Introduction:
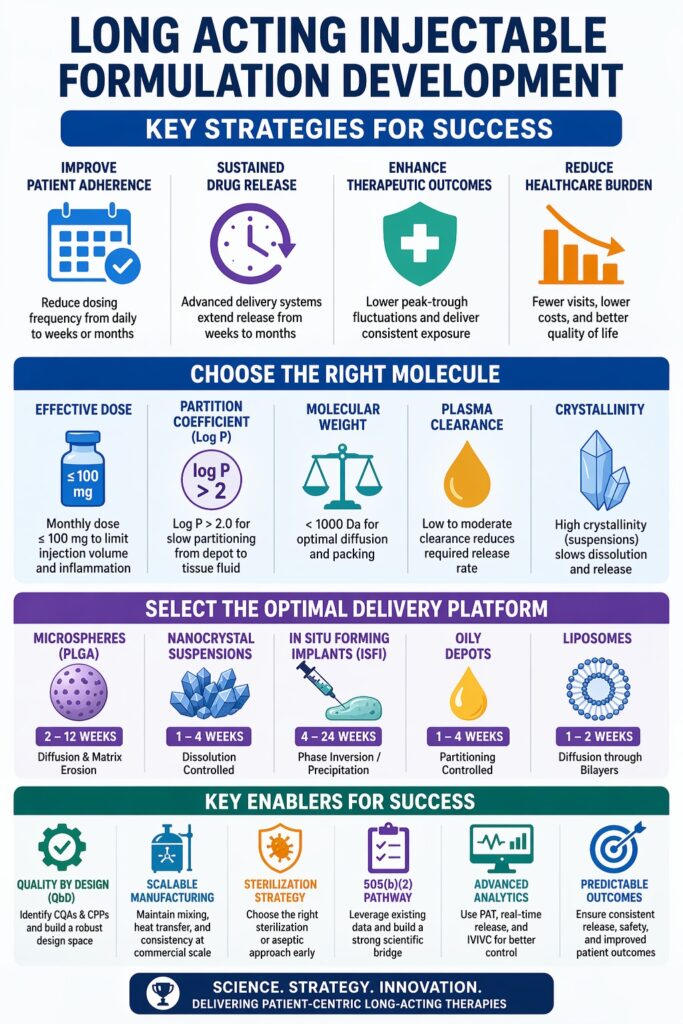
Long Acting Injectable Formulation Development is becoming a preferred strategy for pharmaceutical companies aiming to improve patient adherence and extend product lifecycle. The success of Long Acting Injectable Formulation Development depends on aligning molecular potency, polymer degradation behavior, and a strong scientific bridge for regulatory approval through the 505(b)(2) pathway. Using microencapsulation and nanocrystal technologies, companies can extend drug release from weeks to months. This reduces dosing frequency, improves treatment continuity, and lowers healthcare burden. Long-acting injectables are widely explored in antipsychotics, oncology, hormones, and anti-infectives. A well-designed formulation can improve bioavailability, reduce peak-trough fluctuations, and deliver consistent therapeutic outcomes.
Learn more about the technical process: Comprehensive Guide to the Generic Drug Development Process for ANDA
Share via:
Advanced Molecular Selection and Physicochemical Screening Criteria in Long Acting Injectable Formulation Development
Selecting the right drug candidate for Long Acting Injectable Formulation Development requires careful evaluation of potency, clearance rate, and lipophilicity. Because intramuscular or subcutaneous injections typically allow only 1 mL to 3 mL volume, the dose must be concentrated within a controlled-release system. Molecules with long half-lives and moderate clearance are generally better suited for depot formulations. Chemical stability at physiological pH and temperature must also be confirmed during early screening. Compatibility with polymers and excipients is essential to maintain stability during storage and administration.
The Critical Role of LogP and Effective Dosing in Long Acting Injectable Formulation Development
Successful long-acting injectable candidates usually show a balance between water solubility and permeability. This is measured using the partition coefficient (log P). For PLGA microspheres or oily depots, a higher log P (>2) helps the drug slowly partition from the depot into surrounding tissue. Highly hydrophilic molecules may show burst release unless modified into lipophilic prodrugs or encapsulated in complex systems. Drug ionization and pKa also influence release behavior. Optimizing physicochemical properties helps maintain sustained exposure and reduce toxicity risk.
Effective dose is often the most limiting factor. FDA-approved long-acting injectables usually require a monthly dose of 100 mg or less. Higher doses increase injection volume and may cause injection site reactions and poor syringeability. High-dose molecules may require nanocrystal suspensions or implants. Dose also affects needle selection, injection force, and patient comfort. Manufacturing feasibility must be considered for high-load formulations.
