Identify Post-Translational Modifications (PTMs): (see GLP-1 peptide sequencing analytical techniques)
Detects modifications such as oxidation, deamidation, or glycosylation. - Detect Impurities and Degradation Products:
Identifies unwanted variants or breakdown products that may impact safety or efficacy.
(learn more: GLP-1 peptide impurity characterization) - Ensure Batch-to-Batch Consistency:
Verifies reproducibility and quality across manufacturing lots.
This technique is essential for pharmaceutical development, quality control, and regulatory compliance of GLP-1-based therapeutics.
2: Why High-Resolution Mass Spectrometry is Essential for GLP-1 Mapping
High-Resolution Mass Spectrometry (HRMS) is essential for GLP-1 mapping because it provides the accuracy, sensitivity, and resolution required to detect even the smallest structural variations in GLP-1 peptides.
GLP-1 and its analogs often contain subtle modifications, sequence variants, and low-level impurities that cannot be reliably detected using conventional mass spectrometry. HRMS enables precise characterization, ensuring product quality, safety, and regulatory compliance.
For deeper technical challenges, refer to GLP-1 peptide sequencing challenges and de novo GLP-1 peptide sequencing accuracy.
Advantages of HRMS:
- High Mass Accuracy (ppm-level precision):
Enables exact identification of peptide fragments and molecular compositions. - Enhanced Sensitivity:
Detects trace-level impurities and degradation products that may impact efficacy. - Improved Resolution:
Distinguishes closely related peptide species, including isobaric compounds. - Comprehensive Structural Information:
Supports detailed sequence confirmation and post-translational modification (PTM) analysis.
Comparison: Conventional MS vs HRMS
| Parameter | Conventional MS | HRMS |
|---|---|---|
| Mass Accuracy | Low | High (ppm level) |
| Resolution | Moderate | Very High |
| Impurity Detection | Limited | Highly Sensitive |
| Data Reliability | Moderate | Excellent |
In GLP-1 peptide mapping workflows, HRMS is not just an enhancement—it is a necessity for achieving high-confidence analytical results, especially in regulated pharmaceutical environments.
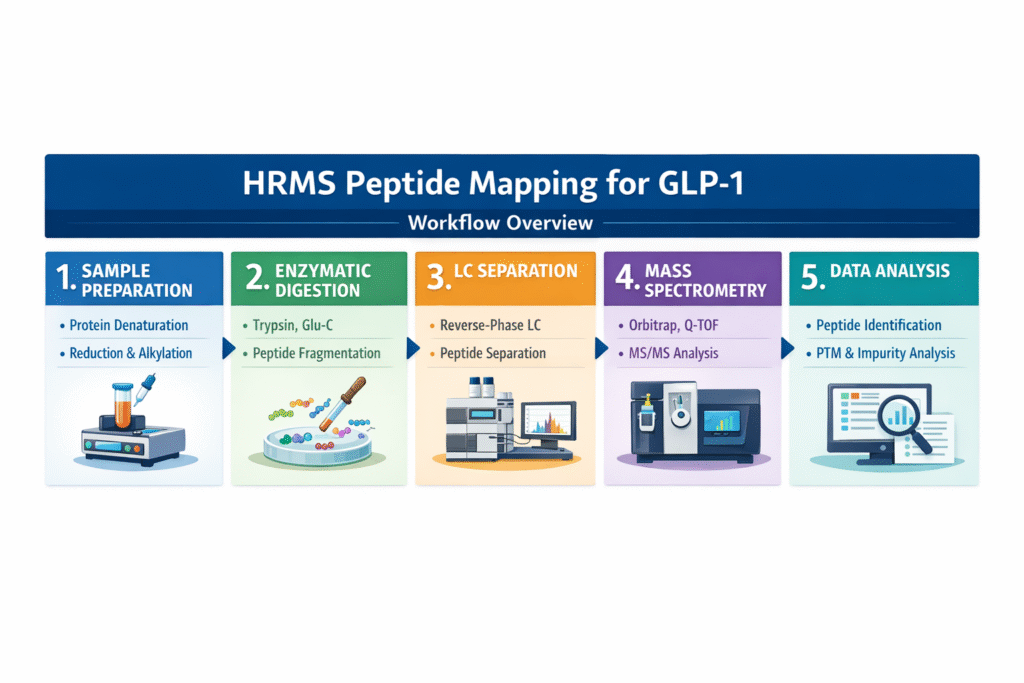
3: Workflow of HRMS Peptide Mapping for GLP-1
The HRMS Peptide Mapping GLP-1 workflow consists of five key steps—sample preparation, enzymatic digestion, chromatographic separation, mass spectrometry detection, and data analysis—to ensure accurate structural characterization.
