Introduction
Leuprolide Depot IVRT Methods are essential for establishing equivalence between reference and generic PLGA-based microsphere depots by accurately measuring multiphasic peptide release under physiologically relevant conditions. These long-acting systems require testing approaches capable of capturing slow polymer degradation and prolonged drug release. The complexity of PLGA depots makes conventional dissolution testing unsuitable for meaningful comparison. A properly designed IVRT method provides reliable and reproducible release data across extended durations.
Unlike conventional dissolution testing, leuprolide depot IVRT must address polymer degradation-driven release, peptide adsorption, burst effects, and long-duration release extending from weeks to months. These characteristics demand long-term sampling strategies and stable analytical detection methods. The IVRT design must also minimize experimental artifacts that may distort release kinetics. Careful control of test conditions improves accuracy.
For generic developers, IVRT is not only a characterization tool but also a quality control and formulation development method. It supports process optimization, batch comparison, and screening during early development. Additionally, IVRT helps identify formulation parameters that influence in vivo performance. This makes IVRT critical throughout product development.
Learn more about our specialized support for ANDA submissions: Generic Drug Development Process for ANDA
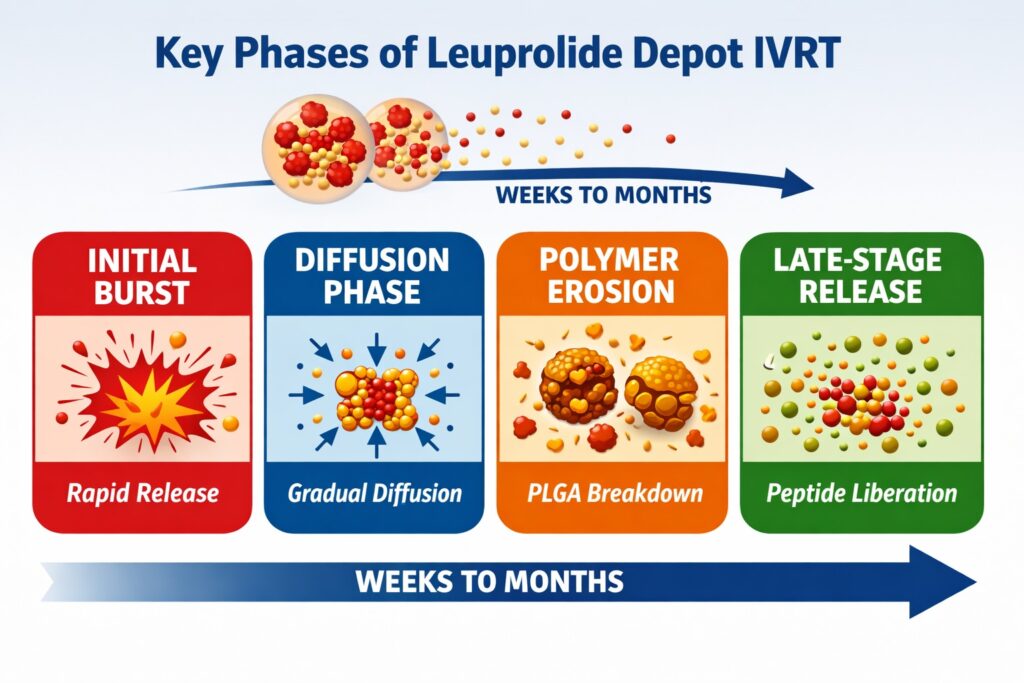
The complexity of PLGA microspheres requires carefully optimized test systems capable of capturing:
- Initial burst release
- Diffusion-controlled phase
- Polymer erosion-controlled release
- Late-stage peptide liberation
These mechanistic stages must be preserved in Leuprolide Depot IVRT Methods to ensure meaningful comparison with reference listed drug (RLD) products. Maintaining the complete release profile allows developers to evaluate equivalence across the entire dosing interval. Missing any phase may lead to misleading conclusions about formulation similarity.
Share via:
Critical Design Considerations for Leuprolide Depot IVRT Methods
The most important design factors include maintaining sink conditions, controlling polymer degradation, minimizing peptide adsorption, and capturing the full multiphasic release profile. These parameters determine whether the IVRT method reflects true release behavior. Improper control can lead to artificial plateauing or exaggerated burst release. Therefore, optimization is necessary.
