Introduction: The Formulation Science Behind Long Acting Injectable Drug Delivery Technologies
Long Acting Injectable Drug Delivery Technologies are changing how medicines are given by moving from daily oral dosing to long-lasting injectable treatments. These systems are designed to maintain steady drug levels in the body for days, weeks, or even months. Scientists use material science principles such as polymer degradation, lipid membrane permeability, and phase transition behavior to control how the drug is released. By carefully adjusting these parameters, formulators can design products that match specific therapeutic needs and dosing schedules. Each platform has its own release mechanism, critical quality attributes, and manufacturing considerations that must be understood for reliable performance.
In addition, formulation scientists must evaluate drug properties, injection site biology, and dosing limitations. Factors such as depot size, tissue interaction, and degradation products can affect in vivo behavior. Regulatory agencies also expect a strong mechanistic understanding to ensure consistency during scale-up and commercial production. Modern Long Acting Injectable Drug Delivery Technologies therefore rely on both formulation science and analytical validation. This article explains the major platform technologies and how they function in practical development settings.
Learn more about polymer-based formulation: Long Acting Injectable Formulation Development Services
Share via:
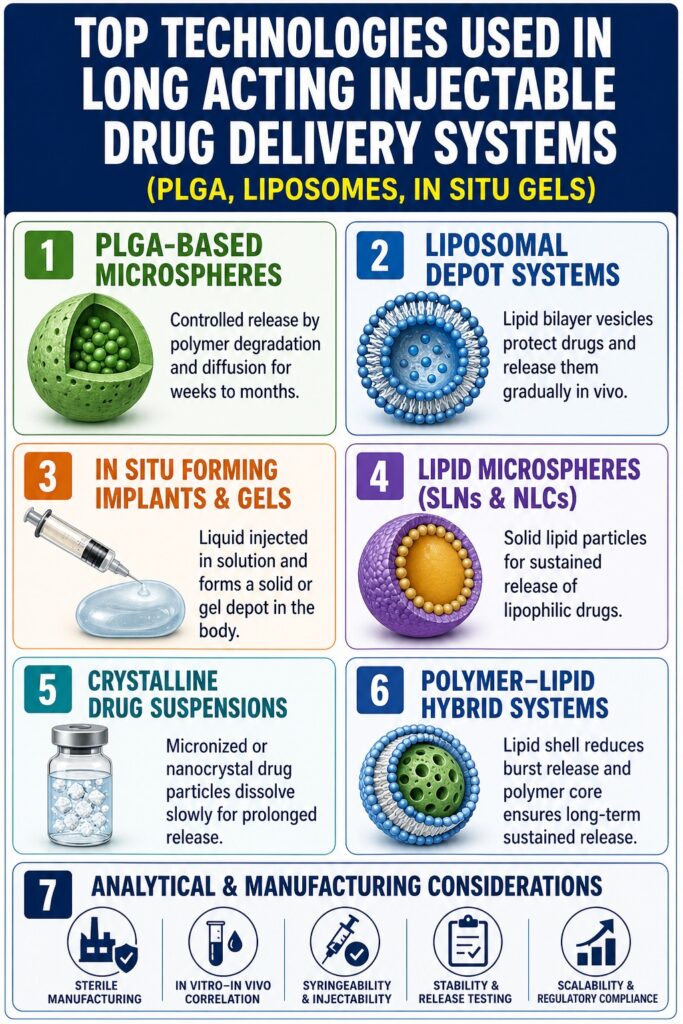
1. PLGA-Based Microspheres and Nanoparticles in Long Acting Injectable Drug Delivery Technologies
PLGA (poly lactic-co-glycolic acid) systems provide controlled release by combining drug diffusion with gradual polymer erosion. This dual mechanism allows formulators to adjust both diffusion rate and degradation speed. Drug molecules are encapsulated inside microspheres or nanoparticles and released slowly as the polymer absorbs water and breaks down. These systems are widely used for peptides, small molecules, and CNS therapies that require sustained exposure. PLGA is biodegradable and biocompatible, which supports regulatory acceptance and long-term safety.
