Introduction
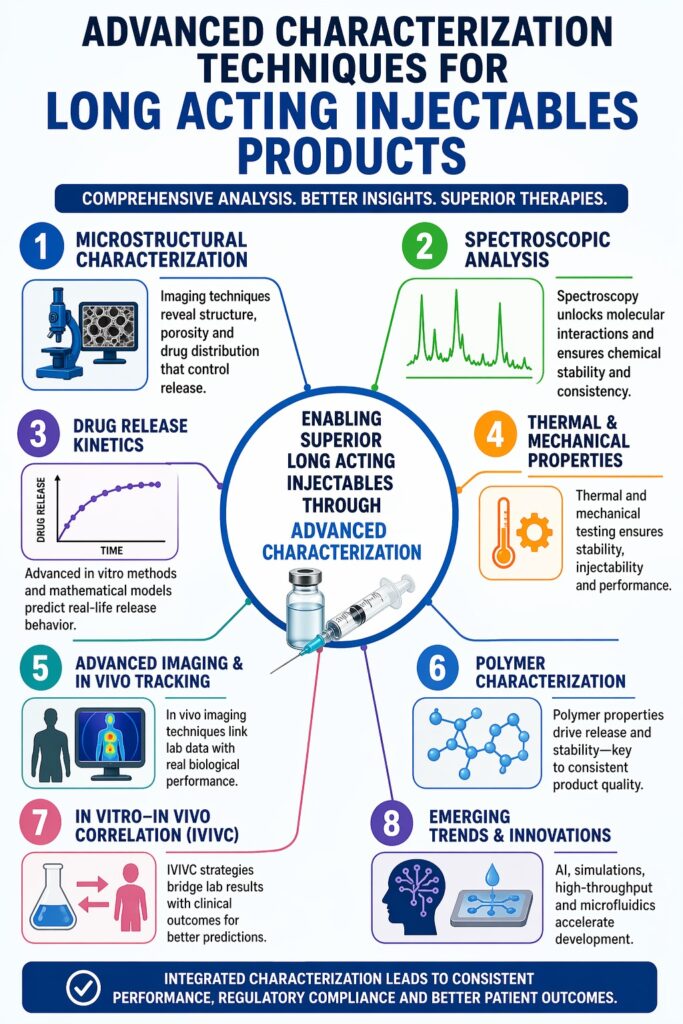
Long Acting Injectables Characterization focuses on understanding microstructure, release mechanisms, and polymer-drug interactions using advanced analytical techniques. This approach allows scientists to study how different formulation factors affect product performance. It also supports the development of safer and more effective drug delivery systems.
Long-acting injectables (LAIs), especially polymer-based microspheres and depot systems, are complex in nature. Their structure is not uniform, and they release drugs over a long period. Because of this complexity, simple testing methods are not enough. Researchers must use a combination of analytical tools to fully understand these systems.
Explore the essential requirements for ANDA submission of generic drugs to ensure regulatory compliance.
Unlike traditional dosage forms, Long Acting Injectables Characterization requires combining structural, chemical, and kinetic data. This integrated approach ensures that the product performs consistently and meets strict regulatory standards. It also helps reduce risks during development and approval.
Modern characterization goes beyond basic measurements like particle size. It focuses on connecting physical and chemical properties with actual in vivo performance. This connection is critical to ensure consistent drug delivery and patient safety over time.
Learn how a pharmaceutical CDMO in the US and Canada can streamline your development pipeline.
Share via:
1. How Do Microstructural Characterization Techniques Influence LAI Performance?
Microstructure plays a key role in drug release behavior, making imaging techniques very important in Long Acting Injectables Characterization. The internal structure of the formulation directly affects how the drug diffuses and how the polymer breaks down. Even small changes in structure can lead to noticeable differences in release patterns.
Advanced imaging tools provide detailed information about porosity, internal structure, and drug distribution. These features are essential for controlling both initial burst release and long-term sustained release. Clear visualization helps scientists improve formulation design and consistency.
Key Techniques:
- Scanning Electron Microscopy (SEM)
Reveals surface structure and changes in pore formation over time. It helps detect defects that may impact drug release. - Transmission Electron Microscopy (TEM)
Provides nanoscale images of drug and polymer distribution. It is useful for identifying phase separation. - X-ray Microcomputed Tomography (micro-CT)
Offers non-destructive 3D imaging of internal structure. It allows repeated analysis of the same sample. - Confocal Laser Scanning Microscopy (CLSM)
Tracks drug movement in real time using fluorescent markers. This helps in understanding release behavior.