Leuprolide depot microspheres exhibit complex release driven by PLGA hydrolysis. IVRT design must control environmental variables affecting polymer erosion and peptide solubility. Factors such as buffer composition, agitation, and temperature significantly influence degradation rate. Careful optimization ensures consistent release measurements.
Explore our comprehensive services for complex generic formulations: CRO for Leuprolide Depot Development
Key Method Design Parameters
| Parameter | Impact on IVRT | Consideration |
|---|---|---|
| Temperature | Controls polymer degradation rate | 37°C real-time; 45–55°C accelerated |
| Agitation | Influences boundary layer diffusion | Orbital shaking or USP apparatus |
| Surfactant | Maintains sink conditions | Tween 80 / SDS optimization |
| Buffer pH | Affects peptide stability | pH 7.4 PBS commonly used |
| Sampling approach | Influences accuracy | Continuous vs discrete |
| Volume | Prevents saturation | High volume recommended |
| Microsphere load | Avoids aggregation | Low solid content |
| Container type | Prevents adsorption | Low binding materials |
Studies demonstrate that peptide adsorption to vessel walls or filters can artificially lower release values. Low-binding containers and validated filtration recovery are therefore essential. Adsorption effects are particularly important for peptide drugs like leuprolide. Recovery experiments should be incorporated into method development.
Sample-and-Separate Leuprolide Depot IVRT Methods
Sample-and-separate methods provide direct release measurement and strong discriminatory capability but require careful handling to avoid microsphere loss. These methods are commonly used during early development because they are flexible. They also allow rapid comparison of multiple prototype formulations. Proper handling improves reproducibility.
This approach suspends microspheres in release media and periodically separates them by centrifugation or filtration. Sampling intervals can be customized based on release duration. The method enables direct measurement of released leuprolide without membrane barriers. This makes interpretation straightforward.
Discover how to navigate the complexities of Leuprolide formulation: How to Develop Generic Leuprolide Depot
Advantages
- Direct measurement of released leuprolide
- Good discrimination between formulations
- Flexible media modification
- Suitable for accelerated testing
These advantages make the sample-and-separate approach ideal for screening studies. It allows quick adjustment of surfactant concentration and buffer composition. Developers often use this method during formulation optimization. It is widely used in early development.
Limitations
- Handling variability
- Microsphere aggregation risk
- Sampling disturbance
- Labor-intensive long-term studies
These limitations become more significant for extended release studies lasting several weeks. Repeated sampling may disturb microsphere distribution. Careful handling procedures help minimize variability. Standardized centrifugation conditions are recommended.
For Leuprolide Depot IVRT Methods, centrifugation-based separation is often preferred over filtration to minimize adsorption and peptide loss. Filtration membranes may retain peptide or microspheres. Validating recovery ensures reliable quantification. This improves accuracy.
Dialysis-Based Leuprolide Depot IVRT Methods
Dialysis IVRT isolates microspheres from sampling media and reduces particle loss but introduces membrane diffusion limitations. These systems are useful for long-duration release studies. They also improve reproducibility by minimizing handling. Proper validation is required.
Dialysis methods encapsulate microspheres inside dialysis tubing or devices, allowing released leuprolide to diffuse into surrounding media. Sampling is performed from the external compartment. This reduces disturbance of microspheres during testing. It improves consistency.
Access our expert analysis on overcoming sustained-release hurdles: Leuprolide Depot Case Study: Sustained Release
When Dialysis IVRT Is Preferred
- Long duration release studies
- Reduced microsphere handling
- Continuous release monitoring
- Improved reproducibility
These benefits make dialysis suitable for confirmatory testing. It is also useful when sample-and-separate methods show variability. Proper validation ensures accurate interpretation. Method comparison is recommended.
Critical Dialysis Parameters
- Membrane molecular weight cutoff
- Surface area-to-volume ratio
- Diffusion lag validation
- Membrane binding studies
- Agitation uniformity
Improper membrane selection may create diffusion-controlled artifacts. This can mask actual PLGA erosion-driven release. Diffusion lag must therefore be evaluated experimentally. Compatibility studies are essential.
