Introduction:
The pharmaceutical industry is steadily moving away from daily oral medicines and toward advanced depot systems. This change is mainly driven by ongoing challenges with patient adherence and the need for more consistent drug performance in the body. When comparing Long Acting Injectable vs Oral Drug Delivery, it becomes clear that injectable options can offer more stable and predictable results. These systems help maintain steady drug levels in the bloodstream and reduce issues linked to digestion and absorption. In addition, companies are using these formulations to extend product value as patents expire. Because of these benefits, depot systems are becoming a major focus in modern drug development.
Explore the broader landscape of modern drug production: Pharmaceutical CDMO Services in the US and Canada
Share via:
The Pharmacokinetic Advantage of Long Acting Injectable vs Oral Drug Delivery
One of the key benefits of Long Acting Injectable vs Oral Drug Delivery is the ability to maintain a stable therapeutic range while avoiding the digestive system. Oral drugs must pass through several biological steps before reaching the bloodstream, which can lead to inconsistent absorption. This variation can affect how well the drug works in different patients.
In contrast, long-acting injectables deliver medication directly into muscle or under the skin, where it is released slowly over time. This controlled release leads to fewer ups and downs in drug levels. For medicines that require precise dosing, this stability is very important. Over time, it supports better treatment results and lowers the risk of side effects caused by sudden peaks in drug concentration.
Learn more about the technical hurdles in formulation: Addressing Analytical Challenges in Leuprolide Depot Development
Bypassing First-Pass Metabolism and Improving Bioavailability
Oral drug delivery comes with several natural challenges. After a pill is swallowed, it must survive stomach acid and enzymes before being absorbed in the intestine. Once absorbed, the drug travels to the liver, where a portion may be broken down before reaching circulation. This process, known as first-pass metabolism, can reduce the effectiveness of the drug.
Because of this, higher doses are often needed, which may increase the chance of side effects. Differences in liver enzyme activity between patients can also lead to unpredictable results. This makes it harder to maintain consistent treatment outcomes across different individuals.
Long-acting injectables avoid this issue by delivering the drug directly into the body’s tissues. From there, the medication is slowly released into the bloodstream, bypassing the liver’s first-pass effect. This leads to higher and more consistent availability of the drug. As a result, lower doses are often enough, which improves safety and reduces the risk of toxicity over long-term use.
Understand the core components of successful market entry: CMC Strategy for Leuprolide Depot Formulations
Understanding Flip-Flop Kinetics in Long Acting Injectable vs Oral Drug Delivery
In the discussion of Long Acting Injectable vs Oral Drug Delivery, the concept of flip-flop kinetics helps explain why depot systems last longer. With oral drugs, absorption is usually quick, and the body’s elimination process determines how long the drug stays active. This often leads to short durations of action.
However, in depot systems, the drug is designed to be released slowly from the injection site. This means absorption becomes the limiting factor rather than elimination. As a result, the drug remains active for a longer period, even if it normally has a short half-life.
This extended activity allows patients to take medication less often, sometimes only once a month or even less frequently. It also creates a smoother drug profile in the body, avoiding the highs and lows commonly seen with oral medications.
| Pharmacokinetic Parameter | Oral Dosage Forms | Long-Acting Injectables (Depot) |
| Primary Route | Enteral (GI Tract) | Parenteral (IM or SC) |
| First-Pass Effect | Significant; can deactivate up to 90% of API | Bypassed; direct systemic entry |
| Bioavailability ($F$) | Often < 50% due to GI/Liver barriers | High; often approaching 100% |
| Plasma Profile | Rapid peaks ($C_{max}$) and deep troughs | Consistent steady-state concentrations |
| Elimination Half-Life | Determined by clearance ($Cl$) | Determined by absorption rate ($k_a$) |
| Metabolic Impact | Higher hepatic load per dose | Lower systemic dose requirement |
Clinical Shift Toward Depot Systems
The move toward depot systems is strongly supported by improved clinical outcomes. In many conditions, missing even a few doses of oral medication can lead to serious complications. This includes disease progression, relapse, or resistance to treatment.
Depot systems help solve this issue by ensuring consistent drug delivery over time. Healthcare providers can more easily track whether a patient is following treatment. At the same time, patients benefit from simpler routines, which improves overall quality of life. These advantages make depot systems an attractive option in long-term disease management.