Another advantage of PLGA platforms is their flexibility in tuning release duration. By modifying polymer composition and molecular weight, developers can design formulations that last from weeks to several months. Particle size, drug loading, and encapsulation efficiency also influence release behavior. Manufacturing techniques such as solvent evaporation, spray drying, and double emulsion methods help control these attributes. Because of this versatility, PLGA remains one of the most established Long Acting Injectable Drug Delivery Technologies.
1.1 How the Release Kinetics Are Actually Controlled
PLGA degrades through hydrolysis of ester linkages, producing lactic and glycolic acid. The lactide-to-glycolide (L:G) ratio is a primary factor controlling degradation rate.
| L:G Ratio | Degradation Rate | Approximate Release Duration |
|---|---|---|
| 50:50 | Fastest | 1–2 months |
| 65:35 | Moderate | 3–4 months |
| 75:25 | Slow | 4–6 months |
| 85:15 | Slowest | 6+ months |
Molecular weight also affects performance. Lower molecular weight PLGA degrades faster, while higher molecular weight grades extend release. End-cap chemistry influences internal hydrolysis, with acid-terminated polymers degrading faster than ester-terminated ones. Other factors such as polymer crystallinity, solvent residue, and drug loading also modify release behavior. Together, these variables create a flexible design space for predictable depot performance.
1.2 The Triphasic Release Problem
PLGA microspheres often show a triphasic release profile. The first phase is an initial burst caused by drug near the particle surface. The second phase is a lag period where little drug is released while the polymer remains intact. The third phase occurs when polymer erosion accelerates and bulk drug release begins. This pattern can create gaps in therapeutic exposure if not controlled.
To reduce burst release and minimize the lag phase, formulators adjust particle size distribution and drug loading uniformity. Encapsulation efficiency and internal morphology are also important. Emulsification method, homogenization speed, and solvent evaporation rate all influence pore formation. Careful optimization helps achieve smooth and continuous drug release. These controls are essential for consistent performance in Long Acting Injectable Drug Delivery Technologies.
1.3 Analytical Demands in PLGA Development
PLGA formulations require multiple analytical techniques for full characterization:
- GPC/SEC for polymer molecular weight and dispersity
- DSC and TGA for crystallinity and thermal behavior
- In vitro release testing under biorelevant conditions
- SEM/TEM for morphology and pore structure
- Residual solvent analysis using headspace GC
These tests help establish structure-function relationships in the formulation. Monitoring polymer degradation over time supports long-term stability predictions. Particle size analysis is also used for batch consistency. Strong analytical control is necessary for regulatory submission and commercial manufacturing.
Deep dive into complex characterization: Characterization of Long Acting Injectables
2. Liposomal Long Acting Injectable Drug Delivery Technologies: Membrane Architecture as a Release Barrier
Liposomes act as sustained-release depots by trapping drugs inside lipid bilayer structures. Hydrophilic drugs remain in the aqueous core, while lipophilic drugs are incorporated into the membrane. Drug release occurs gradually as the membrane structure changes in vivo. This architecture protects sensitive drugs and reduces systemic toxicity. Liposomes also provide flexibility across many drug classes.
For injectable depot use, size, lamellarity, and lipid composition determine release rate. Larger vesicles typically release drug more slowly than small vesicles. Surface charge affects tissue interaction and depot retention. Lipid composition influences membrane rigidity and permeability. These structural elements are carefully balanced to achieve controlled delivery.
2.1 Structural Engineering for Parenteral Depot Performance
Different liposome types behave differently in injectable applications. Multilamellar vesicles and large unilamellar vesicles are often preferred for depot delivery. These structures provide higher drug loading and slower diffusion. Lipid composition is a key design variable.