Explore characterization strategies: Analytical Challenges in Characterizing Long-Acting Injectables
| Physicochemical Attribute | Ideal Range for LAI Development | Technical Justification |
|---|---|---|
| Effective Monthly Dose | < 100 mg | Minimizes depot volume and prevents excessive inflammation at the injection site |
| Partition Coefficient (log P) | > 2.0 | Facilitates slow partitioning from polymer matrix to tissue fluid |
| Molecular Weight | < 1000 Da (small molecules) | Influences diffusion rate and packing density |
| Plasma Clearance | Low to Moderate | Reduces required release rate |
| Crystallinity | High (for suspensions) | Slows dissolution according to Noyes-Whitney equation |
Prodrug Engineering for Solubility Modulation in Long Acting Injectable Formulation Development
When a drug shows poor solubility, lipophilic prodrug design is a proven approach in Long Acting Injectable Formulation Development. Fatty acid chains such as decanoate, palmitate, or enanthate can be attached to increase lipophilicity. This allows the drug to dissolve in oils or triglycerides and form long-acting depots. This approach is widely used in antipsychotic formulations. The fatty acid chain length affects Tmax and Cmax by controlling hydrolysis rate at the injection site. Prodrug stability, enzymatic cleavage, and storage conditions must be carefully evaluated to ensure predictable performance.
Comparative Analysis of Long Acting Injectable Delivery Platforms
Choosing between microspheres, nanocrystals, and in situ forming implants requires balancing drug loading, manufacturing complexity, and release duration. Each platform uses a different mechanism such as polymer erosion or dissolution control. Platform selection also depends on drug properties, dose requirement, and clinical considerations. A suitable delivery platform improves patient compliance and therapeutic performance.
Compare development models: Choosing Between a Peptide CDMO vs CRO for Complex Formulations
Polymeric Microspheres: PLGA-Based Sustained Release
Polymeric microspheres are widely used in Long Acting Injectable Formulation Development for peptides, proteins, and small molecules. The drug is encapsulated in PLGA and released through diffusion, pore formation, and polymer erosion. PLGA degradation occurs through ester bond hydrolysis. Polymer molecular weight and lactide:glycolide ratio determine release duration. Residual solvent control and particle morphology are important for consistent release. Emulsification parameters must be optimized for reproducible batches.
Nanocrystal Suspensions: The High-Load Alternative
Nanocrystal suspensions reduce particle size to sub-micron levels to enable high drug loading. Concentrations up to 300 mg/mL are possible. Drug release is controlled by dissolution of crystalline particles. Stabilizers prevent aggregation and maintain particle size. Milling and high-pressure homogenization are common manufacturing methods. Physical stability and polymorphic control are key development challenges.
In Situ Forming Implants (ISFI) and Phase Separation
In situ forming implants create a depot after injection. Polymer and drug dissolved in solvent precipitate when exposed to body fluid. This forms a solid matrix. ISFIs are easy to manufacture but may show initial burst release. Solvent selection, polymer concentration, and phase inversion kinetics influence performance. Proper optimization reduces burst release and improves sustained delivery.
| Platform Type | Mechanism of Release | Typical Duration | Major Challenge |
|---|---|---|---|
| Microspheres | Diffusion & Matrix Erosion | 2–12 Weeks | Solvent residue & scale-up complexity |
| Nanocrystals | Dissolution-Controlled | 1–4 Weeks | Physical stability & Ostwald ripening |
| ISFI | Phase Inversion / Precipitation | 4–24 Weeks | High initial burst release |
| Oily Depots | Partitioning-Controlled | 1–4 Weeks | Injection site pain & viscosity |
| Liposomes | Diffusion through Bilayers | 1–2 Weeks | Low drug loading capacity |
Quality by Design (QbD) and Critical Quality Attributes (CQAs)
Quality by Design is essential in Long Acting Injectable Formulation Development to ensure consistent product quality. QbD identifies critical material attributes and process parameters that influence safety and efficacy. Design space definition allows controlled flexibility. Statistical tools such as design of experiments help optimize formulation variables.
Read the full breakdown: Particle Characterization Case Study for Lupron Depot
Identification of CQAs in Polymeric Systems
Important CQAs include particle size distribution, porosity, drug loading, and in vitro release. For PLGA microspheres, Span value predicts burst release behavior. Uniform particle size improves reproducibility. Surface morphology and drug distribution affect release kinetics. Monitoring CQAs ensures consistent performance.
Internal porosity is also important. Highly porous particles absorb water quickly and accelerate polymer degradation. Dense particles may cause delayed release. Imaging tools like SEM help evaluate structure. Controlled porosity balances burst release and lag phase.
Critical Process Parameters (CPPs) in Microencapsulation
Key CPPs during solvent evaporation include homogenization speed, temperature, and solvent removal rate. These factors influence particle morphology and encapsulation efficiency.