Continuous Flow Leuprolide Depot IVRT Methods
Continuous-flow IVRT provides improved sink conditions and is highly suitable for regulatory-quality release testing. These systems maintain constant media exchange. They are particularly useful for long-acting depot formulations. Automation improves reproducibility.
Flow-through systems continuously circulate media across microspheres, preventing saturation and enabling long-term testing. This reduces accumulation of released peptide. The approach improves reproducibility and reduces manual sampling. It is ideal for QC testing.
Benefits include:
- Constant sink conditions
- Reduced sampling error
- Automation capability
- High reproducibility
- Regulatory suitability
These advantages make continuous-flow systems attractive for validated testing. They allow precise control of flow rate and temperature. However, instrumentation complexity may limit early use. Setup optimization is required.
See how a specialized CDMO can streamline your development timeline: Specialized Peptide CDMO Services
Accelerated Leuprolide Depot IVRT Methods
Accelerated IVRT reduces test duration by increasing polymer degradation rate without altering release mechanism. This is critical for long-acting depot products. Accelerated testing supports rapid development decisions. It also enables faster screening.
Accelerated methods are essential for generics development where real-time release may exceed 30–90 days. These methods allow quicker formulation comparison. They also enable screening of multiple batches. Development timelines are shortened.
Common Acceleration Strategies
- Elevated temperature (45–60°C)
- Hydroalcoholic media
- Basic pH buffers
- Surfactant-enhanced release
- Enzyme-assisted degradation
These approaches increase polymer hydrolysis and peptide diffusion. The selected strategy should maintain mechanistic similarity. Comparative studies help confirm equivalence. Real-time comparison is recommended.
Understand the differences between CDMO and CRO partnerships for peptides: Peptide CDMO vs CRO
Discriminatory Power in Leuprolide Depot IVRT Methods
A valid IVRT method must detect changes in polymer molecular weight, particle size, and drug loading. This capability ensures sensitivity to formulation differences. Regulatory acceptance depends on demonstrated discrimination. Statistical comparison is often required.
Discriminatory testing should differentiate:
- PLGA molecular weight differences
- Lactide:glycolide ratio changes
- Particle size distribution
- Encapsulation efficiency
- Residual solvent content
These factors directly influence leuprolide release kinetics. Small formulation changes can significantly alter release behavior. The IVRT method must reliably detect these differences. This confirms method suitability.
Analytical Considerations for Leuprolide IVRT
LC-based peptide quantification with stability-indicating capability is required for accurate release measurement. Analytical sensitivity must be sufficient for low-level detection. Stability assessment is also essential. Proper validation improves reliability.
Common analytical approaches:
- RP-HPLC with UV detection
- LC-MS peptide quantification
- Stability-indicating degradation monitoring
- Adsorption recovery studies
These methods provide reliable quantification of released peptide. LC-MS offers improved specificity. UV detection remains widely used due to simplicity. Selection depends on sensitivity requirements.
Method validation should include:
- Accuracy
- Precision
- Specificity
- Linearity
- Filter compatibility
Proper validation ensures reliable IVRT results. Analytical robustness directly impacts release interpretation. Recovery testing confirms sample integrity. This improves confidence in data.
Deep dive into the specific hurdles of Leuprolide analysis: Leuprolide Depot Analytical Challenges
Establishing IVIVC Using Leuprolide Depot IVRT Methods
IVRT can support IVIVC when release mechanisms match in vivo polymer degradation. Establishing correlation strengthens generic justification. It may reduce clinical study requirements. Regulatory confidence increases.
Mechanistic correlation approaches:
- Level A correlation (cumulative release)
- Deconvolution-based modeling
- Weibull fitting
- Diffusion-erosion hybrid models
These approaches compare in vitro and in vivo release patterns. Model selection depends on release behavior. Successful IVIVC enhances predictability. This supports regulatory submissions.
Reliable IVIVC strengthens generic justification and reduces clinical burden. It also supports formulation optimization. Consistent correlation improves predictability. Development decisions become easier.
Recommended Workflow for Developing Leuprolide Depot IVRT Methods
- Screen sample-and-separate method
- Evaluate dialysis-based approach
- Optimize sink conditions
- Introduce accelerated conditions
- Validate discriminatory capability
- Confirm reproducibility
- Establish reference comparison
- Develop QC method
This workflow ensures systematic method development. Each step refines experimental conditions. Following a structured approach improves reliability. It also supports regulatory expectations.