Studies show that porosity and internal channels strongly influence both burst and sustained release. Proper control of these features leads to more predictable and stable drug delivery outcomes.
Understand the specific analytical requirements for ANDA generic drugs to avoid development delays.
2. What Role Do Spectroscopic Techniques Play in Long Acting Injectables Characterization?
Spectroscopic methods are essential in Long Acting Injectables Characterization because they provide molecular-level information. These techniques help identify chemical interactions that cannot be seen through imaging alone. They are critical for understanding stability and drug release mechanisms.
Core Spectroscopic Tools:
- FTIR
Identifies functional group interactions and checks drug-polymer compatibility. - Raman Spectroscopy
Provides chemical mapping within the formulation without damaging the sample. - Solid-State NMR
Gives insight into molecular movement and crystallinity changes. - UV-Vis Spectroscopy
Commonly used for measuring drug concentration and release rates.
Raman mapping helps detect uneven drug distribution, which is important for batch consistency. Solid-state NMR adds further understanding by tracking structural changes over time. Together, these tools create a complete molecular profile.
3. How Are Drug Release Kinetics Characterized in LAIs?
Drug release studies combine lab testing with modeling to predict real-life behavior. This combined approach improves both understanding and accuracy. It is a core part of Long Acting Injectables Characterization.
Advanced In Vitro Release Testing Methods:
- Dialysis-based systems
- Sample-and-separate methods
- USP apparatus (e.g., flow-through cells)
- Microfluidic systems
These methods aim to mimic real physiological conditions as closely as possible. Choosing the right method is important for reliable data.
Discover how a CDMO can accelerate generic drug development in the US and Canada through advanced kinetics studies.
Key Analytical Outputs:
- Burst release phase
- Diffusion-controlled release
- Polymer degradation-driven release
Mathematical Models Used:
- Higuchi model
- Korsmeyer–Peppas model
- Weibull model
These models help interpret results and predict long-term drug release. They are also useful for regulatory documentation and validation.
4. Why Is Thermal and Mechanical Characterization Critical?
Thermal and mechanical properties are essential for ensuring product stability and performance. These factors affect both manufacturing and patient experience. In Long Acting Injectables Characterization, they help predict how the formulation behaves during and after injection.
Thermal Techniques:
- Differential Scanning Calorimetry (DSC)
Measures glass transition temperature, which affects drug diffusion. - Thermogravimetric Analysis (TGA)
Evaluates thermal stability and degradation behavior.
Mechanical & Rheological Testing:
- Viscosity and injectability studies
- Needle force testing
- Depot formation analysis
Understanding these properties ensures smooth administration and consistent drug delivery.
5. How Do Advanced Imaging and In Vivo Tracking Techniques Improve Characterization?
Modern imaging methods allow scientists to observe drug behavior inside the body. This provides insights that cannot be obtained from lab studies alone. These techniques enhance Long Acting Injectables Characterization by linking lab data with real outcomes.
Emerging Techniques:
- MRI – Tracks depot formation and swelling
- PET – Monitors drug distribution in the body
- FRET – Studies drug release at molecular level
These methods improve IVIVC and help refine formulation strategies based on real biological data.
6. What Is the Role of Polymer Characterization in LAIs?
Polymer properties are the main drivers of drug release and stability. Understanding these properties is essential in Long Acting Injectables Characterization. Small changes in polymer composition can lead to large differences in performance.
Critical Polymer Attributes:
- Molecular weight
- Lactide:glycolide ratio
- Crystallinity
- Degradation rate
Analytical Techniques:
- GPC – Measures molecular weight
- XRD – Determines crystallinity
- Mass Spectrometry – Identifies degradation products
These tools ensure consistent quality and performance across batches.
Read our case study on Leuprolide Depot sustained release for real-world polymer performance data.
7. How Are In Vitro–In Vivo Correlations (IVIVC) Established?
IVIVC connects lab data with clinical performance. This relationship is essential for predicting how a drug will behave in patients. It is a key part of Long Acting Injectables Characterization.
Strategies:
- Level A IVIVC
- PBPK modeling
- Mechanistic modeling
These approaches combine different data types to improve prediction accuracy and reduce development risks.
