Introduction:
The development of long-acting injectable formulations requires a strong scientific bridge between laboratory testing and human pharmacokinetics. IVIVC for Leuprolide Depot plays a critical role in connecting complex PLGA-based in vitro dissolution behavior with in vivo drug exposure. A well-designed correlation integrates mathematical deconvolution, mechanistic release modeling, and robust dissolution testing to predict clinical performance. Establishing a Level A correlation is widely accepted as the primary regulatory pathway for demonstrating bioequivalence and supporting biowaivers. The predictive reliability of IVIVC for Leuprolide Depot depends on a reproducible in vitro release method and a validated transformation model linking in vitro and in vivo datasets. A properly developed correlation helps identify formulation variability early, reduces development risk, and supports regulatory justification without repeated clinical trials.
Learn more about the specific steps in the development of generic drugs: Generic Drug Development Process for ANDA
Share via:
Summary:
- IVIVC for Leuprolide Depot enables predictive bioequivalence by linking PLGA in vitro release with in vivo pharmacokinetics using Level A correlation and mechanistic modeling.
- Regulatory success depends on robust IVIVC strategy, including product-specific guidance, PK metrics (Cmax, AUC), and parallel bioequivalence study designs.
- Level A IVIVC supports biowaivers and strength scaling, reducing clinical study burden when Q1/Q2/Q3 sameness and dissolution similarity are demonstrated.
- PLGA microstructure and composition control release behavior, with polymer ratio, molecular weight, and morphology directly impacting burst, lag, and erosion phases.
- Biopredictive dissolution and mathematical modeling improve predictability, using USP Apparatus 4, deconvolution methods, and time-scaling correlations.
- PBPK modeling and optimized manufacturing enhance IVIVC reliability, while addressing peptide stability, acylation, and process variability in depot development.
Strategic Regulatory Framework for Leuprolide Bioequivalence
The regulatory pathway for generic leuprolide acetate requires demonstration of bioequivalence using both in vivo pharmacokinetic studies and detailed in vitro characterization. For ResolveMass Laboratories Inc., implementing IVIVC for Leuprolide Depot provides a scientific bridge between formulation attributes and clinical outcomes. This approach ensures that batches meet the required 80.00–125.00% confidence interval standards. The framework emphasizes consistent drug release, predictable pharmacokinetics, and uniform product performance across strengths. Regulatory agencies expect strong scientific reasoning linking formulation design with in vivo exposure. A structured IVIVC strategy reduces the need for repeated clinical trials and supports lifecycle management. It also helps evaluate post-approval changes within regulatory limits.
Explore the essential requirements for a successful generic drug application: Requirements for ANDA Submission in Generic Drugs
Product-Specific Guidance (PSG) and Bioequivalence Metrics
The FDA has released multiple product-specific guidances for leuprolide acetate, emphasizing strength-specific bioequivalence studies. For example, the February 2018 guidance (revised November 2021) for the 11.25 mg 3-month depot recommends randomized, single-dose, parallel studies in patients with endometriosis. A key parameter in these studies is the $AUC_{7-t}$ metric, which evaluates sustained drug release after the initial burst phase. These pharmacokinetic endpoints capture early exposure and long-term therapeutic coverage. Appropriate sampling schedules are required to define lag phase and erosion-controlled release. Statistical comparisons must confirm equivalence in both peak exposure and total exposure. These requirements ensure that generic formulations match reference product performance.
| Dosage Form | Targeted Patients | Primary BE Analytes | Essential PK Parameters |
| 1-Month Depot (3.75 mg) | Endometriosis | Leuprolide, Norethindrone | C{max}, AUC{0-t}, AUC{7-t} |
| 3-Month Depot (11.25 mg) | Endometriosis | Leuprolide, Norethindrone | C{max}, AUC_{0-t}, AUC{7-t} |
| 1-Month Depot (7.5 mg) | Prostatic Carcinoma | Leuprolide | C{max}, AUC{0-t}, AUC{7-t} |
| 3-Month Depot (30 mg) | Prostatic Carcinoma | Leuprolide | C_{max}, AUC{0-t}, AUC{7-t} |
| 6-Month Depot (45 mg) | Prostatic Carcinoma | Leuprolide | C_{max}, AUC{0-t}, AUC{7-t} |
Parallel study designs are required due to the long-acting nature of depot formulations. Crossover designs are impractical because of long washout periods. Balanced patient populations help reduce variability in pharmacokinetic interpretation. Hormone suppression differences and baseline physiology can influence results. Proper stratification and sample size planning improve predictability. These considerations directly support development of reliable IVIVC models.