Partner with a leader in North American pharmaceutical development: Pharmaceutical CDMO US and Canada
Common Pitfalls in Leuprolide Depot IVRT Development
- Peptide adsorption losses
- Insufficient sink conditions
- Membrane diffusion limitation
- Microsphere aggregation
- Inadequate agitation
- Accelerated method altering mechanism
- Sampling volume errors
Avoiding these ensures robust Leuprolide Depot IVRT Methods. Early identification of pitfalls improves efficiency. Proper controls help prevent variability. Consistent handling is important.
Regulatory Expectations for Leuprolide Depot Generics
Regulators expect IVRT methods to be discriminatory, reproducible, and mechanistically relevant. The method must support equivalence claims. Documentation should demonstrate scientific justification. Validation is required.
Key expectations:
- Method validation
- Reference vs generic comparison
- Batch variability detection
- Accelerated and real-time correlation
- Stability-indicating analytics
IVRT becomes a critical quality attribute for long-acting injectable generics. Regulatory agencies rely on IVRT data for evaluation. Robust methods reduce approval risk. Strong justification improves acceptance.
Conclusion
Leuprolide Depot IVRT Methods must be carefully designed to capture polymer degradation-driven release, maintain sink conditions, and demonstrate discriminatory power between formulations. Accurate characterization of multiphasic release is essential for generic equivalence. Method robustness ensures reliable long-term testing. Properly optimized IVRT improves confidence.
Sample-and-separate, dialysis, and continuous-flow approaches each provide unique advantages, while accelerated testing is essential for efficient generics development. Selecting the appropriate method depends on development stage and regulatory goals. Combining approaches may improve confidence. Method selection should be justified.
A robust IVRT method strengthens formulation optimization, regulatory submissions, and equivalence demonstration for leuprolide depot generics. It also supports lifecycle management and batch consistency evaluation. Properly validated IVRT becomes a cornerstone of depot product development.
For method development support and technical consultation, contact us
FAQs – Leuprolide Depot IVRT Methods
Sample-and-separate methods are commonly used during early development because they offer flexibility and strong discrimination. Dialysis or continuous-flow systems are preferred for validated testing due to improved reproducibility. The final choice depends on release duration and regulatory strategy. Often, developers transition from screening to controlled systems.
Leuprolide solubility limits can cause artificial release plateaus if sink conditions are not maintained. Proper sink conditions ensure release is governed by polymer degradation instead of saturation. Surfactants and large media volumes are typically used. This improves accuracy of release profiles.
Accelerated IVRT shortens timelines but must preserve the same release mechanism. Elevated temperature or modified media should not change PLGA degradation behavior. Real-time confirmation is still recommended. Correlation between accelerated and real-time data is important.
Discriminatory power is shown by intentionally varying formulation parameters such as polymer molecular weight or particle size. The IVRT method should detect clear differences in release profiles. Statistical comparison is usually performed. This confirms method sensitivity.
Agitation reduces boundary layer thickness and improves mass transfer. Inconsistent agitation can lead to variability in release profiles. Controlled shaking or apparatus settings are recommended. Uniform mixing improves reproducibility.
Typical phases include initial burst release, diffusion-controlled release, lag phase, polymer erosion phase, and late-stage release. These stages reflect PLGA degradation behavior. IVRT must capture each phase. Missing phases can misrepresent performance.
Reference:
- U.S. Food and Drug Administration, Center for Drug Evaluation and Research. (2011). Clinical review: NDA 20-517/S025, S030, S032 — Leuprolide acetate 45 mg. U.S. Food and Drug Administration. https://www.accessdata.fda.gov/drugsatfda_docs/nda/2011/020517orig1s025s030s032Review.pdf
- U.S. Food and Drug Administration, Center for Drug Evaluation and Research. (2001). Administrative correspondence: NDA 20-708/S011 — Lupron Depot (leuprolide acetate for depot suspension). U.S. Food and Drug Administration. https://www.accessdata.fda.gov/drugsatfda_docs/nda/2001/20-708S011_Lupron_admincorres_P1.pdf



