Introduction: Why Nitrosamine Testing for Ranitidine Demands Special Attention
Nitrosamine Testing for Ranitidine is one of the most complex tasks in pharmaceutical impurity evaluation. The challenge exists because ranitidine itself can form NDMA due to its chemical structure, even without contamination from manufacturing or raw materials. Unlike many APIs where nitrosamines come from solvents, reagents, or process conditions, ranitidine contains built-in functional groups that can react and form nitrosamines. This makes traditional risk assessments incomplete and requires a molecule-specific evaluation strategy. Storage temperature, humidity, and formulation environment can also increase the chances of internal NDMA formation over time.
The global recall of ranitidine products between 2019 and 2020 by FDA, EMA, and Health Canada highlighted major gaps in structure-based risk evaluation. Many early assessments focused only on manufacturing risks and ignored the intrinsic instability of ranitidine. This resulted in unexpected NDMA levels during stability testing and storage. Regulatory agencies later emphasized lifecycle risk evaluation from API synthesis to finished product storage. Nitrosamine Testing for Ranitidine now requires a comprehensive approach covering structure, synthesis, formulation, packaging, and analytical testing. This article provides a complete overview of ranitidine nitrosamine risk, including API synthesis, degradation pathways, finished products, and analytical method considerations.
Learn More: Discover our comprehensive Nitrosamine Analysis Services and how we address complex molecular challenges.
Share via:
📋 Quick Summary — What This Article Covers
- Ranitidine has a unique molecular structure with a secondary amine (–NH–) and a dimethylaminomethyl tertiary amine. Both groups can undergo nitrosation reactions. This increases the risk of NDMA and NDSRI formation. Because of this, ranitidine is considered a high-risk API for nitrosamine impurities.
- The API synthesis process introduces multiple nitrosamine precursors. These include dimethylamine, thioethylamine intermediates, and nitrovinyl-containing reagents. Each step may create conditions for nitrosamine formation. Careful control is required during manufacturing.
- Finished-product manufacturing adds additional risk. Excipients such as microcrystalline cellulose and corn starch may contain nitrite impurities. Moisture exposure can accelerate degradation. Ranitidine’s thermal instability may also generate NDMA during storage.
- Regulatory agencies including FDA, EMA, and Health Canada identified NDMA as the main impurity. Other impurities include N-nitroso ranitidine and dimethylamine-derived nitrosamines. These must be considered during risk assessment. Comprehensive testing is therefore required.
- Validated analytical techniques include LC-MS/MS, GC-MS/MS, and GC-HRMS. These methods provide sensitive nitrosamine detection. Current guidance follows ICH M7(R2). The acceptable intake limit for NDMA is 96 ng per day.
