Introduction to Nitrosamine Lifecycle Management
Nitrosamine Lifecycle Management is essential for maintaining effective control of nitrosamine impurities after pharmaceutical products receive regulatory approval. Once a drug enters the commercial market, manufacturing conditions, suppliers, and packaging systems may change over time. These changes can influence chemical reactions and may create new pathways for nitrosamine formation.
Regulatory authorities expect pharmaceutical companies to continuously monitor and manage nitrosamine risks throughout the entire product lifecycle. Manufacturers must ensure that impurity levels remain within acceptable regulatory limits during commercial production and distribution.
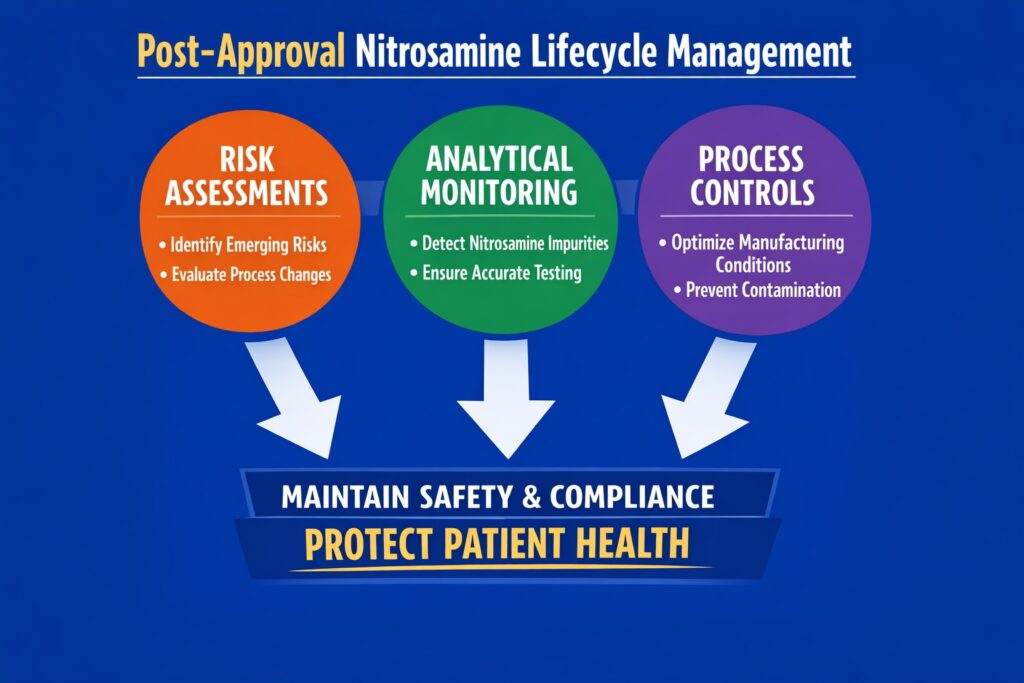
A strong lifecycle management strategy combines risk assessments, analytical monitoring, quality management systems, and regulatory compliance processes. Together, these systems help pharmaceutical companies maintain product safety, ensure regulatory compliance, and control nitrosamine risks during long-term commercial manufacturing.
Explore our comprehensive services for pharmaceutical impurity control: Nitrosamine Analysis and Testing Services
Share via:
Summary of Key Insights
- Nitrosamine Lifecycle Management after product approval requires continuous monitoring, analytical verification, and risk reassessment throughout the product’s commercial lifecycle.
- Regulatory agencies such as FDA, EMA, and Health Canada require manufacturers to maintain ongoing risk evaluation and control strategies for nitrosamine impurities even after market authorization.
- Post-approval lifecycle management includes periodic risk assessments, control strategy updates, supplier qualification, and process monitoring.
- Analytical lifecycle management ensures validated, sensitive testing methods remain suitable as processes, raw materials, or manufacturing conditions change.
- Companies must implement change control frameworks to manage formulation changes, process modifications, and packaging variations that may affect nitrosamine formation.
- Regulatory reporting and documentation are essential to demonstrate continuous compliance and product safety.
- Advanced testing technologies such as LC-MS/MS and high-resolution mass spectrometry are increasingly used to support long-term monitoring programs.
- A structured Nitrosamine Lifecycle Management program integrates quality systems, risk management, and analytical surveillance to maintain product safety across the product lifecycle.
Why Nitrosamine Lifecycle Management Is Critical After Product Approval
Nitrosamine Lifecycle Management ensures that impurity control strategies remain effective throughout the commercial manufacturing and distribution stages of pharmaceutical products. While extensive testing is conducted during drug development, additional risks can appear once production moves to a larger commercial scale.