Biowaiver Strategies and Strength Scaling Using IVIVC for Leuprolide Depot
Establishing Level A IVIVC for Leuprolide Depot can support biowaiver requests for additional strengths. For instance, guidance for the 30 mg strength suggests that a waiver for the 22.5 mg strength may be granted if bioequivalence is demonstrated for the 30 mg dose, Q1/Q2 sameness is shown, and comparative dissolution profiles are provided. This approach reduces clinical burden and speeds development timelines. Strength scaling must be supported by proportional formulation design and consistent release mechanisms. Dissolution similarity testing is essential for demonstrating equivalence. Accurate IVIVC modeling ensures proportional exposure across strengths.
Understand the regulatory filings and requirements specifically for Leuprolide: Leuprolide Depot ANDA Requirements
Microstructural Equivalence (Q3) and PLGA Architecture
Achieving Q3 microstructural equivalence is essential for predictive IVIVC for Leuprolide Depot, as polymer matrix structure directly controls release kinetics. Even when Q1/Q2 composition matches the reference product, manufacturing differences can produce distinct microstructures. Parameters such as porosity, particle size distribution, and polymer density strongly influence drug diffusion. Processing conditions including solvent removal rate and emulsification energy affect microsphere morphology. These structural variations may alter burst release and lag phase duration. Therefore, Q3 sameness is a key determinant of IVIVC predictability.
The Impact of PLGA Composition and Sequence
PLGA is the primary excipient controlling leuprolide release. The lactide:glycolide ratio, molecular weight ($M_w$), and polymer architecture determine degradation and diffusion behavior. Different L:G ratios such as 50:50 versus 75:25 influence hydrophobicity and hydrolysis rates. These changes affect water penetration, swelling, and erosion dynamics. Molecular weight impacts matrix density and diffusion resistance. End-group chemistry also modifies degradation behavior. Together, these factors define the mechanistic release profile required for IVIVC modeling.
| Polymer Attribute | Effect on Leuprolide Release | Impact on IVIVC |
| L:G Ratio (50:50) | Faster degradation due to higher glycolide content | Shorter lag phase, faster secondary release |
| L:G Ratio (75:25) | Slower degradation, more hydrophobic | Longer lag phase, sustained long-term delivery |
| Molecular Weight (Mw) | High Mw increases matrix density | Slows diffusion and erosion-controlled release |
| End-Group Chemistry | Acid-capped vs. Ester-capped | Acid-capped polymers degrade faster (autocatalysis) |
| Polymer Architecture | Linear vs. Star-branched | Influences chain entanglement and erosion kinetics |
Detailed characterization of PLGA is essential. Minor sequence variations can change microclimate pH inside microspheres. This affects peptide stability and release behavior. Analytical tools such as Gel Permeation Chromatography (GPC) are used to measure molecular weight distribution. Sequence analysis confirms similarity to the reference product. These evaluations support consistent degradation patterns for predictive IVIVC.
Explore specialized services for complex peptide drug development: Specialized Peptide CDMO Services
Advanced Morphological Characterization (Q3 Sameness)
To establish Q3 sameness, manufacturers analyze particle size distribution, surface morphology, internal porosity, and drug distribution. Scanning Electron Microscopy (SEM) and nitrogen adsorption techniques are commonly used. Uniform pore distribution typically results in predictable burst release and lag phase behavior. These measurements connect microstructure with release kinetics. Morphological consistency improves IVIVC reliability. Quantitative imaging also supports regulatory submissions.