- ResolveMass Laboratories provides complete Nitrosamine Testing for Ranitidine. Services include risk assessment, method development, and validated testing. Both API and finished dosage forms are supported. Regulatory documentation assistance is also available.
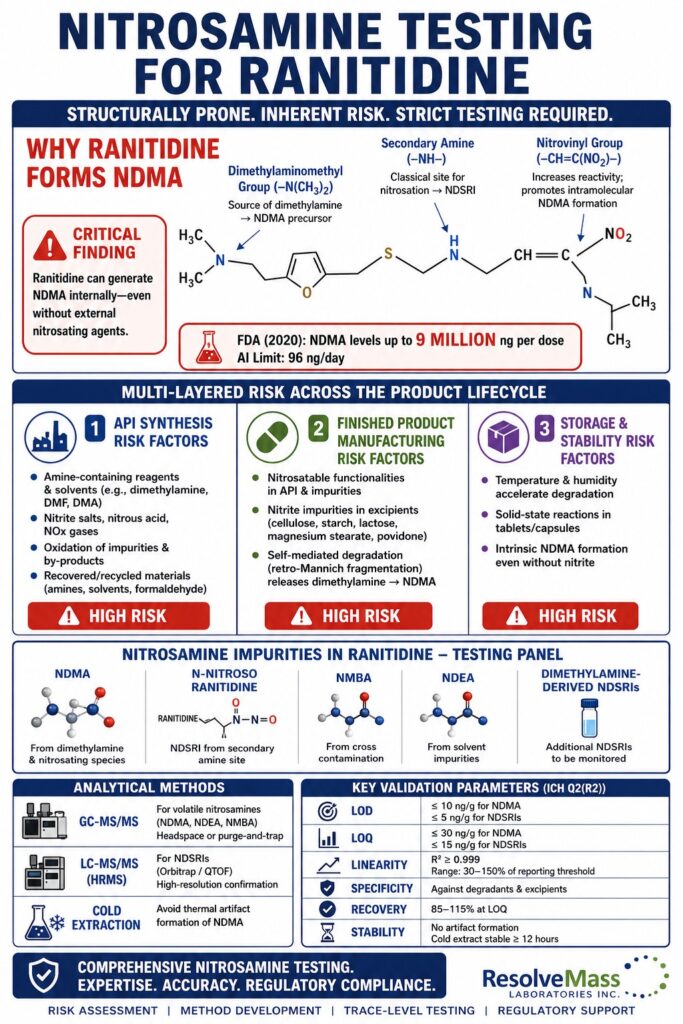
Structural Analysis of Ranitidine: Identifying Nitrosatable Functionalities
The molecular structure of ranitidine plays a central role in nitrosamine formation risk. The compound contains multiple nitrogen-containing groups capable of undergoing nitrosation reactions. These groups may react under acidic, thermal, or storage-related stress conditions. Their proximity to electron-withdrawing groups increases chemical instability and promotes degradation. Both intermolecular and intramolecular pathways can form NDMA. Because of this, ranitidine is considered structurally prone to nitrosamine formation compared to many other APIs.
Dimethylaminomethyl Group (–N(CH₃)₂)
This tertiary amine is attached to the furan ring through a methylene bridge. Under nitrosating conditions, partial demethylation may produce a secondary amine intermediate. This intermediate can directly convert to NDMA in the presence of nitrosating species. The reaction may occur during storage, manufacturing, or stability testing. The formation of dimethylamine significantly increases NDMA risk. This group is also sensitive to oxidation and hydrolysis.
Secondary Amine (–NH–) in the Ethylamine Side Chain
This secondary amine is located between the thioether chain and the nitrovinyl group. It represents a classical nitrosatable functionality that reacts easily with nitrite impurities. Even trace nitrite levels may trigger nitrosamine formation. Solid dosage form microenvironments may also support this reaction. Mildly acidic conditions in tablets can increase reactivity. This site is therefore critical during finished product risk assessment.
Nitrovinyl Group (–CH=C(NO₂)–)
The nitro group does not directly cause nitrosation, but it increases reactivity of nearby amines. Its electron-withdrawing effect promotes molecular fragmentation under stress conditions. This allows intramolecular reactions that can generate NDMA without external nitrosating agents. Such self-contained reactivity is unusual and explains ranitidine’s instability. This behavior is often observed during long-term stability studies. It is a major reason for strict Nitrosamine Testing for Ranitidine requirements.
⚠️ Critical Finding: FDA laboratory investigations in 2020 demonstrated that ranitidine can generate NDMA at extremely high levels under simulated storage and gastrointestinal conditions. Some results exceeded 9 million ng per dose, far above the acceptable intake limit of 96 ng/day. These findings confirmed that NDMA formation is mainly due to intrinsic structural instability. Temperature, humidity, and storage duration significantly affect impurity levels. These observations ultimately led to global product withdrawals.
Technical Insight: Explore the Impact of ICH M7(R2) Updates on Nitrosamine Risk Assessment to understand evolving regulatory expectations.