Post-approval manufacturing environments are often more complex than development settings. Pharmaceutical companies may operate multiple production sites, use global suppliers, and manufacture larger batch volumes. These operational changes can influence chemical reactions and create new pathways for nitrosamine formation.
Several factors make lifecycle monitoring essential:
- Process optimization during commercial scale manufacturing
- Supplier changes or raw material variability
- Packaging interactions
- Storage conditions affecting degradation pathways
- New regulatory limits or toxicological findings
Research has shown that several nitrosamine contamination incidents occurred after process adjustments were made during commercial production. One widely known example involved angiotensin receptor blockers, where contamination appeared after manufacturing changes in active pharmaceutical ingredient (API) synthesis.
These cases highlight the importance of maintaining strong lifecycle oversight. Even small changes in production conditions may lead to unexpected impurity formation if proper monitoring systems are not in place.
Learn more about managing risks for specific pharmaceutical categories: Nitrosamine Testing for High-Risk Drug Classes
Post-Approval Risk Drivers
| Risk Factor | Potential Impact on Nitrosamine Formation |
|---|---|
| API synthesis changes | Introduction of nitrosating agents |
| Excipient interactions | Secondary amine reactions |
| Solvent reuse | Accumulation of nitrosating intermediates |
| Packaging components | Nitrite migration |
| Storage conditions | Degradation-induced nitrosamine formation |
Lifecycle management frameworks allow manufacturers to detect, investigate, and mitigate these risks early. Identifying issues at an early stage helps companies maintain regulatory compliance while protecting patient safety.
Post-Approval Nitrosamine Lifecycle Management Framework
A structured Nitrosamine Lifecycle Management framework integrates risk evaluation, monitoring systems, and pharmaceutical quality processes to maintain control strategies during commercial production. This systematic approach ensures that impurity risks are continuously evaluated throughout the product lifecycle.
Regulatory guidance from agencies such as FDA, EMA, and ICH M7 emphasizes the importance of lifecycle-based impurity management. These guidelines encourage pharmaceutical manufacturers to maintain active oversight even after market authorization has been granted.
A comprehensive framework ensures that all operational activities are monitored for potential nitrosamine risks. It also provides clear procedures for evaluating manufacturing changes and implementing corrective actions when required.
Stay updated on the latest regulatory changes affecting risk assessments: Impact of ICH M7(R2) Updates on Nitrosamine Risk Assessment
Core Components of Nitrosamine Lifecycle Management
- Periodic risk assessment
- Analytical monitoring programs
- Manufacturing process control
- Change management procedures
- Regulatory reporting and documentation
- Supplier and raw material oversight
Each of these components supports continuous risk evaluation. Together, they form a strong control system that maintains product quality and regulatory compliance throughout the product lifecycle.
Continuous Risk Assessment in Nitrosamine Lifecycle Management
Continuous risk assessment is one of the most important elements of Nitrosamine Lifecycle Management. It helps identify new pathways for nitrosamine formation that may appear during manufacturing, distribution, or long-term product storage.
Post-approval risk evaluations must consider several operational factors. These include manufacturing deviations, process condition changes, raw material variability, and stability data generated during shelf-life studies. Each of these factors can influence impurity formation.
Companies should also monitor market complaints and new toxicological research findings that may indicate emerging impurity risks. Including new scientific insights in the risk evaluation process ensures that control strategies remain updated and effective.
Understand the complexities of NDSRIs and the CPCA framework: Nitrosamine CPCA Approach for NDSRIs
Key Elements of Ongoing Risk Evaluation
- Review of API synthesis routes
- Evaluation of amine and nitrosating agent presence
- Assessment of degradation pathways
- Packaging interaction studies
- Monitoring nitrite levels in excipients
Risk assessments should be conducted:
- After any process change
- During annual product quality reviews
- When new regulatory guidance becomes available
A structured risk-based framework helps pharmaceutical companies identify emerging risks early and implement effective mitigation strategies.
Analytical Lifecycle Management for Nitrosamine Monitoring
Analytical lifecycle management ensures that testing methods remain reliable, sensitive, and compliant throughout the product lifecycle. Because nitrosamine limits are extremely low, laboratories must use highly sensitive analytical techniques capable of detecting trace concentrations.
Many regulatory limits for nitrosamines are measured in nanograms per day, which requires advanced analytical instruments. Testing methods must also be robust enough to produce consistent results across different manufacturing batches and laboratory environments.