Sequential Semi-Solvent Vapor (SSV), also known as SASSI/SAVI, allows evaluation of morphological changes before and after environmental exposure. This method visualizes polymer rearrangement during degradation. It helps identify mechanistic signatures associated with release phases. Such understanding strengthens IVIVC model accuracy and improves prediction of in vivo performance.
Deep dive into the analytical hurdles of Leuprolide development: Leuprolide Depot Analytical Challenges
Mechanistic Release Stages and IVIVC for Leuprolide Depot Predictability
Leuprolide depot formulations typically show a triphasic release pattern including initial burst, lag phase, and secondary erosion-controlled release. A multi-stage IVIVC for Leuprolide Depot treats each phase as a separate kinetic event. Predictive accuracy improves when dissolution methods simulate transitions between these phases. Each stage reflects different physical processes within the PLGA matrix. Capturing these transitions is essential for accurate modeling.
Phase I: Initial Burst and Surface Phenomena
The initial burst occurs within the first 24 hours and is driven by surface-associated drug. Manufacturing methods such as double emulsion or spray drying strongly influence this phase. Polymer concentration during solvent evaporation also affects burst magnitude. Excessive burst may lead to testosterone flare in prostate cancer patients. In vivo absorption can mask this effect, making Level A correlation challenging. Controlling surface drug loading is therefore critical.
Phase II: The Lag Phase and Polymer Hydration
After the burst, a lag phase with minimal release is observed. Water penetrates the PLGA matrix and initiates ester bond hydrolysis. Duration depends on polymer molecular weight and L:G ratio. In vitro lag phase may be shortened using acidic buffers or plasticizers such as triethyl citrate. Accurate modeling must capture this delay. Failure to represent lag time may overpredict early exposure.
Phase III: Secondary Release and Autocatalytic Erosion
The final stage is dominated by bulk erosion of PLGA. Acidic oligomers reduce internal pH and accelerate hydrolysis through autocatalysis. This stage often challenges IVIVC models due to differences between in vitro and in vivo buffering.
Rate of Hydrolysis
𝑘 [𝐻+] [Ester]
Secondary release typically follows near zero-order kinetics. Accurate point-to-point correlation requires matching erosion rates. Physiological buffering may alter degradation dynamics. Model calibration improves prediction of $C_{max}$ and $AUC$.
Establishing Level A IVIVC for Leuprolide Depot: Mathematical Framework
Level A IVIVC for Leuprolide Depot establishes a point-to-point relationship between fraction dissolved in vitro ($F_d$) and fraction absorbed in vivo ($F_a$). This allows prediction of plasma concentration-time profiles using dissolution data. Validation must meet FDA percent prediction error criteria. Reliable datasets and statistical robustness improve regulatory acceptance.
Deconvolution Techniques: Wagner-Nelson and Loo-Riegelman
Wagner-Nelson Method
𝐹𝑎(t) = (𝐶𝑡 + 𝑘𝑒 ∫0t 𝐶(τ)dτ) / (𝑘𝑒 ∫0∞ 𝐶(τ)dτ)
This method is suitable for one-compartment models and simplifies absorption profiling. It provides linear transformation for correlation development and is frequently applied to depot datasets. The approach is computationally efficient and supports early IVIVC modeling.
Loo-Riegelman Method
This method is preferred for multi-compartment kinetics. Leuprolide shows complex distribution following injection. The method accounts for drug transfer between compartments and improves absorption estimates. It enhances precision in IVIVC development.
Time-Scaling and Non-Linear Correlation Functions
In vitro release often differs from in vivo absorption rates, requiring time scaling. Non-linear regression such as the Hill equation aligns $t_{50%}$ values. This improves correlation between datasets and enhances predictability.
| Model Component | Description | Parameter Example |
| Release Compartment | Describes the drug leaving the microsphere | E1, E2, E_3 (release constants) |
| Time Scale Factor | Aligns in vitro and in vivo time axes | T{in vivo} = a \cdot (T_{in vitro})^b |
| Correlation Coefficient | Measure of the model’s strength | R^2 \geq 0.95 |
| Prediction Error (%PE) | Validation of the model’s predictive power | \leq 10\%$ (average), \leq 15\%(individual) |
Representative studies report %PE values near 5.46%, demonstrating strong predictability. Accurate dissolution data improves model performance. Statistical validation ensures reliability for regulatory submissions.