Nitrosamine Risk Evaluation: Ranitidine API Synthesis Process
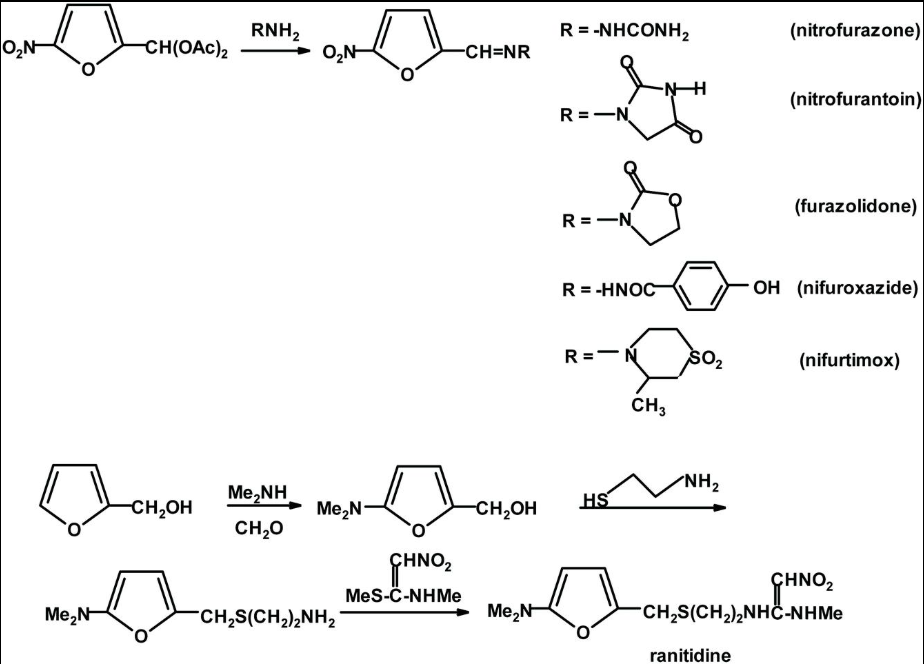
A stepwise review of ranitidine synthesis shows multiple stages where nitrosamine formation may occur. The synthesis generally includes three main steps: Mannich aminomethylation of furfuryl alcohol, thioalkylation with cysteamine, and coupling with the nitrovinyl methylamino reagent. Each step introduces amines, reactive intermediates, or conditions that support nitrosation. Residual reagents carried forward between steps can increase risk. Process temperature, solvent reuse, and pH also influence impurity formation. Therefore, Nitrosamine Testing for Ranitidine must include a full process risk evaluation.
Risk Factor 1 — Amine-Containing Starting Materials, Reagents, and Solvents
HIGH RISK
Dimethylamine (Me₂NH) is used in Step 1 during the Mannich reaction. This reagent introduces the dimethylaminomethyl group onto the furan ring. Dimethylamine is a direct precursor for NDMA formation. Residual dimethylamine carried into later steps may react with nitrite impurities. Even trace contamination can produce measurable NDMA. Strict residual control is therefore required.
Cysteamine (HSCH₂CH₂NH₂) is used in Step 2 to introduce the aminoethylthiomethyl side chain. Although primary amines usually form unstable nitrosamides, incomplete reactions may leave reactive intermediates. These intermediates can degrade into nitrosamines under certain conditions. Over-nitrosation may also produce nitrogen-containing by-products. Process optimization is necessary to minimize this risk. Monitoring of residual amines is recommended.
Methylamine residues from coupling reagents also contribute to risk. These residues may remain in recycled solvent systems. If nitrite is present, NDMA formation may occur. Solvent recovery systems must therefore be carefully evaluated. Routine monitoring helps prevent impurity buildup. Dedicated solvent handling reduces cross contamination.
Polar aprotic solvents such as DMF, DMA, and NMP may degrade into dimethylamine. This degradation increases under acidic or high-temperature conditions. Released dimethylamine can directly form NDMA. This pathway is well documented in pharmaceutical manufacturing. Long storage times further increase the risk. Solvent quality control is essential.
Quaternary ammonium catalysts may also degrade and release tertiary amines. These amines can participate in nitrosation reactions. Degradation may occur during heating or hydrolysis. Catalyst selection therefore impacts nitrosamine risk. Impurity monitoring should include catalyst-derived amines.
🔍 Key Assessment Point: Recovered dimethylamine and recycled solvents should always be considered potential NDMA sources. Nitrite testing is recommended before reuse. Recovered DMF or DMA should also be evaluated for secondary amines. Standard purification may not remove all impurities. Routine monitoring reduces nitrosamine formation risk.
Expert Guide: See our specialized approach for Nitrosamine Testing for High-Risk Drug Classes.
Risk Factor 2 — Nitrite Salts, Esters, and Other Nitrosating Agents
HIGH RISK
The nitrovinyl reagent used in final coupling increases reactivity of nearby amines. Although not a direct nitrosating agent, it promotes nitrosation under suitable conditions. Residual amines further increase this risk. Careful control of nitrosating sources is therefore necessary. Raw material quality plays an important role. Impurity monitoring should include nitrite content.
Sodium nitrite contamination can create a direct nitrosamine pathway. Nitrite may originate from water systems, reagents, or buffers. When combined with dimethylamine, NDMA can form rapidly. Even low nitrite levels may generate detectable impurities. Raw material testing is essential. Utility systems should also be monitored.