Discover advanced techniques for trace-level impurity detection: High-Resolution Mass Spectrometry (HRMS) for Nitrosamine Testing
Common Analytical Methods
| Technique | Purpose |
|---|---|
| LC-MS/MS | Highly sensitive quantification |
| GC-MS | Detection of volatile nitrosamines |
| HRMS | Structural confirmation |
| SFC-MS | Multi-nitrosamine screening |
Analytical lifecycle management includes several ongoing activities:
- Updating method validation
- Cross-laboratory method transfer
- Continuous monitoring of method performance
- Re-validation after process modifications
Modern analytical strategies increasingly include universal nitrosamine detection workflows and phase-appropriate validation approaches. These systems help ensure consistent monitoring across many pharmaceutical products and formulations.
Manufacturing Process Controls in Nitrosamine Lifecycle Management
Manufacturing process controls are essential for minimizing nitrosamine formation during large-scale pharmaceutical production. As production volumes increase, operational complexity also grows, which makes strong process control measures even more important.
Commercial manufacturing environments may include solvent recycling systems, multi-product facilities, and process optimization initiatives. While these changes improve efficiency, they may also create conditions that allow impurities to form if controls are not properly maintained.
Process Control Strategies
- Control of nitrite levels in raw materials
- Avoidance of secondary amines and nitrosating agents
- Optimized reaction conditions such as pH and temperature
- Controlled solvent recovery processes
- Dedicated cleaning protocols to prevent cross-contamination
These strategies help maintain stable manufacturing conditions and reduce the possibility of nitrosamine formation during production.
Learn how to optimize your manufacturing through targeted mitigation:Nitrosamine Mitigation via Solvents and Catalysts
Example Process Risk Matrix
| Manufacturing Step | Risk Level | Control Measure |
|---|---|---|
| API synthesis | High | Nitrite monitoring |
| Solvent reuse | Medium | Purification testing |
| Formulation stage | Medium | Excipient qualification |
| Packaging | Low-Medium | Nitrite migration testing |
Strong process control systems support effective Nitrosamine Lifecycle Management and ensure consistent impurity control during commercial manufacturing.
Change Management and Post-Approval Variations
Change management systems are essential for evaluating any modification that could influence nitrosamine formation. Pharmaceutical companies often introduce process improvements after product approval, but each change must be carefully reviewed before implementation.
Regulatory agencies require manufacturers to perform detailed risk assessments for post-approval changes. Even minor modifications may alter chemical reaction pathways and create new opportunities for nitrosamine formation.
Common Post-Approval Changes Requiring Evaluation
- API manufacturing route changes
- New raw material suppliers
- Process parameter adjustments
- Excipient substitutions
- Packaging modifications
- Manufacturing site transfers
Each proposed change should undergo:
- Risk assessment
- Analytical verification
- Regulatory impact evaluation
This structured approach ensures that process improvements do not unintentionally introduce new impurity risks.
Ensure your submissions meet regulatory standards during process changes: Nitrosamine Risk Assessment for ANDA Submission
Regulatory Expectations for Nitrosamine Lifecycle Management
Global regulatory authorities expect pharmaceutical manufacturers to maintain documented Nitrosamine Lifecycle Management strategies for all marketed products. These expectations reflect the increasing focus on mutagenic impurity control within pharmaceutical regulation.
Regulators require companies to demonstrate that they have implemented risk assessments, confirmatory testing programs, and monitoring systems for nitrosamine impurities in marketed medicines.
Key Regulatory Expectations
- Completion of risk assessments for all marketed products
- Implementation of confirmatory testing programs
- Submission of post-approval variations when required
- Maintenance of validated analytical testing methods
Major Regulatory Agencies and Guidance
| Agency | Guidance Focus |
|---|---|
| US FDA | Risk evaluation and confirmatory testing |
| EMA | Lifecycle risk management for nitrosamines |
| Health Canada | Post-authorization monitoring |
| ICH | Mutagenic impurity risk management |
Regulatory authorities emphasize that Nitrosamine Lifecycle Management should be treated as a continuous process rather than a one-time evaluation conducted during development.
Compare international safety thresholds and limits: Nitrosamine Acceptable Intake (AI) Limits Comparison
Documentation and Quality System Integration
Lifecycle management of nitrosamine control strategies must be fully integrated into the pharmaceutical quality management system. Proper documentation helps demonstrate that manufacturers are actively managing impurity risks and maintaining regulatory compliance.
Maintaining detailed records also allows companies to track manufacturing changes, analytical testing activities, and risk assessment updates that may influence nitrosamine formation.