- Homogenization Speed: Controls droplet size and final particle size
- Solvent Extraction Rate: Rapid removal may trap solvent and fracture particles
- Phase Ratio: Influences encapsulation efficiency and emulsion stability
Proper optimization ensures consistent release and stability.
Engineering Challenges in Manufacturing Scale-Up
Scaling Long Acting Injectable Formulation Development from lab to commercial scale is challenging due to changes in fluid dynamics and solvent removal. Small-scale equipment behaves differently from large reactors. Scale-up requires modeling of mixing, heat transfer, and shear forces.
Maintaining Geometric and Kinematic Similarity
Successful scale-up requires maintaining similar power-to-volume ratio and impeller tip speed. As vessel size increases, solvent evaporation slows. Computational fluid dynamics helps predict mixing. Proper scale-up reduces variability and maintains particle size distribution.
Downstream Processing: Purification and Isolation
After formation, particles must be washed and concentrated. Tangential Flow Filtration (TFF) is widely used. TFF enables closed processing, reduces contamination risk, and supports large-scale manufacturing. Membrane selection and washing cycles affect purity and stability.
| Scale-Up Variable | Laboratory Scale (100 mL) | Commercial Scale (100 L) | Impact of Misalignment |
|---|---|---|---|
| Mixing Energy | High localized shear | Distributed shear | Broad particle size |
| Surface/Volume Ratio | High | Low | Slow solvent extraction |
| Heat Transfer | Rapid | Slower | Variable polymer solidification |
| Purification Method | Centrifugation | TFF | Aggregation risk |
| Drying Process | Small lyophilization | Industrial freeze drying | Residual moisture variation |
Discover specialized support: CRO Services for Complex Injectable Development
Sterilization Protocols in Long Acting Injectable Formulation Development
Sterility is a major requirement in Long Acting Injectable Formulation Development. Regulatory agencies prefer terminal sterilization, but complex formulations often require aseptic processing. Sterilization strategy must be selected early in development.
The Preference for Terminal Sterilization
Terminal sterilization occurs in the final container and provides high sterility assurance. Common methods include:
- Moist Heat (Autoclaving) – unsuitable for PLGA systems
- Gamma or E-beam Radiation – may degrade API or polymer
- Ethylene Oxide Gas – limited use due to residual toxicity
Each method requires stability validation.
Aseptic Processing and Cleanroom Requirements
If terminal sterilization is not possible, aseptic processing is used. Components are sterilized separately and assembled in cleanrooms. Environmental monitoring, media fills, and operator training are critical. This approach increases cost but supports sensitive formulations.
Regulatory Strategy and the 505(b)(2) Pathway
The 505(b)(2) pathway is widely used in Long Acting Injectable Formulation Development to reduce development time and cost. This pathway allows reliance on existing safety data for a reference drug. It is suitable for converting oral or short-acting drugs into long-acting injectables.
Building the Scientific Bridge
The scientific bridge includes comparative pharmacokinetic and bioavailability studies. The goal is to show similar therapeutic exposure without new safety risks. Modeling, animal studies, and human PK studies may be required.
CMC Challenges and Complete Response Letters (CRLs)
Many 505(b)(2) delays occur due to CMC issues such as:
- Impurity profiles
- Stability and shelf life
- J-code and payer strategy
Regulatory agencies require strong process validation and batch consistency.
Understand the regulatory requirements: Analytical Requirements for ANDA Generic Drug Submissions
Analytical Innovations: PAT and Real-Time Release Kinetics
Advanced analytics are essential in Long Acting Injectable Formulation Development. Process Analytical Technology allows real-time monitoring. These tools improve process control and reduce batch failures.
Real-Time Monitoring with Inline NMR and FTIR
Inline spectroscopy tracks solvent concentration and chemical changes during manufacturing. These tools are useful for nanocrystal production and crystallization monitoring. Real-time feedback improves batch consistency.
Chemical Shift Imaging (CSI) for In Situ Release
CSI-NMR enables visualization of depot formation and drug diffusion. This non-destructive method provides insight into burst release and sustained phase behavior.
Establishing Level A IVIVC in Long Acting Injectable Formulation Development
Level A IVIVC predicts in vivo performance from in vitro data. This helps manage lifecycle changes and reduce bioequivalence studies. Strong correlation improves regulatory acceptance.
USP Apparatus 4 and Biorelevant Media
USP Apparatus 4 is preferred for long-acting injectables. It uses flow-through cells and biorelevant media. Scientists typically:
- Prepare formulations with different release rates
- Conduct animal PK studies
- Apply deconvolution methods for correlation
This approach supports robust IVIVC development.
