Introduction
In pharmaceutical manufacturing and chemical synthesis, Identification of In-Process Organic Compounds using LCMS plays a vital role in maintaining product quality, improving process efficiency, and ensuring regulatory compliance. Liquid Chromatography–Mass Spectrometry (LC-MS) is widely recognized as a powerful analytical technique for this purpose due to its high sensitivity, specificity, and versatility in analyzing complex mixtures. As a leading Contract Research Organization (CRO) specializing in custom synthesis and analytical services, Resolvemass Laboratories utilizes advanced LC-MS technology for the accurate Identification of In-Process Organic Compounds using LCMS, helping clients monitor reaction progress, characterize intermediates and impurities, and optimize their manufacturing processes to achieve consistent, high-quality results.
Explore our specialized services for complex molecules: > Custom Polymer Synthesis
Share via:
Article Summary
- LC-MS is a powerful technique used to identify and monitor intermediates, impurities, and degradation products during pharmaceutical manufacturing.
- It helps improve process efficiency, product quality, and ensures regulatory compliance.
- The LC-MS workflow includes sample preparation, separation, detection, and data analysis for accurate compound identification.
- Reference standards, validated methods, and spectral databases improve reliability and confidence in results.
- Advanced tools like HRMS and PAT enhance process monitoring, accuracy, and overall manufacturing control.
Understanding In-Process Organic Compound Identification
In-process organic compounds include starting materials, intermediates, by-products, and degradation products formed during chemical reactions. Accurate identification and quantification of these compounds are essential for:
- Process Optimization: Understanding reaction pathways and identifying key intermediates and by-products can help optimize reaction conditions, improve yields, and reduce waste.
- Quality Control: Ensuring that the final product meets specified quality standards by monitoring and controlling impurities and degradation products throughout the manufacturing process.
- Regulatory Compliance: Meeting regulatory requirements for impurity profiling and ensuring the safety and efficacy of pharmaceutical products.
Need detailed impurity analysis? Learn more here: > Impurity Profiling Using LCMS
LC-MS combines the separation capabilities of liquid chromatography (LC) with the detection and identification power of mass spectrometry (MS). This synergy makes LC-MS an ideal tool for the analysis of complex mixtures, providing detailed information on the composition and structure of organic compounds. Key advantages of LC-MS include:
- High Sensitivity and Specificity: LC-MS can detect and quantify trace levels of compounds with high specificity, distinguishing between structurally similar molecules.
- Versatility: Suitable for a wide range of compounds, from small molecules to large biomolecules, in various matrices.
- Structural Elucidation: MS provides molecular weight and structural information, aiding in the identification of unknown compounds.
Importance of Method Development and Validation in LC-MS
Method development and validation are fundamental steps that ensure LC-MS delivers reliable and reproducible results during in-process compound identification. A properly developed method considers factors such as chromatographic resolution, ionization efficiency, and signal stability. Scientists carefully evaluate different columns, solvent systems, and gradients to achieve optimal separation of all relevant compounds, including minor impurities and closely related intermediates. This step minimizes the risk of co-eluting compounds that could lead to inaccurate identification or quantification.
Method validation further confirms that the developed LC-MS method is suitable for its intended purpose. Key validation parameters include accuracy, precision, specificity, linearity, detection limits, and robustness. By validating these parameters, laboratories ensure consistent performance even when small variations occur in operating conditions. This is especially important in pharmaceutical environments, where regulatory authorities require documented proof that analytical methods can reliably detect impurities and monitor process changes. A validated LC-MS method ultimately builds confidence in the analytical data used for critical manufacturing decisions.