Nitrous acid may form under acidic aqueous conditions. Trace nitrite contamination can generate this species in situ. Nitrous acid reacts easily with secondary amines. Reaction rates increase at lower pH values. Such conditions may occur during washing or isolation steps. Process controls should minimize this risk.
Nitrogen oxide gases (NOx) may also act as nitrosating agents. These gases can enter reactors through ventilation systems. In the presence of moisture and amines, nitrosation reactions may occur. Open reactor systems increase exposure. Gas purity should be evaluated. Reactor sealing helps reduce risk.
Risk Factor 3 — Oxidation of Hydrazine, Hydrazides, and Related Intermediates
MODERATE RISK
Ranitidine synthesis does not intentionally use hydrazine intermediates. However, nitroaminoguanidine-type impurities may form as by-products. These species are structurally related to hydrazones. Under oxidative conditions, they may degrade into reactive nitrogen compounds. These compounds can contribute to nitrosamine formation. Such reactions may occur during cleaning or stress testing.
Hydrogen peroxide, hypochlorite sanitizers, and atmospheric oxygen may drive oxidation. These conditions can generate reactive nitroso species. Although secondary, this pathway remains relevant in multi-product facilities. Cross contamination from other processes may increase risk. Cleaning validation is therefore important. Segregation practices should be followed.
Risk Factor 4 — Contaminated, Recovered, or Recycled Materials
HIGH RISK
Recovered dimethylamine and recycled solvents are major contamination sources. Distillation may concentrate nitrosamine impurities. Reuse of contaminated streams can increase NDMA levels. Continuous solvent recovery systems present higher risk. Monitoring of recovered fractions is essential. Dedicated solvent handling reduces contamination.
Recovered DMF and DMA may contain residual dimethylamine. Incomplete purification allows these impurities to persist. Subsequent reuse introduces NDMA precursors. Risk increases when solvents are shared across products. Separate solvent systems may help reduce exposure. Regular testing is recommended.
Formaldehyde used in the Mannich reaction may introduce nitrite impurities. Contaminated storage containers may be the source. Reaction with nitrite generates nitrosating species. These species can react with amines. Storage conditions must therefore be controlled. Raw material qualification is necessary.
Acidic aqueous streams from thioalkylation should also be monitored. Residual nitrite may remain in these streams. Reuse without testing may introduce contamination. Proper disposal and monitoring are required. These controls help prevent unintended nitrosamine formation.
Nitrosamine Risk Evaluation: Ranitidine Finished Product Manufacturing
Even when API meets specifications, finished product manufacturing introduces additional risks. Excipients may contain nitrite impurities. Moisture and temperature accelerate reactions in solid dosage forms. Packaging materials may influence stability. Solid-state reactions can occur during shelf life. Therefore, Nitrosamine Testing for Ranitidine must include finished product evaluation.
Risk Factor 5 — Nitrosatable Functionalities in the API and Its Impurities
HIGH RISK
Ranitidine’s secondary amine is the primary nitrosatable site. Trace nitrosating agents may react with this group. This reaction forms N-nitroso ranitidine (NDSRI). Even minimal nitrite levels can initiate conversion. Solid-state reactions may occur during storage. Monitoring is essential throughout shelf life.
Ranitidine sulfoxide retains the same reactive amine. Oxidative degradation increases its concentration. This increases nitrosamine risk over time. Storage conditions influence this behavior. Stability testing should include oxidative impurities. Monitoring helps control risk.
Ranitidine N-oxide may revert to dimethylamine. This creates additional NDMA precursor capacity. Tablet matrix interactions may influence this reaction. Microenvironmental pH also affects conversion. This creates additional complexity. Shelf-life studies must evaluate this pathway.
Ranitidine desmethyl impurity contains a secondary methylamine. This group is highly reactive toward nitrosation. Trace nitrite may produce nitrosamines. Process-related impurities also contribute. These species increase overall reactive nitrogen content. Combined effects elevate risk.