Key Documentation Elements
- Risk assessment reports
- Analytical method validation records
- Change control documentation
- Supplier qualification records
- Periodic product quality reviews
Quality systems should integrate:
- ICH Q9 risk management principles
- ICH Q10 pharmaceutical quality systems
- Continuous improvement frameworks
These systems ensure that nitrosamine control strategies remain effective throughout the product lifecycle and that regulatory inspections can easily verify compliance.
Learn how stability data impacts your lifecycle documentation: Nitrosamine Testing in Stability Studies
Emerging Trends in Nitrosamine Lifecycle Management
Scientific advancements and new technologies are shaping the future of Nitrosamine Lifecycle Management. As regulatory expectations increase, pharmaceutical companies are adopting innovative tools to detect and prevent impurity formation more effectively.
Advanced analytical technologies and digital quality systems are helping manufacturers improve monitoring efficiency and risk prediction accuracy. These solutions allow earlier identification of potential nitrosamine formation pathways.
Emerging Approaches
- Predictive risk modeling for nitrosamine formation
- Advanced mass spectrometry workflows
- Digital quality management systems
- AI-assisted impurity prediction
These developments support more proactive lifecycle management strategies and allow pharmaceutical companies to respond quickly to emerging risks.
Conclusion
Effective Nitrosamine Lifecycle Management ensures that impurity control strategies established during drug development remain effective throughout commercial production and distribution. Even after regulatory approval, several variables can influence nitrosamine formation.
Post-approval stages often include manufacturing optimization, supplier changes, and packaging modifications. These factors may introduce new impurity risks if they are not properly evaluated and monitored.
By combining continuous risk assessments, analytical lifecycle management, manufacturing process controls, change management systems, and regulatory compliance strategies, pharmaceutical companies can maintain safe nitrosamine levels and protect patient health.
As regulatory expectations continue to evolve, companies must adopt proactive and scientifically robust lifecycle management programs. A strong Nitrosamine Lifecycle Management strategy helps maintain product quality, ensure compliance, and safeguard patient safety.
Frequently Asked Questions (FAQs)
Nitrosamine Lifecycle Management after drug approval involves continuous monitoring, periodic risk assessments, and regular updates to impurity control strategies. Pharmaceutical companies must verify that nitrosamine levels remain within regulatory limits during commercial production. This also includes maintaining validated analytical methods and documenting all risk management activities.
Major regulatory agencies including the US FDA, EMA, Health Canada, and ICH require pharmaceutical companies to maintain ongoing monitoring programs for nitrosamine impurities. These agencies emphasize lifecycle risk management and expect manufacturers to continuously evaluate and control impurity risks.
Risk assessments should be performed regularly throughout the product lifecycle. They are commonly conducted during annual product quality reviews, after manufacturing or formulation changes, and whenever new regulatory guidance becomes available. Regular reviews help ensure impurity risks remain controlled.
Change control systems ensure that any modification in manufacturing processes, raw materials, or packaging is evaluated before implementation. This evaluation helps determine whether the change could create conditions that lead to nitrosamine formation. Proper change management prevents unexpected impurity risks.
Companies maintain compliance by performing continuous risk assessments, implementing analytical testing programs, and maintaining strong documentation systems. They also manage suppliers carefully and submit regulatory updates whenever required. These actions ensure that nitrosamine risks remain controlled throughout the product lifecycle.
New technologies such as advanced analytics, predictive modeling, AI-based risk assessment, and digital quality systems are improving impurity monitoring. These innovations allow pharmaceutical companies to detect potential risks earlier and strengthen lifecycle control strategies.
Reference:
- Tarafder, A., Vega, E., Beck, H. P., Mundal, D., Tilala, M., & Wang, S. (2025). Nitrosamine control: From risk assessment to analytical testing with emphasis on sample preparation and phase-appropriate method validation. Organic Process Research & Development, 29(11). https://doi.org/10.1021/acs.oprd.5c00158
- Manchuri, K. M., & colleagues. (2024). Analytical methodologies to detect N-nitrosamine impurities in pharmaceutical products: A comprehensive review. ACS Omega. https://pmc.ncbi.nlm.nih.gov/articles/PMC12135945/
- U.S. Food and Drug Administration. (2024). Control of nitrosamine impurities in human drugs: Guidance for industry. https://www.fda.gov/media/141720/download
- Badia, L., Camps, H., Lloveras, I., Handler, N., & others. (2024). Strategy to assess and control nitrosamine formation in API during storage: A sitagliptin case study. Organic Process Research & Development, 28(1). https://doi.org/10.1021/acs.oprd.4c00088