Biopredictive Dissolution Methodology: USP Apparatus 4
Developing a biopredictive dissolution method is essential for IVIVC for Leuprolide Depot. USP Apparatus 4 (Flow-Through Cell) is widely used for long-acting microspheres. The system maintains sink conditions and reduces hydrodynamic variability. It enables detection of subtle manufacturing differences and improves reproducibility.
Advantages of the Flow-Through Cell (USP 4)
USP Apparatus 4 is suitable for microsphere formulations. Continuous flow prevents media saturation. Glass beads maintain uniform exposure. Media changes simulate physiological conditions. Hydrodynamic control enhances reproducibility and IVIVC predictability.
Accelerated vs. Real-Time Dissolution Methods
Real-time release testing for long-acting depots requires extended study durations. Accelerated methods reduce timelines using temperature or pH modifications. Mechanistic equivalence must be preserved. Excessive acceleration may change release mechanisms. Validation against real-time data is required.
| Acceleration Factor | Mechanism of Action | Consideration |
| Elevated Temperature | Increases polymer chain mobility and water uptake | Must not exceed the glass transition temperature (Tg) |
| Altered pH | Catalyzes ester bond hydrolysis | Acidic pH can cause peptide degradation/acylation |
| Organic Solvents | Increases polymer swelling and drug solubility | May lead to unrealistic burst release |
| Surfactants | Improves wetting and prevents aggregation | Essential for hydrophobic PLGA microspheres |
Successful accelerated methods show near 1:1 correlation with real-time release. These approaches improve development efficiency and support IVIVC model generation.
Discover the benefits of partnering with a North American CDMO: Pharmaceutical CDMO in US and Canada
PBPK Modeling and Virtual Bioequivalence (VBE)
Physiologically Based Pharmacokinetic (PBPK) modeling provides a mechanistic extension of IVIVC for Leuprolide Depot. These models integrate formulation attributes with physiological parameters. Virtual bioequivalence studies simulate population variability. This reduces dependence on clinical trials and supports regulatory decisions.
Mechanistic PBPK and Virtual Bioequivalence Analysis
PBPK models incorporate polymer ratio, molecular weight, drug loading, and porosity. Model verification involves comparison with variant formulations. Monte Carlo simulations generate virtual populations. Statistical evaluation confirms bioequivalence. These models help assess formulation risk and guide development.
Credibility Matrix and Platform Qualification
Regulators require credibility assessment for PBPK models. High-impact models must show robust validation. Multiple batches should be evaluated. Documentation must justify assumptions. Qualified platforms may support waiver of additional clinical studies.
Technical Challenges: Peptide Stability and Acylation
Chemical instability within PLGA matrix complicates IVIVC for Leuprolide Depot. Polymer degradation lowers internal pH and promotes peptide acylation. This reduces active drug release and complicates correlation between in vitro and in vivo results. Stabilization strategies are therefore necessary.
The Role of Micro-pH and the Serine Residue
Internal pH may drop to around 2.0 during erosion. The serine residue is a primary acylation site. Polymer-peptide adducts form under acidic conditions and alter release kinetics. Additives such as $Mg(OH)_2$ or $ZnCO_3$ help reduce acidity. Analytical methods distinguish acylated variants.
Polymer Variability and Autocatalysis
PLGA variability affects autocatalytic degradation. Differences in polydispersity index lead to uneven erosion. This disrupts point-to-point correlation. Strict raw material specifications are required. GPC analysis monitors polymer consistency. Controlled variability improves predictability.