- DPPC and DSPC provide rigid membranes and slower release
- Cholesterol reduces membrane fluidity and leakage
- PEGylated lipids extend circulation time but reduce loading
Chain length and saturation also influence packing density. Higher transition temperature lipids generally slow drug release. Surface charge affects aggregation and retention at the injection site. These design considerations are central to liposomal Long Acting Injectable Drug Delivery Technologies.
2.2 In Situ Depot Formation via Liposome Aggregation
Charged liposomes can aggregate at the injection site and form a depot. Cationic lipids interact with negatively charged tissue components, creating a semi-solid matrix. This structure holds the drug and releases it gradually. Aggregation behavior can be tuned by adjusting lipid composition and ionic strength. Such systems are explored for local anesthetics and anti-inflammatory therapies. Controlled aggregation improves retention while limiting systemic exposure.
2.3 Key Stability Challenges
Liposomal formulations face several stability risks:
- Phospholipid hydrolysis producing lysolipids
- Lipid oxidation controlled using antioxidants
- Vesicle aggregation during storage
- Drug leakage during freezing conditions
Lyophilization is often used to improve shelf life. Cryoprotectants such as sucrose or trehalose protect vesicle structure. Storage temperature and container systems also affect stability. Stability testing must monitor both particle size and drug retention.
3. In Situ Forming Implants and Thermosensitive Gels in Long Acting Injectable Drug Delivery Technologies
In situ forming systems are injected as liquids and transform into solid or gel depots inside the body. This transformation may occur due to solvent exchange, temperature change, or pH shift. The formed depot releases drug slowly over time. These systems eliminate the need for surgical implantation. They also allow high drug loading and flexible release duration.
Such technologies are increasingly used for sustained hormone therapy, pain management, and oncology applications. Injection viscosity and phase transition behavior must be carefully optimized. The resulting implant structure determines release kinetics. These systems are considered a growing segment of Long Acting Injectable Drug Delivery Technologies.
3.1 The Atrigel® Mechanism (Solvent Exchange ISFIs)
Atrigel-type systems use PLGA dissolved in a water-miscible solvent such as NMP. After injection, solvent diffuses into surrounding tissue while water diffuses inward. This causes polymer precipitation and implant formation. The drug becomes trapped inside the porous matrix. Release then occurs by diffusion followed by polymer erosion.
Critical formulation parameters for Atrigel-type ISFIs:
| Parameter | Effect on Performance |
|---|---|
| PLGA concentration (w/v) | Controls implant porosity and initial burst |
| Polymer Mw and L:G ratio | Governs erosion rate and release duration |
| NMP volume | Affects injection viscosity and precipitation rate |
| Drug particle size | Influences initial burst magnitude |
| Drug-polymer miscibility | Determines drug distribution uniformity |
Balancing solvent content and polymer concentration is important for injectability. Implant morphology affects long-term release behavior. Both formulation and injection conditions influence final performance.
3.2 Thermosensitive Gel Systems: Poloxamers, PLGA-PEG-PLGA, and Chitosan Derivatives
Thermosensitive polymers remain liquid at room temperature and gel at body temperature. This creates an injectable depot without organic solvents. Common polymers include:
- Poloxamer 407 (Pluronic F127)
- PLGA-PEG-PLGA triblock copolymers
- Chitosan-based thermoresponsive gels
Gelation occurs due to micelle aggregation and network formation. Drug molecules become trapped in the gel structure. Release depends on gel erosion and diffusion. These systems are useful for localized drug delivery and minimally invasive treatment.
Compare development models: CRO vs In-House ANDA Development
4. Lipid Microspheres, Solid Lipid Nanoparticles (SLNs), and Nanostructured Lipid Carriers (NLCs)
Lipid-based particulate systems provide biocompatible matrices for sustained release. Drugs are incorporated into solid lipid cores that slowly release payload by diffusion and matrix erosion. These platforms are suitable for lipophilic compounds. They also improve stability and reduce injection site irritation. Manufacturing usually involves high-pressure homogenization or melt emulsification.