For complete mapping strategies, explore peptide mapping of GLP-1 peptides and semaglutide peptide mapping.
Each step plays a critical role in generating reliable, high-resolution data for GLP-1 peptide analysis.
Step-by-Step Process:
1. Sample Preparation
Proper sample preparation ensures peptide stability and reproducibility of results.
- Protein Denaturation:
Unfolds the GLP-1 peptide to expose cleavage sites. - Reduction and Alkylation:
- Reduction breaks disulfide bonds (if present).
- Alkylation prevents reformation, improving digestion efficiency.
2. Enzymatic Digestion
Controlled enzymatic digestion generates predictable peptide fragments for mapping.
- Common Enzymes:
- Trypsin
- Glu-C
- Purpose:
- Produces specific cleavage patterns
- Enables sequence coverage and reproducibility
3. Chromatographic Separation (LC)
Liquid chromatography separates peptide fragments before MS detection.
- Technique:
Reverse-phase LC (RP-LC) - Separation Principle:
Based on hydrophobic interactions - Outcome:
Reduces sample complexity and improves detection sensitivity
4. Mass Spectrometry Detection
High-resolution MS accurately detects and characterizes peptide fragments.
- Instruments Used:
- Orbitrap
- Q-TOF
- Key Features:
- High mass accuracy (ppm level)
- MS/MS fragmentation for sequence confirmation
- Detection of PTMs and impurities
5. Data Analysis
Advanced software tools convert raw data into meaningful peptide information.
- Peptide Identification:
Matches observed spectra with theoretical sequences - PTM Analysis:
Detects modifications such as oxidation or deamidation - Impurity Profiling:
Identifies unknown or low-level variants
This structured workflow ensures that HRMS Peptide Mapping GLP-1 delivers precise, reproducible, and regulatory-compliant results for pharmaceutical development and quality control.
4: Best Practices for HRMS Peptide Mapping GLP-1
Following best practices in HRMS Peptide Mapping GLP-1 ensures high accuracy, reproducibility, and regulatory compliance across analytical workflows.
For outsourcing and CRO-based expertise, check CRO for GLP-1 peptide characterization and outsource GLP-1 peptide sequencing services.
Implementing optimized methods at every stage—from sample preparation to data analysis—minimizes variability and improves confidence in peptide characterization.
1. Optimize Sample Preparation
Proper sample preparation is critical to maintain peptide integrity and ensure consistent results.
- Use high-purity reagents to avoid background interference
- Prevent contamination and degradation during handling
- Maintain controlled pH and temperature to preserve peptide stability
2. Select Appropriate Enzymes
Choosing the right enzyme ensures efficient and reproducible peptide digestion.
- Select enzymes (e.g., Trypsin, Glu-C) based on GLP-1 sequence specificity
- Ensure complete digestion to achieve full sequence coverage
- Validate digestion efficiency through test runs and controls
3. Optimize LC Conditions
Efficient chromatographic separation enhances detection sensitivity and resolution.
- Use high-resolution columns (C18) for better separation
- Maintain consistent gradient profiles for reproducibility
- Control flow rate and column temperature to stabilize retention times
4. Fine-Tune HRMS Parameters
Optimizing instrument settings is essential for accurate mass detection and characterization.
- Set high resolving power (≥30,000) for precise mass measurement
- Optimize ionization conditions (ESI) to improve signal intensity
- Use appropriate scan ranges to capture all relevant peptide ions
5. Ensure Calibration and Validation
Regular calibration and validation ensure data reliability and compliance.
- Perform routine instrument calibration
- Use certified reference standards for accuracy
- Conduct system suitability tests before analysis
6. Implement Robust Data Analysis
Accurate data interpretation is crucial for reliable peptide mapping results.