Check out our specialized Leuprolide Depot generic drug development services for expert IVIVC support.
8. What Are the Emerging Trends in Long Acting Injectables Characterization?
New technologies are making Long Acting Injectables Characterization faster and more accurate. These innovations focus on prediction and efficiency.
Key Innovations:
- Molecular simulations
- AI/ML-based prediction models
- High-throughput screening
- Microfluidic systems
These tools allow faster development and better decision-making during formulation design.
Navigating the regulatory pathway for complex peptide injectables requires staying ahead of these trends.
Conclusion
Long Acting Injectables Characterization is becoming more advanced and data-driven. It combines imaging, spectroscopy, and modeling to fully understand complex drug delivery systems. This approach ensures both performance and regulatory compliance.
Linking microstructure, polymer properties, and release kinetics is essential for developing reliable products. Strong analytical strategies lead to consistent therapies and better patient outcomes. This is especially important for long-term treatments.
See how we address Leuprolide Depot analytical challenges to ensure superior product quality.
As regulatory expectations grow, the use of advanced and reliable techniques becomes even more important. These methods improve product quality and reproducibility. They also increase confidence in clinical performance.
Organizations that invest in modern characterization approaches can speed up development and meet market demands more effectively. This benefits both manufacturers and patients.
Compare your options: Peptide CDMO vs. CRO to determine the best fit for your next project.
👉 For expert support in advanced analytical characterization, connect here:
https://resolvemass.ca/contact/
FAQs on Long Acting Injectables Characterization
The most important factors include particle size, polymer molecular weight, drug loading, and release profile. These elements decide how the drug is released and how long it remains effective. Careful measurement of these parameters helps ensure consistent product quality. Advanced analytical tools are often required due to the complex nature of these systems.
Microstructure defines how the drug is arranged inside the formulation and how the polymer matrix is built. Features like pores and internal channels control how quickly or slowly the drug moves out. Even small structural differences can change the release pattern. This is why detailed imaging is essential during development.
Commonly used techniques include GPC for molecular weight, DSC for thermal behavior, and XRD for crystallinity. Each method provides unique information about the polymer properties. These insights help predict how the polymer will behave during drug release. Together, they support consistent formulation development.
Burst release is typically measured using in vitro testing with early time-point sampling. This helps capture the initial rapid release of the drug. Imaging tools like SEM can also be used to understand structural causes. Using multiple approaches improves the reliability of the results.
One major challenge is maintaining conditions that closely match the human body. It can also be difficult to keep results consistent across different tests. Complex release mechanisms add another layer of difficulty. Because of this, customized testing methods are often needed.
Spectroscopy helps identify chemical interactions between the drug and polymer. Techniques like FTIR and Raman provide detailed molecular information. They can detect compatibility issues and distribution patterns. This makes them valuable tools for ensuring formulation stability.
Polymer characteristics such as molecular weight, composition, and crystallinity directly affect release behavior. These properties control how the polymer breaks down and how the drug diffuses. Selecting the right polymer is critical for achieving the desired release profile. Small changes can lead to noticeable differences.
Reference:
- Alidori, S., et al. (2024). Patient-centric long-acting injectable and implantable drug delivery systems. Pharmaceutics. https://pmc.ncbi.nlm.nih.gov/articles/PMC11372838/
- U.S. Food and Drug Administration. (2015). FY2015 regulatory science research report: Long-acting injectable formulations. https://www.fda.gov/industry/generic-drug-user-fee-amendments/fy2015-regulatory-science-research-report-long-acting-injectable-formulations
- Bauer, A., Berben, P., Chakravarthi, S. S., Chattorraj, S., Garg, A., Gourdon, B., Heimbach, T., Huang, Y., Morrison, C., Mundhra, D., Palaparthy, R., Saha, P., Siemons, M., Shaik, N. A., Shi, Y., Shum, S., Thakral, N. K., Urva, S., Vargo, R., Koganti, V. R., & Barrett, S. E. (2023). Current state and opportunities with long-acting injectables: Industry perspectives from the Innovation and Quality Consortium “Long-Acting Injectables” working group. Pharmaceutical Research, 40(7), 1601–1631. https://doi.org/10.1007/s11095-022-03391-y
- U.S. Food and Drug Administration. (2019). Long-acting injectable and implantable drug products: Considerations for product development and regulatory approval (Draft guidance for industry). https://www.fda.gov/media/129010/download