4.1 SLNs vs. NLCs for LAI Applications
| Feature | Solid Lipid Nanoparticles (SLN) | Nanostructured Lipid Carriers (NLC) |
|---|---|---|
| Lipid matrix | Solid lipid only | Blend of solid + liquid lipids |
| Drug loading capacity | Limited | Higher |
| Drug release mechanism | Diffusion through solid matrix | Diffusion + matrix melting |
| Physical stability | Risk of polymorphic transitions | More stable long-term |
| Burst release tendency | Higher | Lower |
SLNs may undergo polymorphic transitions that push drug out of the matrix. This can cause burst release during storage. NLCs reduce this issue by mixing solid and liquid lipids. The resulting structure improves stability and drug loading. Because of these benefits, NLCs are increasingly used for injectable depot formulations.
5. Crystalline Drug Suspensions: Micronization and Nanocrystal Engineering
Crystalline suspensions rely on slow dissolution of poorly soluble drug particles. The drug is micronized or nanocrystallized and suspended in an injectable vehicle. After injection, particles dissolve gradually at the site. This provides sustained release over time. This approach is widely used in antipsychotic and hormonal therapies.
Particle size directly controls dissolution rate. Smaller particles dissolve faster, while larger particles extend duration. Stabilizers prevent aggregation and maintain uniform suspension. Viscosity modifiers improve syringeability and dose consistency. Control of crystal form and size distribution is essential for predictable performance.
Critical variables:
- Particle size distribution (D10, D50, D90)
- Crystal polymorphic form
- Surfactant or stabilizer system
- Viscosity modifier such as carboxymethylcellulose
These factors must be tightly controlled during manufacturing. Stability testing monitors particle growth and settling. Suspension rheology also affects injectability.
Read a relevant case study: Leuprolide Depot Case Study: Sustained Release
6. Polymer-Lipid Hybrid Systems: The Next-Generation Long Acting Injectable Drug Delivery Technologies
Hybrid systems combine polymer and lipid components in a single structure. A lipid shell surrounds a PLGA core to suppress burst release. The polymer core then controls sustained erosion-based release. This design improves stability for peptides and proteins. It also reduces water penetration into the polymer matrix.
These systems allow independent tuning of diffusion and degradation mechanisms. Encapsulation efficiency is often higher for sensitive biologics. Manufacturing approaches include double emulsion followed by lipid coating. Microfluidic assembly is also used for precise particle control. Hybrid systems represent an emerging direction in Long Acting Injectable Drug Delivery Technologies.
7. Analytical and Manufacturing Considerations Across All LAI Platforms
All long-acting injectable platforms require sterile manufacturing. This may involve aseptic processing or terminal sterilization. Radiation sterilization can affect polymer molecular weight and lipid stability. Therefore, compatibility must be evaluated early in development. Process control ensures consistent batch quality.
In vitro–in vivo correlation is critical for regulatory approval. Agencies often expect Level A IVIVC supported by mechanistic modeling. Dose dumping studies assess risk of rapid release under stress conditions. Syringeability and injectability must also be validated using clinical needle sizes. Rheology and particle settling behavior influence administration performance.
ResolveMass Laboratories Inc. provides analytical support across PLGA, liposomal, in situ gel, lipid nanoparticle, and crystalline suspension platforms. Services include physicochemical characterization, release testing, and regulatory support. Advanced instrumentation enables mechanistic understanding of release behavior. These capabilities help accelerate development timelines while maintaining compliance.
Understand the submission process: Requirements for ANDA Submission for Generic Drugs
Conclusion: Choosing the Right Long Acting Injectable Drug Delivery Technology
There is no single best option among Long Acting Injectable Drug Delivery Technologies. The optimal choice depends on drug properties, therapeutic goals, and manufacturing capabilities. PLGA microspheres provide well-established regulatory history for long duration release. Liposomes are useful for sensitive molecules requiring protection. In situ gels offer simple administration and implant formation. Crystalline suspensions provide a straightforward option for poorly soluble drugs. Lipid nanoparticles and hybrid systems continue to expand formulation possibilities.