Risk Factor 6 — Nitrite Content in Excipients
HIGH RISK
Common excipients used in tablets and capsules may contain nitrite impurities. These impurities can promote nitrosation of ranitidine. Solid-state reactions may occur even at low moisture levels. Storage conditions further influence the reaction. Excipient qualification is therefore critical.
| Excipient | Nitrite Source / Mechanism | Risk Level | Recommended Action |
|---|---|---|---|
| Microcrystalline Cellulose (MCC) | Nitrate/nitrite impurities from acid hydrolysis of cellulose; residual mineral acid contamination | HIGH | Vendor qualification + lot-by-lot nitrite testing (<0.1 ppm) |
| Corn Starch / Maize Starch | Nitrate-reducing microbial activity during storage; soil-derived nitrate residues | HIGH | Storage condition controls; nitrite specification in CoA |
| Lactose Monohydrate | Reducing sugar-mediated Maillard byproducts can generate reactive nitrogen species at elevated temperature/humidity | MEDIUM | Avoid lactose in ranitidine FDF where possible; use controlled humidity storage |
| Magnesium Stearate | Nitrite contamination from stearate salt impurities; variable by source | MEDIUM | Vendor and grade qualification; preference for pharmaceutical-grade sources |
| Povidone (PVP) | Trace peroxide impurities can oxidize nitrogen-containing impurities to NOₓ species | MEDIUM | Peroxide specification <400 ppm; avoid PVP with ranitidine in high-humidity packaging |
| Colloidal Silicon Dioxide | NOₓ adsorption and surface-catalyzed release under acidic micro-environments | MEDIUM | Evaluate grade; consider alternative glidants |
Mitigation Strategy: Read about using a Secondary Amine Scavenger for Nitrosamine Mitigation to protect your formulation.
Risk Factor 7 — Degradation of Ranitidine by Inherent Reactivity
HIGH RISK
The most critical risk factor is self-mediated NDMA generation. Ranitidine undergoes retro-Mannich fragmentation during storage. This reaction releases dimethylamine. The released amine can form NDMA. This process may occur without external nitrosating agents. This makes ranitidine unique.
Thermal stress accelerates cleavage of the dimethylaminomethyl fragment. This releases dimethylamine. Acidic hydrolysis produces nitrosating species. These species react with dimethylamine. This intramolecular pathway explains spontaneous NDMA formation. Temperature significantly affects reaction rate.
This reaction is independent of excipients. Even controlled formulations may generate NDMA. Elevated storage conditions worsen impurity levels. Stability studies must evaluate temperature impact. Long-term testing is required.
NDMA generation increases with temperature. Studies at high temperatures show significant levels. Accelerated stability conditions may produce measurable NDMA. This places ranitidine in a high-risk category. Careful regulatory evaluation is required.
🧪 Implication for Reformulation Efforts: Reformulation must demonstrate inhibition of structural fragmentation. Absence of nitrite alone is not sufficient. Storage and in-use conditions must be evaluated. Full shelf-life stability data is required. Comprehensive Nitrosamine Testing for Ranitidine is essential.
Case Study: Review our NDMA Root Cause Investigation Case Study for practical insights into identifying contamination sources.
Published NDSRIs for Ranitidine: Regulatory Guidance Landscape
Regulatory authorities have identified multiple nitrosamine impurities in ranitidine. These impurities define the minimum testing panel. Acceptable intake limits are established for each compound. Testing must cover both volatile nitrosamines and NDSRIs. Regulatory expectations require comprehensive coverage.
| Nitrosamine Impurity | Structure / Origin | AI Limit | Regulatory Reference |
|---|---|---|---|
| NDMA (N-Nitrosodimethylamine) | Dimethylamine + nitrous acid / self-fragmentation of ranitidine | 96 ng/day | FDA (2020), EMA (EMA/428592/2020), Health Canada |
| N-Nitroso Ranitidine (NDSRI) | Nitrosation at the secondary amine of the ranitidine side chain | ICH M7(R2) TTC-based (≤18 ng/day for <10 yr treatment) | EMA Q&A on Ranitidine (2020); Health Canada Ranitidine Review |
| NMBA (N-Nitrosomethylbenzylamine) | Cross-contamination from benzylamine-based intermediates in multi-product facilities | 96 ng/day | FDA Nitrosamine Guidance 2023 (facility-level risk) |
| NDEA (N-Nitrosodiethylamine) | Diethylamine impurities in recovered solvents (ethanol, ethyl acetate) + nitrite | 26.5 ng/day | ICH M7(R2); EMA/FDA cohort of concern |
| Dimethylamine-derived NDSRI | Residual dimethylamine from Mannich step carried into finished product | 96 ng/day (as NDMA equivalent) | FDA Laboratory Alert (Sep 2019); Health Canada Product Recall Guidance |
Compliance Tip: Understand the new Nitrosamine CPCA Approach for NDSRIs to determine acceptable intake limits.