- Use validated and compliant software tools
- Apply strict acceptance criteria for peptide identification
- Confirm findings with MS/MS fragmentation data
By adhering to these best practices, laboratories can maximize the effectiveness of HRMS Peptide Mapping GLP-1, ensuring high-quality data that meets both scientific and regulatory expectations.

5: Common Challenges and Solutions
HRMS Peptide Mapping GLP-1 can encounter challenges in digestion efficiency, sensitivity, and data interpretation—but these can be effectively managed with optimized strategies.
For impurity-focused analysis, refer to GLP-1 peptide impurity sequencing analysis.
Understanding these challenges and applying targeted solutions ensures reliable, high-quality analytical outcomes.
Key Challenges in HRMS Peptide Mapping GLP-1
- Incomplete Enzymatic Digestion:
Leads to poor sequence coverage and unreliable mapping results. - Signal Suppression:
Matrix effects or co-eluting compounds can reduce ionization efficiency, impacting detection sensitivity. - Complex Data Interpretation:
Large datasets with overlapping spectra make peptide identification and PTM analysis difficult. - Detection of Low-Level Impurities:
Trace impurities may be masked by dominant peptide signals.
Effective Solutions and Best Approaches
- Optimize Digestion Protocols:
- Adjust enzyme-to-substrate ratio
- Optimize incubation time and conditions
- Use complementary enzymes if needed
- Use Internal Standards:
- Improve quantification accuracy
- Monitor system performance and variability
- Enhance Chromatographic Separation:
- Optimize LC gradients
- Use high-resolution columns
- Reduce co-elution of interfering compounds
- Apply Advanced Data Processing Algorithms:
- Use AI-enabled or validated software tools
- Automate peak detection and peptide identification
- Improve confidence in PTM and impurity analysis
By proactively addressing these challenges, laboratories can significantly enhance the performance and reliability of HRMS Peptide Mapping GLP-1, ensuring accurate characterization and compliance with regulatory standards.
6: Regulatory Considerations
HRMS Peptide Mapping GLP-1 must comply with global regulatory guidelines to ensure data integrity, method reliability, and product quality throughout the analytical lifecycle.
Regulatory compliance is essential for supporting drug development, quality control, and successful submissions to health authorities.
Learn more about compliance here:
- Regulatory requirements for GLP-1 peptide characterization
- GLP-1 peptide characterization regulatory requirements
Key Regulatory Guidelines
- ICH Q6B (Specifications for Biotechnological Products):
Defines acceptance criteria for identity, purity, and quality attributes of peptide-based therapeutics. - ICH Q2(R2) (Analytical Method Validation):
Provides updated guidance on validating analytical methods, including robustness and lifecycle management. - FDA and EMA Guidelines:
Outline expectations for peptide characterization, impurity profiling, and analytical method performance in regulated environments.
Core Compliance Requirements
- Method Validation:
Ensure the method is scientifically sound and reproducible by evaluating:- Accuracy
- Precision
- Specificity
- Linearity and robustness
- Documentation and Traceability:
Maintain complete records of:- Sample preparation
- Instrument parameters
- Data processing steps
This ensures full audit readiness and traceability.
- Data Integrity (ALCOA Principles):
Analytical data must be:- Attributable
- Legible
- Contemporaneous
- Original
- Accurate
Adhering to these regulatory considerations ensures that HRMS Peptide Mapping GLP-1 delivers trustworthy, high-quality data that meets global compliance standards and supports safe, effective peptide therapeutics.
7: Applications of HRMS Peptide Mapping in GLP-1 Analysis
HRMS Peptide Mapping GLP-1 is widely applied across drug development, manufacturing, and quality control to ensure structural accuracy, purity, and consistency of GLP-1 therapeutics.
Its high sensitivity and resolution make it an indispensable tool throughout the product lifecycle.
For stability-focused insights, visit GLP-1 peptide stability analytical methods.
For sequencing services, explore GLP-1 peptide sequencing CRO services.