Addressing Non-Adherence in Mental Health
Mental health conditions such as schizophrenia often involve challenges with medication adherence. Patients may forget doses or stop treatment due to lack of awareness about their condition. This can lead to repeated relapses and hospital visits.
Long-acting injectables provide a reliable solution by offering controlled dosing through scheduled injections. This ensures that medication is consistently delivered without requiring daily effort from the patient. Clinicians can quickly identify missed appointments and take action before symptoms worsen.
Studies show that patients using injectable treatments experience fewer hospitalizations and improved stability. Over time, this reduces the burden on caregivers and healthcare systems while improving patient outcomes.
Transforming HIV Treatment and Prevention
Depot systems are also changing how HIV is treated and prevented. Instead of taking daily pills, patients can now receive long-acting injections that last for weeks or months. This greatly reduces the risk of missed doses.
Clinical research has shown that these injectable therapies improve adherence and overall outcomes. They are especially helpful for individuals who struggle with daily medication routines. In prevention, long-acting options provide continuous protection without requiring daily action.
This approach supports better public health outcomes by lowering transmission rates and improving long-term disease control.
See how these systems apply to complex drug categories: Regulatory Pathways for Complex Peptide Injectables
Expanding Use Across Therapeutic Areas
Depot systems are no longer limited to a few medical fields. In oncology, they are used for sustained hormone therapy in cancers such as prostate cancer. In endocrinology, they help maintain stable hormone levels over time.
In immunology, longer dosing intervals reduce the need for frequent clinic visits. This makes treatment more convenient and less stressful for patients. As a result, depot systems are becoming an important part of care across many specialties.
Strategic Value in Long Acting Injectable vs Oral Drug Delivery
From a business perspective, Long Acting Injectable vs Oral Drug Delivery also plays a key role in lifecycle management. When patents expire, pharmaceutical companies face competition from generic drugs, which can reduce revenue.
By developing long-acting versions of existing medications, companies can extend product life and maintain market share. These improved formulations also offer better patient experiences, which strengthens brand value in competitive markets.
Accelerate your development timeline: How CDMOs Speed Up Generic Drug Development
The Patent Cliff and Evergreening Strategies
The patent cliff is a major challenge for drug manufacturers. Once a patent expires, generic versions enter the market and quickly lower prices. This leads to a sharp drop in revenue for the original product.
To manage this, companies use strategies like evergreening. This involves creating new versions of existing drugs, such as long-acting injectables, that can be protected by new patents. These updated products help maintain revenue while offering improved treatment options for patients.
Technical Barriers That Protect Innovation
Depot systems are complex and require advanced technology to develop and manufacture. This creates a barrier for generic competitors, who must match both the chemical composition and release behavior of the original product.
Achieving the same performance is difficult and costly, which limits competition. As a result, branded depot products often retain their value for longer periods compared to standard oral medications.
| Business Driver | Impact on Strategy | Outcome for Pharma |
| Patent Cliff | Mitigates revenue loss from generic entry | Sustained profitability |
| Evergreening | Extends exclusivity via new delivery systems | Market share protection |
| Technical Complexity | High barrier to generic competition | Reduced number of competitors |
| Market Differentiation | u-LAIs offer “premium” convenience | Higher unit pricing power |
| HCP Engagement | Professional administration builds provider ties | Brand loyalty and monitoring |
Advanced Technologies in Depot Systems
Modern depot systems use a range of advanced technologies to control how drugs are released. These include biodegradable polymers, lipid-based carriers, and nanotechnology platforms. Each method offers unique advantages in controlling drug delivery.
These technologies allow scientists to design treatments with precise release patterns. This improves effectiveness and reduces side effects. Ongoing innovation continues to expand the possibilities for long-acting formulations.
Deep dive into specialized formulation tech: Long-Acting Injectable Drug Delivery Technologies
PLGA Microsphere Platforms
PLGA microspheres are widely used in depot formulations because they are safe and biodegradable. The drug is enclosed within the polymer and released slowly as the material breaks down in the body.
By adjusting the polymer structure, scientists can control how long the drug is released. This flexibility allows treatments to be tailored to specific medical needs. The breakdown products are naturally removed by the body, making this approach suitable for long-term use.