All these platforms require strong analytical characterization and process control. Scalability, sterilization compatibility, and stability must be considered early. Regulatory expectations emphasize predictable release and reproducible manufacturing. Successful development therefore integrates formulation science, process engineering, and analytical validation. These combined elements define modern excellence in Long Acting Injectable Drug Delivery Technologies.
Partner with experts for your project: CRO for Complex Injectables
Frequently Asked Questions (FAQs)
PLGA (poly lactic-co-glycolic acid) is widely used in long-acting injectable formulations because it is biodegradable, biocompatible, and well accepted by regulatory agencies. Its degradation rate can be adjusted by changing the lactide-to-glycolide ratio and molecular weight, allowing flexible release durations. The polymer breaks down into lactic and glycolic acid, which are naturally processed by the body. This strong safety and performance record makes PLGA a preferred starting material for many formulations.
Burst release usually happens when drug molecules remain near the surface of microspheres. Formulators reduce this effect by improving drug distribution inside the polymer matrix and optimizing emulsification conditions. Techniques such as reducing drug particle size, adjusting viscosity, and applying polymer coatings can help control initial release. These strategies aim to create uniform particles and provide smoother, sustained drug delivery.
In situ forming implants are injected as liquids that solidify inside the body, while PLGA microspheres are pre-made solid particles suspended in a liquid. ISFIs are easier to manufacture but may cause local irritation due to solvent exchange at the injection site. Pre-formed microspheres generally offer more predictable release because their structure is fixed before injection. Clinical selection depends on release consistency, patient tolerance, and manufacturing considerations.
Liposomal systems can encapsulate proteins and peptides within their aqueous core, making them useful for sensitive biologics. These systems help protect molecules from degradation and allow gradual release. However, maintaining protein stability during formulation and storage remains challenging. Additional steps such as lyophilization and careful processing are often required to preserve structure and activity.
IVRT for long-acting injectables is designed to measure drug release over extended periods. Common methods include dialysis membrane testing, sample-and-separate techniques, and flow-through cell systems. These tests use media that simulate physiological conditions to maintain sink conditions. The goal is to generate release data that can later be correlated with in vivo pharmacokinetic behavior.
Thermosensitive gels can degrade over time due to hydrolysis of polymer chains. This degradation may shift the gelation temperature and weaken the gel structure. Changes in polymer molecular weight can also affect drug release behavior. Additional concerns include drug precipitation during storage and phase separation after freeze-thaw cycles.
Particle size directly controls how quickly a drug dissolves at the injection site. Smaller particles dissolve faster and shorten the duration of action, while larger particles extend release. Maintaining a consistent particle size distribution is therefore critical for predictable performance. Manufacturing processes such as wet milling are carefully controlled to prevent variation.
Nanostructured lipid carriers are composed of mixed solid and liquid lipids that create a disordered internal structure. This design allows higher loading of lipophilic drugs compared to traditional liposomes. Liposomes contain a lipid bilayer with an aqueous core, making them better for hydrophilic or amphiphilic drugs. NLCs also offer improved physical stability and reduced risk of drug expulsion during storage.
Reference:
- U.S. Food and Drug Administration. (2014). Bioavailability Studies Submitted in NDAs or INDs — General Considerations: Guidance for Industry. CDER. https://www.fda.gov/media/88254/download
- European Medicines Agency. (2014). Guideline on the pharmacokinetic and clinical evaluation of modified release dosage forms (EMA/CHMP/EWP/280/96 Rev1). https://www.ema.europa.eu/en/documents/scientific-guideline/guideline-pharmacokinetic-clinical-evaluation-modified-release-dosage-forms_en.pdf
- ICH. (2009). Pharmaceutical Development Q8(R2). International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use. https://www.ich.org/page/quality-guidelines