Learn how a CRO can help manage complex Leuprolide projects: CRO for Leuprolide Depot Development
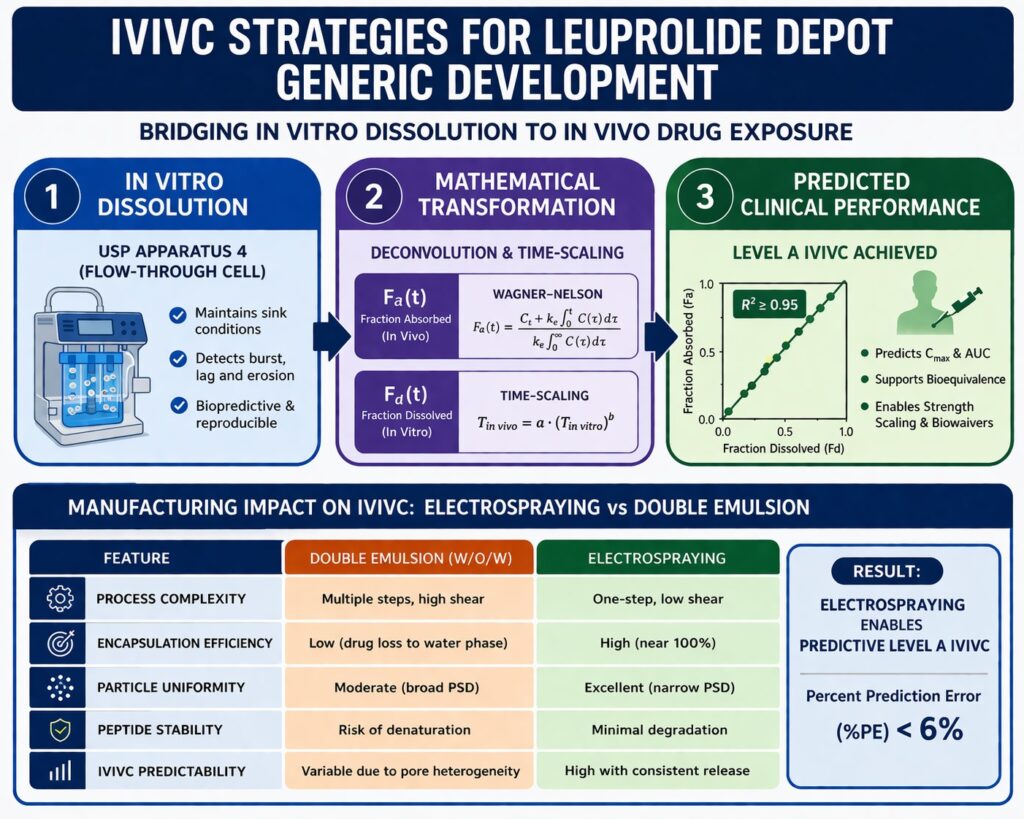
Case Study: Electrospraying vs. Double Emulsion in Depot Development
Manufacturing technology influences IVIVC for Leuprolide Depot. Double emulsion methods may create heterogeneous structures. Electrospraying produces controlled particle formation. Encapsulation efficiency improves and residual solvent decreases. Release kinetics become more predictable.
| Feature | Double Emulsion (W/O/W) | Electrospraying |
| Complexity | Multiple steps, high shear | One-step, low shear |
| Encapsulation Efficiency | Low (drug escapes to water phase) | High (near 100%) |
| Particle Uniformity | Moderate (broad PSD) | Excellent (narrow PSD) |
| Peptide Stability | Risk of denaturation at interfaces | Minimal (no heat or high shear) |
| IVIVC Predictability | Variable due to pore heterogeneity | High due to uniform internal structure |
Electrosprayed microspheres typically show reduced burst release and consistent secondary release. Predictive Level A IVIVC becomes achievable. Percent prediction error below 6% has been reported. Precision manufacturing improves regulatory success.
Read the full breakdown of sustained-release performance: Leuprolide Depot Case Study: Sustained Release
Frequently Asked Questions (FAQs)
The AUC_{7-t} parameter evaluates drug exposure during the sustained release phase after the initial burst period. It helps confirm that the formulation maintains consistent leuprolide levels throughout the dosing interval. Regulatory agencies rely on this metric to assess long-term delivery performance. If AUC_{7-t} differs significantly, it may indicate changes in polymer degradation and extended release behavior.