In Situ Forming Implants (ISFI) and Atrigel® Technology
ISFI systems are injected as liquids but quickly form solid depots داخل the body. This simplifies the manufacturing process compared to pre-formed particles. The result is a consistent and controlled release of medication over time.
This technology reduces production challenges while maintaining reliable performance. Continued improvements are making these systems even more effective and easier to produce at scale.
Nanocrystalline and NLLC Suspensions
Nanocrystal technology improves drug delivery for compounds that do not dissolve easily. By reducing particle size, the drug dissolves more slowly, which supports sustained release.
NLLC systems form structured gels that control how the drug moves through the body. Together, these approaches expand the range of medicines that can be developed as depot formulations.
Liposomal and Lipid-Based Depots
Liposomal systems use tiny lipid bubbles to carry drugs and release them gradually. They are especially useful for targeted delivery and shorter-duration treatments.
Advanced lipid-based systems improve stability and control over drug release. These innovations continue to support the development of more effective long-acting therapies.
Overcoming Manufacturing Challenges
Developing depot systems requires careful attention to stability, sterility, and scalability. Maintaining drug quality over long periods can be difficult and requires precise formulation.
Manufacturers must also ensure that products perform consistently during large-scale production. Addressing these challenges is essential to deliver safe and reliable treatments to patients.
Optimize your production strategy: CDMO Services for Long-Acting Injectable Formulation
Managing Initial Burst Release
Burst release refers to a rapid release of drug shortly after injection. This can lead to high drug levels initially and reduce how long the treatment lasts.
To control this, scientists adjust formulation properties and surface characteristics. These strategies help create a more balanced and predictable release profile, improving patient safety.
Sterilization and Stability Considerations
Sterility is critical for injectable products, but not all materials can handle traditional sterilization methods. This often requires aseptic manufacturing processes.
Although more complex, these methods ensure that the final product is safe for patient use. Advances in sterile processing are helping improve efficiency and reliability.
Scale-Up and Ostwald Ripening Challenges
Scaling up production introduces new variables that can affect product quality. Factors like temperature, mixing, and storage conditions must be tightly controlled.
Ostwald ripening, a process where particles grow over time, can impact stability in suspensions. Advanced manufacturing techniques are used to prevent this and maintain consistency.
Regulatory Requirements in Long Acting Injectable vs Oral Drug Delivery
Regulatory approval for depot systems is more demanding than for oral drugs. Companies must provide detailed data showing consistent performance and safety.
These strict requirements ensure that patients receive high-quality treatments. They also maintain trust in advanced drug delivery technologies.
Navigate the complexities of ANDA filings: Analytical Requirements for Generic Drug ANDA Submissions
Demonstrating Bioequivalence in Complex Generics
Generic versions of depot systems must match both the composition and behavior of the original product. This involves detailed pharmacokinetic studies and testing.
Meeting these standards is challenging but necessary to ensure safety and effectiveness for patients.
The Role of IVIVC in Quality Control
IVIVC, or in vitro–in vivo correlation, connects lab test results with actual clinical performance. This helps predict how formulation changes will affect outcomes.
It reduces the need for repeated clinical trials and supports more efficient development processes.
Ensuring Compliance in Sterile Manufacturing
Facilities that produce injectable drugs must meet strict regulatory standards. Advanced systems are used to prevent contamination and maintain product quality.
These measures are essential for ensuring patient safety and maintaining industry standards.
| Technology Trend | Clinical Benefit | Future Outlook |
| u-LAI (6-12 Months) | Eliminates daily pill burden entirely | Standard of care for chronic prophylaxis |
| Biorobotic Depots | Electronically controlled release rates | Personalized, real-time dosing |
| Biodegradable Implants | No surgical removal required | Lower patient trauma and higher compliance |
| AI-Integrated Monitoring | Alerts HCPs to missed doses in real-time | Predictive adherence and relapse prevention |
| Nano-Suspensions | High-payload delivery in small volumes | Broadens molecules suitable for LAI |
Future Trends: Patient-Centric and Digital Approaches
The future of drug delivery is becoming more patient-focused and supported by digital tools. Information must be clear and easy to understand for both healthcare providers and patients.