Nitrosamine Testing for Ranitidine: Analytical Method Considerations
Nitrosamine Testing for Ranitidine requires modified analytical methods. Ranitidine can generate NDMA during sample preparation. Artifact formation must be avoided. Both volatile nitrosamines and NDSRIs must be detected. Matrix complexity requires selective techniques.
GC-MS/MS is preferred for volatile nitrosamines such as NDMA and NDEA. Headspace or purge-and-trap improves sensitivity. Extraction conditions should avoid heating. Cold extraction reduces artifact formation. Method validation is required.
LC-MS/MS with HRMS is required for NDSRIs. High-resolution instruments provide structural confirmation. Orbitrap and QTOF systems are commonly used. These methods detect unknown nitrosamines. This capability is important for ranitidine.
LC-UV may be used for screening. However, specificity is limited. Matrix interference may occur. Confirmatory testing is necessary. LC-UV should not be used alone.
Cold extraction sample preparation is recommended. Thermal extraction may generate NDMA artificially. Early studies showed false positives. Validated cold methods prevent this issue. Sample stability must be demonstrated.
Methodology: Learn about GC-MS Method Development for Nitrosamine Testing
and HRMS for Nitrosamine Testing.
Key Validation Parameters per ICH Q2(R2)
Limit of Detection (LOD) should be ≤10 ng/g for NDMA and ≤5 ng/g for NDSRIs. These limits ensure trace detection. Sensitive instrumentation is required. Validation must confirm performance. Low-level detection is critical.
Limit of Quantitation (LOQ) should be ≤30 ng/g for NDMA and ≤15 ng/g for NDSRIs. Accurate quantitation at low levels is necessary. Calibration curves must be validated. Precision studies should be performed. Reporting thresholds must be defined.
Linearity should demonstrate R² ≥ 0.999. The range should cover 30–150% of reporting threshold. This ensures reliable quantitation. Multiple calibration points are recommended. System suitability should be verified.
Specificity must be demonstrated against degradants. Recovery should be 85–115% at LOQ. Artifact generation studies must confirm stability. Cold extraction stability should be shown for at least 12 hours. These parameters support regulatory acceptance.
Advanced Capability: See how we achieve Ultra-Low Limit of Quantitation (LOQ) in Nitrosamine Testing for sensitive applications.
Conclusion: A Multi-Layered Risk Profile Demanding Expert Testing
Nitrosamine Testing for Ranitidine is not a routine compliance activity. It requires expertise in synthesis chemistry, analytical science, and regulatory guidance. The risk profile is structurally driven and multi-layered. Conventional mitigation strategies are often insufficient. Integrated evaluation is necessary across the product lifecycle.
Ranitidine differs from other recalled APIs because its risk is inherent. Structural fragmentation generates NDMA internally. API synthesis introduces additional amine sources. Formulation excipients further increase risk. Storage conditions amplify degradation pathways. Comprehensive assessment is required.
A complete risk evaluation must consider structural reactivity, synthesis inputs, nitrosating agents, recycled materials, API impurities, excipient nitrite levels, and degradation pathways. Analytical testing must avoid artifact formation. Both NDMA and NDSRIs must be included. Regulatory guidance from FDA, EMA, and Health Canada should be followed.
ResolveMass Laboratories Inc. provides scientifically robust and GMP-compliant Nitrosamine Testing for Ranitidine API and finished products. The laboratory supports risk assessment, method development, and routine testing. Services include trace-level detection and regulatory documentation. Comprehensive testing helps ensure compliance with current nitrosamine guidance.
Partner with Experts: Learn how to optimize your Nitrosamine Testing CRO Selection to ensure high-quality data and regulatory success.
FAQs: Nitrosamine Testing for Ranitidine
Ranitidine was recalled because its molecular structure can generate NDMA on its own. The drug contains a dimethylaminomethyl group positioned near a nitro-related functionality, which allows internal reactions that release dimethylamine and promote nitrosamine formation. Other H2 blockers such as famotidine and cimetidine do not have this structural combination. Without this reactive arrangement, they are less likely to form NDMA through intrinsic degradation. For this reason, the recall was specific to ranitidine and not applied to the entire H2 blocker class.