PLGA molecular weight controls polymer density and degradation speed, which directly affects drug release timing. Higher Mw typically slows hydrolysis and extends the lag phase before secondary release. In IVIVC for Leuprolide Depot, molecular weight distribution becomes a key model input. Differences from the reference product can shift release profiles and impact bioequivalence outcomes.
USP Apparatus 4 provides continuous flow conditions that maintain sink environment during dissolution testing. This helps prevent drug accumulation in the media, which could distort release results. The use of glass beads promotes uniform flow and minimizes particle aggregation. These features improve sensitivity and support biopredictive dissolution for depot microspheres.
A validated Level A IVIVC can support certain formulation or process changes without requiring new clinical studies. When the correlation is reliable, dissolution data may serve as a surrogate for in vivo performance. This approach is useful for scale-up, site transfer, or process optimization. Regulatory acceptance depends on model predictability and validation.
PLGA degradation produces acidic by-products that lower the internal pH inside microspheres. This acidic environment may cause peptide acylation, particularly at reactive residues. Modified drug forms can alter release behavior and reduce active drug availability. As a result, differences between in vitro release and in vivo absorption may occur.
PBPK modeling combines physiological parameters with formulation characteristics to simulate drug absorption. In virtual bioequivalence studies, large virtual populations are generated to compare test and reference products. The model evaluates whether exposure metrics fall within accepted bioequivalence limits. This approach helps identify formulation sensitivity before clinical trials.
Leuprolide depot products have long durations of action, often lasting several months. A crossover design would require very long washout periods, making studies impractical. Parallel studies allow comparison between separate groups receiving test and reference products. This design reduces study duration but requires careful control of subject variability.
The initial burst occurs due to drug present on the surface or within accessible pores of microspheres. When exposed to biological fluids, this portion dissolves quickly. Manufacturing conditions such as polymer concentration and emulsification speed influence surface structure. These factors determine the magnitude of early release.
Reference:
- Swayzer, D. V., & Gerriets, V. (2023). Leuprolide. In StatPearls. StatPearls Publishing. https://pmc.ncbi.nlm.nih.gov/articles/PMC7022573/
- Shen, J., Lee, K., Choi, S., Qu, W., Wang, Y., & Burgess, D. J. (2016). A reproducible accelerated in vitro release testing method for PLGA microspheres. International Journal of Pharmaceutics, 498(1–2), 274–282. https://doi.org/10.1016/j.ijpharm.2015.12.031
- Cheng, Y.-H., Thomas, S., Tsang, Y. C., Almeida, S., Ashraf, M., Fotaki, N., Heimbach, T., Patel, N., Shah, H., Jiang, X., Kim, M.-J., Moody, R., Rostami-Hodjegan, A., Singh, R., Zhao, L., Babiskin, A., & Wu, F. (2025). Advances in physiologically based pharmacokinetic (PBPK) modeling and its regulatory utility to support oral drug product development and harmonization. Pharmaceutical Research, 42(5), 819–833. https://doi.org/10.1007/s11095-025-03849-9
- U.S. Food and Drug Administration. (2021). Product-specific guidance for leuprolide acetate injection (depot). Center for Drug Evaluation and Research. https://www.fda.gov/media/166586/download
- Manolis, E., García-Arieta, A., Lindahl, A., Kotzagiorgis, E., Limberg, J., Holte, Ø., Paixao, P., Versantvoort, C., Musuamba Tshinanu, F., Blake, K., & Van Den Heuvel, M. (2023). Using mechanistic models to support development of complex generic drug products: European Medicines Agency perspective. CPT: Pharmacometrics & Systems Pharmacology, 12(5), 556–559. https://doi.org/10.1002/psp4.12906
- Su, Y., Zhang, B., Sun, R., Liu, W., Zhu, Q., Zhang, X., Wang, R., & Chen, C. (2021). PLGA-based biodegradable microspheres in drug delivery: Recent advances in research and application. Drug Delivery, 28(1), 1397–1418. https://doi.org/10.1080/10717544.2021.1938756