This helps improve decision-making and ensures better treatment outcomes across different populations.
Define your quality standards for the future: Critical Quality Attributes for Long-Acting Injectables
Ultra Long-Acting Injectables (u-LAI)
Ultra long-acting injectables are designed to last for several months or even up to a year. These systems could remove the need for frequent dosing entirely.
They represent the next stage of innovation in drug delivery, offering greater convenience and improved adherence for chronic conditions.
Economic Impact of Depot Systems
Although depot systems may have higher upfront costs, they often reduce overall healthcare expenses. Better adherence leads to fewer complications and hospital visits.
This makes them a cost-effective option in the long term while improving patient outcomes.
Reducing Hospitalizations Through Better Adherence
Patients using depot systems are less likely to miss doses, which leads to more stable treatment. This reduces the need for hospital care and emergency interventions.
Improved consistency also supports better long-term health and quality of life.
Addressing the Cost of Drug Resistance
In diseases like HIV, missed doses can lead to drug resistance, which is expensive and difficult to manage. Depot systems help prevent this by ensuring steady drug levels.
This reduces long-term treatment costs and supports better public health outcomes.
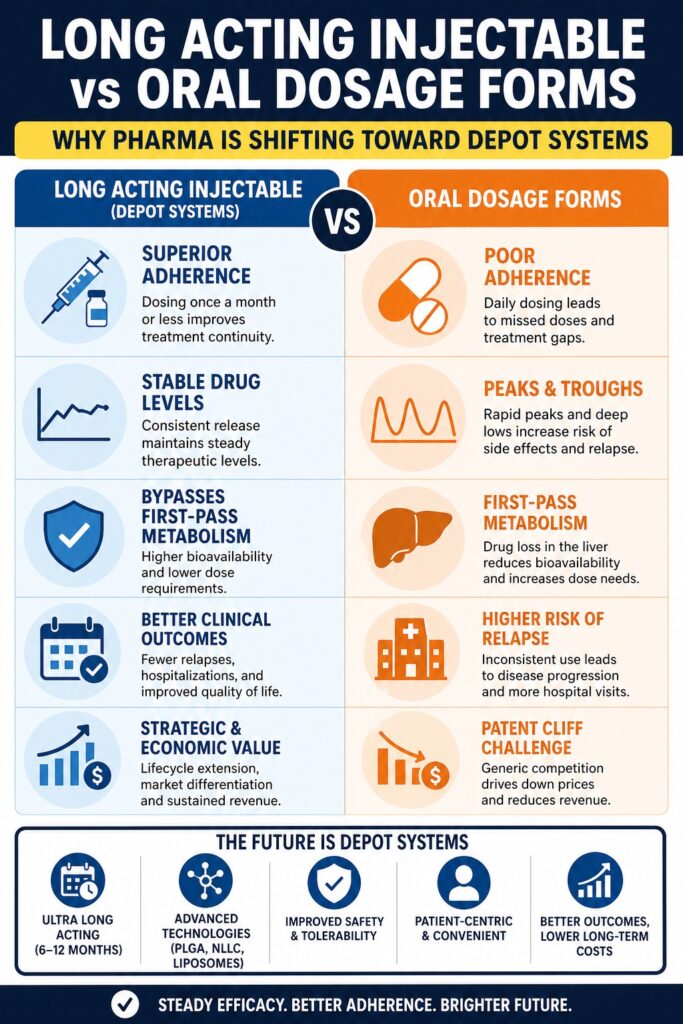
Conclusion: The Future of Long Acting Injectable vs Oral Drug Delivery
The comparison of Long Acting Injectable vs Oral Drug Delivery clearly shows a growing preference for depot systems. These formulations improve adherence, provide stable drug levels, and reduce overall healthcare costs.
With ongoing technological advancements, long-acting injectables are becoming more effective and widely used. For pharmaceutical companies, investing in these systems supports innovation and long-term growth. For patients, they offer simpler and more reliable treatment options, making it easier to manage chronic conditions and maintain a better quality of life.