Conclusion
Long Acting Injectable Formulation Development plays an important role in modern pharmaceutical innovation. By optimizing polymer behavior, selecting the right delivery platform, and implementing Quality by Design, companies can develop reliable long-acting therapies. Strong regulatory strategy, advanced analytics, and scalable manufacturing are essential for success. As analytical technologies and polymer science continue to improve, Long Acting Injectable Formulation Development will support the creation of patient-friendly therapies with predictable release and improved adherence.
Expert Case Study: Leuprolide Depot Sustained Release Development Case Study
For expert partnership in your next formulation project, contact our team at:
https://resolvemass.ca/contact/
Frequently Asked Questions in Long Acting Injectable Formulation Development
For a three-month release, PLGA with a higher lactide content and higher molecular weight is usually preferred. Ratios such as 75:25 or 85:15 slow water uptake and delay polymer breakdown. This reduces hydrolysis speed and extends drug release. End-group chemistry and polymer viscosity should also be considered to fine-tune the duration.
The initial burst mainly happens when the solvent diffuses quickly into surrounding tissue before the polymer solidifies. During this early phase, the drug is not fully trapped in the depot. As a result, a portion of the drug escapes rapidly. Polymer concentration and solvent choice can help reduce this effect.
USP Apparatus 4 uses a flow-through system that keeps particles suspended and prevents settling. This setup better reflects the environment at the injection site. It also allows testing with smaller volumes and controlled flow conditions. These features provide more reliable and discriminatory release data for long-acting injectables.
Terminal sterilization is difficult for peptide-loaded PLGA systems because heat can soften the polymer and cause particle aggregation. Radiation methods may also damage peptides or break polymer chains. These changes can alter release behavior and reduce product stability. Therefore, aseptic manufacturing is often used instead.
A scientific bridge connects the new long-acting product to an already approved drug. This is usually done using comparative pharmacokinetic or bioavailability studies. The goal is to show similar exposure without introducing new safety risks. This approach reduces the need for extensive clinical trials.
Ostwald ripening occurs when smaller particles dissolve and redeposit onto larger ones over time. This causes gradual growth in particle size. Larger particles may change dissolution rate and injection performance. Stabilizers and optimized storage conditions help minimize this effect.
TFF provides a gentle and scalable method for washing and concentrating particles. It operates in a closed system, which helps maintain sterility. Compared to centrifugation, it reduces aggregation and mechanical stress. This makes TFF suitable for large-scale injectable manufacturing.
A validated Level A IVIVC links in vitro release with in vivo drug exposure. Once accepted, it can predict how formulation changes affect pharmacokinetics. If predictions show no meaningful impact, regulators may allow changes without new clinical studies. This speeds up product lifecycle management.
Reference:
- Otte, A., Park, K., & Li, T. (2026). Challenges and innovations in long-acting injectable formulations: Can formulation design space be rationalized? Journal of Pharmacy and Pharmacology, 78(1), rgaf080. https://doi.org/10.1093/jpp/rgaf080
- Bao, Q., Wang, X., Wan, B., Zou, Y., Wang, Y., & Burgess, D. J. (2023). Development of in vitro-in vivo correlations for long-acting injectable suspensions. International Journal of Pharmaceutics, 634, 122642. https://doi.org/10.1016/j.ijpharm.2023.122642
- Nakmode, D. D., Singh, B., Abdella, S., Song, Y., & Garg, S. (2025). Long-acting parenteral formulations of hydrophilic drugs, proteins, and peptide therapeutics: Mechanisms, challenges, and therapeutic benefits with a focus on technologies. Drug Delivery and Translational Research, 15(4), 1156–1180. https://doi.org/10.1007/s13346-024-01747-y
- Thapa Magar, K., Boucetta, H., Zhao, Z., Xu, Y., Liu, Z., & He, W. (2026). Insights into development of long-acting injectable suspensions. Expert Opinion on Drug Delivery. Advance online publication. https://pubmed.ncbi.nlm.nih.gov/41690479/
- Alidori, S., Subramanian, R., & Holm, R. (2024). Patient-centric long-acting injectable and implantable platforms—An industrial perspective. Molecular Pharmaceutics, 21(9), 4238–4258. https://doi.org/10.1021/acs.molpharmaceut.4c00665
- Gonella, A., Grizot, S., Liu, F., López Noriega, A., & Richard, J. (2022). Long-acting injectable formulation technologies: Challenges and opportunities for the delivery of fragile molecules. Expert Opinion on Drug Delivery, 19(8), 927–944. https://doi.org/10.1080/17425247.2022.2105318
- Cleland, J. L., Powell, M. F., & Shire, S. J. (2001). The development of stable protein formulations: A review of protein aggregation, deamidation, and oxidation. Critical Reviews in Therapeutic Drug Carrier Systems, 18(3), 229–279. https://pubmed.ncbi.nlm.nih.gov/10810750/