Discover our approach to comprehensive method validation: > Method Validation and Development Services
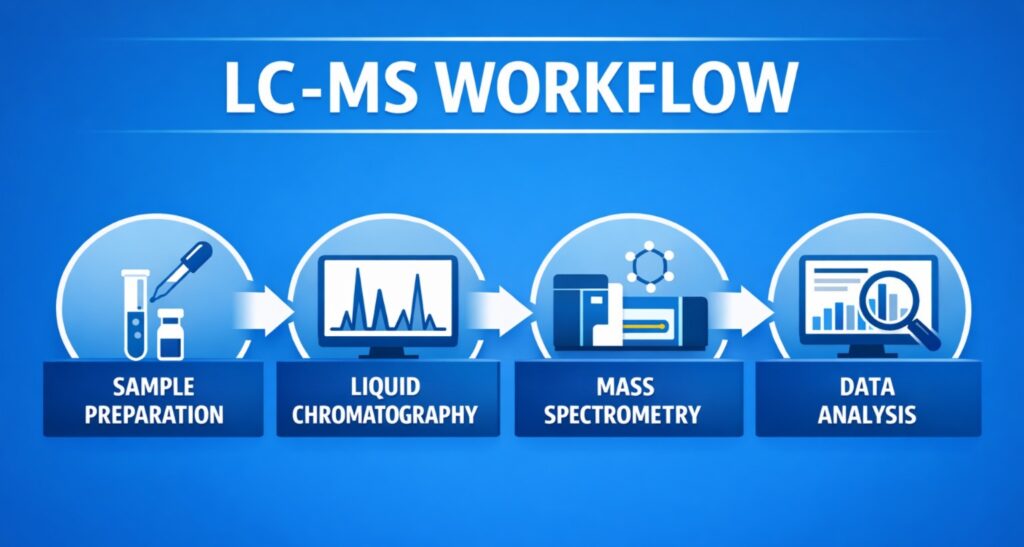
LC-MS Workflow for In-Process Compound Identification
At Resolvemass Laboratories, our LC-MS workflow for identifying in-process organic compounds involves several key steps:
1. Sample Preparation
Proper sample preparation is crucial for accurate LC-MS analysis. Depending on the sample matrix and target compounds, preparation methods may include:
- Filtration: Removing particulate matter to prevent clogging of the LC column and MS ion source.
- Extraction: Isolating target compounds from complex matrices using techniques such as solid-phase extraction (SPE) or liquid-liquid extraction (LLE).
- Dilution: Adjusting the concentration of the sample to fall within the dynamic range of the LC-MS system.
2. Liquid Chromatography
LC separates the components of the sample based on their chemical properties, such as polarity and hydrophobicity. Key considerations for LC include:
- Column Selection: Choosing the appropriate stationary phase (e.g., reversed-phase, normal-phase, or ion-exchange) to achieve optimal separation of target compounds.
- Mobile Phase Optimization: Adjusting the composition and gradient of the mobile phase to enhance resolution and peak shape.
- Flow Rate and Temperature: Optimizing these parameters to balance separation efficiency and analysis time.
3. Mass Spectrometry
MS detects and identifies the separated compounds based on their mass-to-charge (m/z) ratios. Key aspects of MS analysis include:
- Ionization Technique: Choosing the appropriate ionization method (e.g., electrospray ionization (ESI) or atmospheric pressure chemical ionization (APCI)) based on the chemical nature of the compounds.
- Mass Analyzer: Selecting the mass analyzer (e.g., quadrupole, time-of-flight (TOF), or orbitrap) to achieve the desired resolution, accuracy, and sensitivity.
- Data Acquisition: Collecting full-scan MS data or targeted MS/MS data to obtain detailed information on the molecular weight and structure of the compounds.
4. Data Analysis and Interpretation
The data generated by LC-MS are processed and interpreted using advanced software tools. Key steps include:
- Peak Identification: Identifying peaks in the chromatogram and correlating them with the corresponding mass spectra.
- Compound Identification: Comparing the obtained mass spectra with reference libraries or using de novo analysis to identify unknown compounds.
- Quantification: Determining the concentration of identified compounds using calibration curves and internal standards.
- Structural Elucidation: Interpreting MS/MS fragmentation patterns to elucidate the structure of unknown compounds.
For rapid screening without prior chromatography: > Direct Infusion Services
Role of Reference Standards and Databases in Compound Identification
Reference standards and spectral databases play a vital role in improving the accuracy and confidence of LC-MS compound identification. Authentic reference standards provide direct comparison points for retention time, mass spectra, and fragmentation patterns. When an unknown compound matches a known standard, analysts can confirm its identity with a high level of certainty. This approach is especially useful for identifying known intermediates, degradation products, and regulated impurities during pharmaceutical manufacturing.