Key Applications:
- Structural Characterization:
Confirms the complete amino acid sequence and detects structural variants or modifications, ensuring the integrity of GLP-1 peptides. - Impurity Profiling:
Identifies process-related impurities, degradation products, and trace-level contaminants that may affect safety and efficacy. - Stability Studies:
Monitors chemical and physical changes in GLP-1 peptides under various storage and stress conditions, supporting shelf-life determination. - Biosimilar Comparison:
Enables detailed comparison between innovator and biosimilar GLP-1 products to demonstrate structural and functional equivalence. - Batch Release Testing:
Ensures consistency and compliance by verifying that each production batch meets predefined quality specifications.
By leveraging these applications, HRMS Peptide Mapping GLP-1 plays a critical role in delivering safe, effective, and high-quality peptide-based therapeutics to the market.
8: Why Choose ResolveMass Laboratories Inc.
ResolveMass Laboratories Inc. provides expert-driven HRMS peptide mapping services with a focus on accuracy, compliance, and reliability.
Our Expertise:
- Advanced HRMS instrumentation (Orbitrap, Q-TOF)
- Experienced analytical scientists
- Regulatory-compliant workflows
- Customized method development
What Sets Us Apart:
- High data accuracy and reproducibility
- Fast turnaround time
- Comprehensive reporting
- Strong regulatory support
Conclusion:
HRMS Peptide Mapping GLP-1 is an essential analytical technique for ensuring the structural integrity, safety, and efficacy of GLP-1 therapeutics.
By integrating advanced analytical workflows and leveraging specialized services such as GLP-1 peptide sequencing and analytical characterization of GLP-1 peptide drugs, organizations can achieve superior analytical outcomes.
ResolveMass Laboratories Inc. stands as a trusted partner in delivering high-quality HRMS peptide mapping solutions tailored to the evolving needs of the pharmaceutical and biotechnology industries.
Frequently Asked Questions:
HRMS Peptide Mapping GLP-1 is used to confirm peptide sequence, identify modifications, and detect impurities with high accuracy.
It plays a critical role in drug development, quality control, and regulatory submissions. The technique ensures structural integrity and consistency of GLP-1 therapeutics across batches.
HRMS is important because it provides superior mass accuracy and sensitivity compared to conventional MS.
It allows detection of low-level impurities, sequence variants, and post-translational modifications that are essential for ensuring drug safety and efficacy.
Trypsin and Glu-C are the most commonly used enzymes for GLP-1 peptide mapping.
These enzymes generate predictable peptide fragments, enabling accurate sequence coverage and reproducible analytical results.
The key steps include sample preparation, enzymatic digestion, LC separation, MS detection, and data analysis.
Each step contributes to generating high-quality, reliable data for peptide characterization and impurity profiling.
Common challenges include incomplete digestion, signal suppression, and complex data interpretation.
These issues can impact accuracy but can be minimized with optimized workflows, proper sample preparation, and advanced data analysis tools.
HRMS detects impurities with high sensitivity, even at trace levels.
It differentiates closely related compounds and identifies degradation products, ensuring the purity and safety of GLP-1 peptides.
Reference
- Howard JW. The development of mass spectrometry-based methodologies for the high throughput quantitation of peptides in biological matrices (Doctoral dissertation, Loughborough University).https://repository.lboro.ac.uk/articles/The_development_of_mass_spectrometry-based_methodologies_for_the_high_throughput_quantitation_of_peptides_in_biological_matrices/9397871/files/17013215.pdf
- Darji P. Key Analytical Challenges in GLP-1 Peptide Drugs and How CDMOs Solve Them.https://resolvemass.ca/glp-1-peptide-analytical-characterization/
- Evers A, Bossart M, Pfeiffer-Marek S, Elvert R, Schreuder H, Kurz M, Stengelin S, Lorenz M, Herling A, Konkar A, Lukasczyk U. Dual glucagon-like peptide 1 (GLP-1)/glucagon receptor agonists specifically optimized for multidose formulations. Journal of medicinal chemistry. 2018 Jun 7;61(13):5580-93.https://pubs.acs.org/doi/abs/10.1021/acs.jmedchem.8b00292
- Kim SH, Kim SS, Kim HJ, Park EJ, Na DH. Peptide mapping analysis of synthetic semaglutide and liraglutide for generic development of drugs originating from recombinant DNA technology. Journal of Pharmaceutical and Biomedical Analysis. 2025 Apr 15;256:116682.https://www.sciencedirect.com/science/article/pii/S0731708525000238