Regulatory agencies including FDA, EMA, and Health Canada follow an acceptable intake limit of 96 ng per day for NDMA. This limit is based on long-term exposure calculations and a conservative cancer risk model. During investigations, several ranitidine products showed NDMA levels far above this threshold. In some cases, results were hundreds or thousands of times higher than the permitted level. These findings led regulators to remove ranitidine products from the market instead of allowing continued use with labeling changes.
Various reformulation strategies have been explored to reduce NDMA formation in ranitidine products. These include alternative salt forms, antioxidant addition, cyclodextrin complexation, and modified dosage forms. While some approaches lowered NDMA levels, none consistently prevented formation under all storage or use conditions. The core issue remains the inherent instability of the ranitidine structure itself. Because of this, regulatory agencies have not approved any reformulated version that fully resolves the risk. Current scientific understanding suggests formulation alone may not completely eliminate NDMA generation.
DMF solvent can degrade under acidic or basic conditions to form dimethylamine and formic acid. The dimethylamine produced acts as a direct precursor for NDMA formation. If nitrite contamination is present during synthesis, the reaction between dimethylamine and nitrosating agents can generate NDMA. This risk increases when recovered solvents are reused without proper testing. Because ranitidine synthesis already involves reactive amines, additional dimethylamine from DMF further elevates risk. Therefore, tight control of DMF quality is important during Nitrosamine Testing for Ranitidine.
A combination of headspace GC-MS/MS and LC-HRMS is considered the most reliable testing approach. GC-MS/MS is typically used for volatile nitrosamines such as NDMA and NDEA. LC-HRMS methods, including Orbitrap or QTOF systems, are used to detect drug substance-related nitrosamines. A critical requirement for ranitidine is avoiding heat during sample preparation. Elevated temperatures may artificially generate NDMA and produce false results. Cold extraction methods are therefore recommended to ensure accurate Nitrosamine Testing for Ranitidine.
Excipients with potential nitrite contamination should be carefully evaluated before use with ranitidine. Materials such as microcrystalline cellulose, corn starch, and lactose may contain measurable nitrite or nitrate levels. Povidone may also introduce peroxide impurities that promote nitrosation reactions. Agricultural-origin excipients often show variability depending on source and storage conditions. A conservative formulation approach includes minimizing excipient count and using highly purified grades. Establishing nitrite specifications for excipients helps reduce risk in finished products.
NDMA and N-nitroso ranitidine are controlled under different regulatory approaches. NDMA has a fixed acceptable intake limit of 96 ng per day based on carcinogenicity data. N-nitroso ranitidine, classified as an NDSRI, follows the threshold of toxicological concern approach. This typically results in lower acceptable intake limits depending on treatment duration. Additional toxicological evaluation may also be required for NDSRIs. Regulatory guidance recommends compound-specific assessment where necessary.
A nitrosamine risk assessment is a scientific evaluation of possible formation pathways. It considers API structure, synthesis steps, excipient contributions, and storage conditions. This assessment identifies which nitrosamines may form and where the risk exists. A confirmatory test is the laboratory analysis used to measure actual nitrosamine levels. Regulatory agencies require both steps as part of compliance. For ranitidine, the risk assessment usually leads to testing for NDMA and NDSRIs.
Reference:
- Liu, J., Zhao, Z., Yang, X., Jin, Y., Liu, X., Wang, C., & Zhang, Z. (2021). Determination of N-nitrosodimethylamine in ranitidine dosage forms by ESI-LC-MS/MS: Applications for routine laboratory testing. Iranian Journal of Pharmaceutical Research, 20(4), 255–264. https://doi.org/10.22037/ijpr.2021.115222.15258
- Aldawsari, F. S., Alshehry, Y. M., & Alghamdi, T. S. (2021). N-nitrosodimethylamine (NDMA) contamination of ranitidine products: A review of recent findings. Journal of Food and Drug Analysis, 29(1), 39–45. https://doi.org/10.38212/2224-6614.1133
- U.S. Food and Drug Administration. (2020). FDA updates and press announcements on NDMA in Zantac (ranitidine). https://www.fda.gov/drugs/drug-safety-and-availability/fda-updates-and-press-announcements-ndma-zantac-ranitidine
- Venkataraman, R., John, J. T., Shaju, M., & Asha, K. H. (2022). Nitrosamine impurities present in drugs. International Journal of Pharmaceutical Sciences Review and Research, 77(2), 54–61. https://globalresearchonline.net/ijpsrr/v77-2/08.pdf