In addition to physical standards, digital mass spectral libraries significantly enhance analytical efficiency. These libraries contain thousands of stored spectra that can be automatically compared with experimental data using advanced software. This allows rapid screening and tentative identification of unknown compounds even when reference standards are not immediately available. As spectral databases continue to expand, they enable faster decision-making, improved troubleshooting, and better process understanding, making LC-MS an even more powerful tool for routine and investigative analysis.
Applications of LC-MS in In-Process Compound Identification
Resolvemass Laboratories utilizes LC-MS for a wide range of applications in process control and quality assurance:
1. Reaction Monitoring
LC-MS enables real-time monitoring of chemical reactions, providing insights into reaction kinetics, intermediate formation, and by-product profiles. This information is invaluable for optimizing reaction conditions and improving yields.
2. Impurity Profiling
Identifying and quantifying impurities at various stages of the manufacturing process helps ensure product quality and compliance with regulatory standards. LC-MS can detect trace impurities that may impact the safety and efficacy of the final product.
3. Degradation Studies
LC-MS is used to study the stability of compounds under different conditions, identifying degradation products and elucidating degradation pathways. This information is essential for developing robust formulations and ensuring product shelf-life.
4. Process Development and Scale-Up
During process development and scale-up, LC-MS provides detailed information on the composition of reaction mixtures, helping to identify potential issues and optimize process parameters for large-scale production.
Specialized safety analysis for pharmaceuticals: > Nitrosamine Analysis Services
Integration of LC-MS with Process Analytical Technology (PAT)
The integration of LC-MS with Process Analytical Technology (PAT) has significantly improved real-time monitoring capabilities in modern pharmaceutical manufacturing. PAT focuses on designing, analyzing, and controlling manufacturing processes through timely measurements of critical quality attributes. When LC-MS is incorporated into PAT frameworks, it provides highly detailed molecular-level information about reaction mixtures. This allows manufacturers to detect process deviations immediately and implement corrective actions before product quality is affected.
This real-time insight supports a more proactive approach to process control rather than relying solely on end-product testing. Continuous monitoring helps maintain consistency across batches, reduces material loss, and improves overall manufacturing efficiency. Additionally, regulatory agencies encourage the adoption of PAT tools like LC-MS because they promote better process understanding and quality assurance. As a result, LC-MS is increasingly becoming a central component of smart, automated, and highly controlled production environments.
Learn about LCMS applications in natural products: > LCMS Analysis of Plant Extracts
Case Study: Enhancing Process Control in Pharmaceutical Manufacturing
A pharmaceutical company approached Resolvemass Laboratories for LC-MS analysis of their manufacturing process for a new drug compound. The project involved:
Challenges
- Complex Reaction Pathways: The synthesis involved multiple steps with potential for intermediate formation and by-product generation.
- Trace Impurities: Detecting and controlling trace impurities to meet stringent regulatory standards.
- Scalability: Ensuring that the optimized process could be scaled up for commercial production without compromising quality.
Solution
- Comprehensive LC-MS Analysis: We performed LC-MS analysis at various stages of the synthesis, identifying key intermediates, by-products, and impurities.
- Reaction Monitoring: Real-time monitoring of the reaction enabled optimization of reaction conditions, improving yields and reducing by-products.
- Impurity Profiling: Detailed impurity profiling ensured that trace impurities were identified and controlled, meeting regulatory requirements.
- Process Optimization: The insights gained from LC-MS analysis were used to refine the process, ensuring scalability and consistent product quality.
Outcome
The successful identification and control of in-process organic compounds enabled the pharmaceutical company to optimize their manufacturing process, ensuring high-quality production and regulatory compliance. Our comprehensive LC-MS services facilitated the development of a robust and scalable process, accelerating the time-to-market for their new drug compound.
Advantages of High-Resolution Mass Spectrometry (HRMS) in In-Process Analysis
High-Resolution Mass Spectrometry (HRMS) has further enhanced the capabilities of LC-MS in identifying in-process organic compounds with exceptional precision. Unlike conventional mass analyzers, HRMS provides highly accurate mass measurements, often within parts-per-million (ppm) accuracy. This level of precision enables scientists to determine exact molecular formulas, even for unknown compounds present at very low concentrations. Such accuracy is extremely valuable when investigating unexpected impurities or complex degradation products.