Most Frequently Asked Questions: Long Acting Injectable vs Oral Drug Delivery
Long-acting injectables and slow-release tablets both aim to extend how long a drug works, but they do it in very different ways. Oral pills depend on the digestive system, which means food, stomach acid, and gut health can affect how the drug is absorbed. In Long Acting Injectable vs Oral Drug Delivery, injectables form a depot in the tissue and release the drug directly into the bloodstream over time. This leads to more stable and predictable drug levels in the body.
Not all oral drugs can be turned into long-acting injectables. The drug must be strong enough to work in small doses and remain stable in the body for an extended time. It also needs the right physical and chemical properties, such as solubility and stability. ResolveMass Laboratories Inc. evaluates these factors to determine whether a drug is suitable for depot-based delivery systems.
An oral lead-in phase is used to confirm that the patient can tolerate the drug safely. Since long-acting injectables stay in the body for weeks or even months, they cannot be quickly stopped if side effects occur. By first giving the oral version, doctors can monitor for any adverse reactions. Once safety is confirmed, the patient can move to the injectable form with lower risk.
Long-acting injectables usually allow a short grace period if an appointment is missed. However, delaying treatment for too long can lower drug levels and increase the risk of symptoms returning. One advantage of these systems is that missed doses are quickly noticed by healthcare providers. This allows timely follow-up and helps keep the patient on track with their treatment plan.
In most cases, long-acting injectables do not increase side effects and may even reduce them. Oral drugs can create sharp peaks in drug levels, which often trigger unwanted reactions like nausea or dizziness. Depot systems release the drug slowly, avoiding these spikes. The most common additional effect is mild pain or redness at the injection site, which usually goes away quickly.
Although they may look similar, long-acting injectables and vaccines serve very different purposes. A vaccine delivers a quick dose to stimulate the immune system, often in a single visit. In contrast, depot injections are designed to release medication slowly over time. They use advanced carrier systems to control drug release and maintain steady levels in the body.
Long-acting injectables were first widely used in psychiatric care, but their use has expanded significantly. Today, they are used in areas such as cancer treatment, hormone therapy, HIV care, and chronic conditions like asthma and osteoporosis. Ongoing research is also exploring their use in diabetes and heart-related diseases, showing their growing importance in modern medicine.
Reference:
- Correll, C. U., Kim, E., Sliwa, J. K., Hamm, W., Gopal, S., Mathews, M., Venkatasubramanian, R., & Saklad, S. R. (2021). Pharmacokinetic characteristics of long-acting injectable antipsychotics for schizophrenia: An overview. Clinical Pharmacokinetics, 60(1), 39–59. https://doi.org/10.1007/s40263-020-00779-5
- Hodge, D., Back, D. J., Gibbons, S., Khoo, S. H., & Marzolini, C. (2021). Pharmacokinetics and drug–drug interactions of long-acting intramuscular cabotegravir and rilpivirine. Clinical Pharmacokinetics, 60(7), 835–853. https://doi.org/10.1007/s40262-021-01005-1
- World Health Organization. (2025). Guidelines on lenacapavir for HIV prevention and testing strategies for long-acting injectable pre-exposure prophylaxis (PrEP). NCBI Bookshelf. https://www.ncbi.nlm.nih.gov/books/NBK595006/
- Nakmode, D. D., Singh, B., Abdella, S., Song, Y., & Garg, S. (2024). Long-acting parenteral formulations of hydrophilic drugs, proteins, and peptide therapeutics: Mechanisms, challenges, and therapeutic benefits with a focus on technologies. Drug Delivery and Translational Research. https://pmc.ncbi.nlm.nih.gov/articles/PMC11435547/
- Brissos, S., Veguilla, M. R., Taylor, D., & Balanzá-Martínez, V. (2014). The role of long-acting injectable antipsychotics in schizophrenia: A critical appraisal. Therapeutic Advances in Psychopharmacology, 4(5), 198–219. https://doi.org/10.1177/2045125314540297
- Wei, Y., Yan, V. K. C., Kang, W., Wong, I. C. K., Castle, D. J., Gao, L., Chui, C. S. L., Man, K. K. C., Hayes, J. F., Chang, W. C., & Chan, E. W. (2022). Association of long-acting injectable antipsychotics and oral antipsychotics with disease relapse, health care use, and adverse events among people with schizophrenia. JAMA Network Open, 5(7), e2224163. https://doi.org/10.1001/jamanetworkopen.2022.24163