Another important advantage of HRMS is its ability to distinguish between compounds with nearly identical masses. This improves confidence in compound identification and reduces the chances of misinterpretation. HRMS also supports retrospective data analysis, allowing previously acquired data to be re-examined for new impurities without repeating experiments. This capability saves time, reduces costs, and improves the overall efficiency of process investigations. As pharmaceutical processes become more complex, HRMS continues to play a critical role in ensuring product quality and safety.
Get the highest level of analytical precision: > High-Resolution Mass Spectrometry (HRMS) Analysis
Conclusion
The identification of in-process organic compounds is crucial for ensuring process control, product quality, and regulatory compliance in pharmaceutical and chemical manufacturing. LC-MS offers unparalleled sensitivity, specificity, and versatility for this purpose. At Resolvemass Laboratories, we leverage advanced LC-MS technology to provide comprehensive identification and characterization services, helping clients optimize their processes and achieve consistent, high-quality outcomes. Our commitment to excellence, innovation, and customer satisfaction makes us a trusted partner for all your analytical needs.
In addition, ongoing advancements such as high-resolution mass spectrometry, improved spectral databases, and integration with process analytical technologies have further strengthened the effectiveness of LC-MS in modern manufacturing environments. These innovations enable faster identification, improved accuracy, and real-time process monitoring, allowing manufacturers to respond quickly to process variations. By providing deeper insights into reaction pathways, impurity formation, and compound stability, LC-MS supports continuous process improvement and risk reduction. As the pharmaceutical and chemical industries continue to evolve, LC-MS will remain an essential tool for ensuring safe, efficient, and high-quality production.
Contact Us
For more information about our LC-MS services and how we can assist with your process control and quality assurance needs, please Resolvemass laboratories & contact us
Frequently Asked Questions
LC-MS is an analytical technique that combines liquid chromatography separation with mass spectrometry detection. It helps identify and measure organic compounds present during different stages of a chemical reaction. This technique is widely used because it provides accurate molecular information and can detect compounds even at very low concentrations. It is especially useful for monitoring intermediates, impurities, and degradation products.
LC-MS allows scientists to analyze reaction mixtures and track the formation and disappearance of compounds during manufacturing. This helps in understanding reaction progress and identifying unwanted impurities. By monitoring the process in real time or at specific stages, manufacturers can adjust conditions to improve product quality. This ensures consistent and reliable production outcomes.
Identifying in-process compounds helps ensure that the desired product is being formed correctly and safely. It also helps detect impurities or unwanted by-products that could affect product quality. This information allows scientists to optimize reaction conditions and improve efficiency. It is also necessary for meeting regulatory requirements in pharmaceutical manufacturing.
LC-MS is known for its high sensitivity and can detect compounds present in very small amounts. It can identify trace impurities that may not be visible using other methods. This level of sensitivity helps ensure accurate analysis and reliable results. It is particularly important in pharmaceutical applications where impurity control is critical.
Proper sample preparation ensures that the compounds of interest can be analyzed accurately without interference. It may involve filtration, dilution, or extraction to remove unwanted materials. Good preparation improves detection quality and protects the instrument from damage. This step is essential for obtaining consistent and meaningful results.
Reference:
- Blaženović, I., Kind, T., Ji, J., & Fiehn, O. (2018). Software tools and approaches for compound identification of LC-MS/MS data in metabolomics. Metabolites, 8(2), 31. https://doi.org/10.3390/metabo8020031
- Alanazi, S. (2025). Recent advances in liquid chromatography–mass spectrometry (LC–MS) applications in biological and applied sciences. Analytical Science Advances, 6(1), e70024. https://doi.org/10.1002/ansa.70024
- Yang, Y., Peng, Y., Chang, Q., Dan, C., Guo, W., & Wang, Y. (2016). Selective identification of organic iodine compounds using liquid chromatography–high resolution mass spectrometry. Analytical Chemistry, 88(2), 1275–1280. https://doi.org/10.1021/acs.analchem.5b03694



