Introduction:
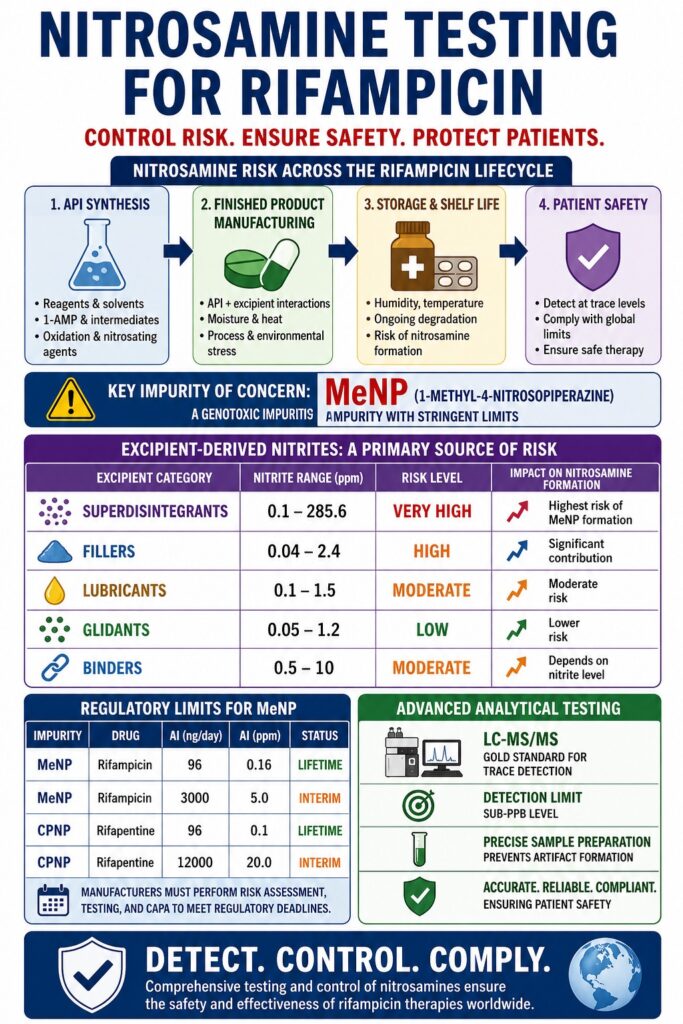
Nitrosamine Testing for Rifampicin is an essential analytical requirement focused on identifying and controlling 1-methyl-4-nitrosopiperazine (MeNP) along with other genotoxic impurities. This process plays a vital role in ensuring patient safety and preserving the effectiveness of global tuberculosis and leprosy treatment programs. Because rifampicin is widely used in high-dose, long-term therapies, even trace-level impurities can have significant clinical implications over time. Regulatory agencies have therefore emphasized strict monitoring to maintain acceptable exposure limits.
This evaluation spans the entire lifecycle of rifampicin, beginning with the fermentation-based synthesis of the rifamycin core and extending to the interactions between the API and excipient-derived nitrites in the finished dosage form. Each stage introduces unique chemical risks that must be carefully assessed. From raw material variability to storage conditions, multiple factors influence impurity formation. A lifecycle-based approach ensures that risks are identified early and controlled effectively.
Learn more about our specialized approach: Comprehensive Nitrosamine Analysis Services
Share via:
Key Highlights – Nitrosamine Testing for Rifampicin
- Lifecycle-based risk assessment is essential — nitrosamine formation in rifampicin can originate from API synthesis, raw materials, solvents, and finished product excipients, requiring evaluation from manufacturing through shelf life.
- Structural features of rifampicin increase nitrosation risk — the piperazine moiety and azomethine linkage can generate reactive intermediates such as 1-amino-4-methylpiperazine, which may oxidize to 1-methyl-4-nitrosopiperazine (MeNP).
- API synthesis introduces multiple contamination sources — solvents (DMF, DMA, NMP), alkyl nitrites, oxygen exposure, and recycled materials can contribute to nitrosamine formation during processing.
- Finished dosage forms remain vulnerable — excipient-derived nitrites, moisture, heat, and manufacturing processes such as wet granulation can promote nitrosamine generation during storage.
- Strict global regulatory limits apply — agencies including FDA, EMA, and Health Canada define acceptable intake limits for MeNP and require risk assessments, confirmatory testing, and CAPA implementation.
- Advanced LC-MS/MS methods are required for detection — highly sensitive analytical strategies with optimized sample preparation and matrix control are necessary to quantify trace-level nitrosamines and ensure compliance.
Structural Vulnerability and Nitrosatable Potential of the Rifampicin Molecule
Nitrosamine Testing for Rifampicin must start with a detailed structural evaluation, as the molecule contains functional groups that are inherently prone to nitrosation and oxidative reactions. Rifampicin is a semi-synthetic macrocyclic antibiotic of the rifamycin class, featuring a naphthofuranic chromophore and a seventeen-membered ansa bridge. These structural features contribute to both its therapeutic activity and its chemical instability under certain conditions. Understanding these vulnerabilities is critical for predicting impurity formation pathways.
The molecule contains secondary and tertiary amine groups that act as precursors for nitrosamine formation. The 1-methylpiperazine substituent at the 3-position introduces a tertiary amine that may undergo dealkylation, forming nitrosopiperazine derivatives. This transformation becomes more likely under oxidative stress or in the presence of reactive impurities. Even minor structural degradation can significantly alter the impurity profile.
A major concern is the azomethine (Schiff base) linkage, which is highly susceptible to hydrolysis, especially in acidic environments. This reaction produces 3-formylrifamycin SV and 1-amino-4-methylpiperazine (1-AMP). The 1-AMP compound is particularly reactive due to its semi-cyclic hydrazine nature and can be readily oxidized to MeNP. This pathway represents one of the most direct routes for genotoxic impurity formation in rifampicin systems.
Technical Insight: Direct Injection vs. Headspace Techniques for Nitrosamines
| Structural Element | Chemical Characterization | Potential for Nitrosation/Oxidation |
|---|---|---|
| Piperazine Moiety | Cyclic tertiary amine | Susceptible to nitrosative dealkylation or oxidation if fragmented |
| Azomethine (-\text{N}=\text{CH}-) | Schiff base linkage | Hydrolytically unstable; releases 1-AMP |
| Ansa Bridge | Macrocyclic aliphatic chain | Affects solubility and electronic distribution |
| Naphthofuran Core | Aromatic system | Complicates UV detection of trace impurities |
Comprehensive Risk Assessment of the API Synthesis Process
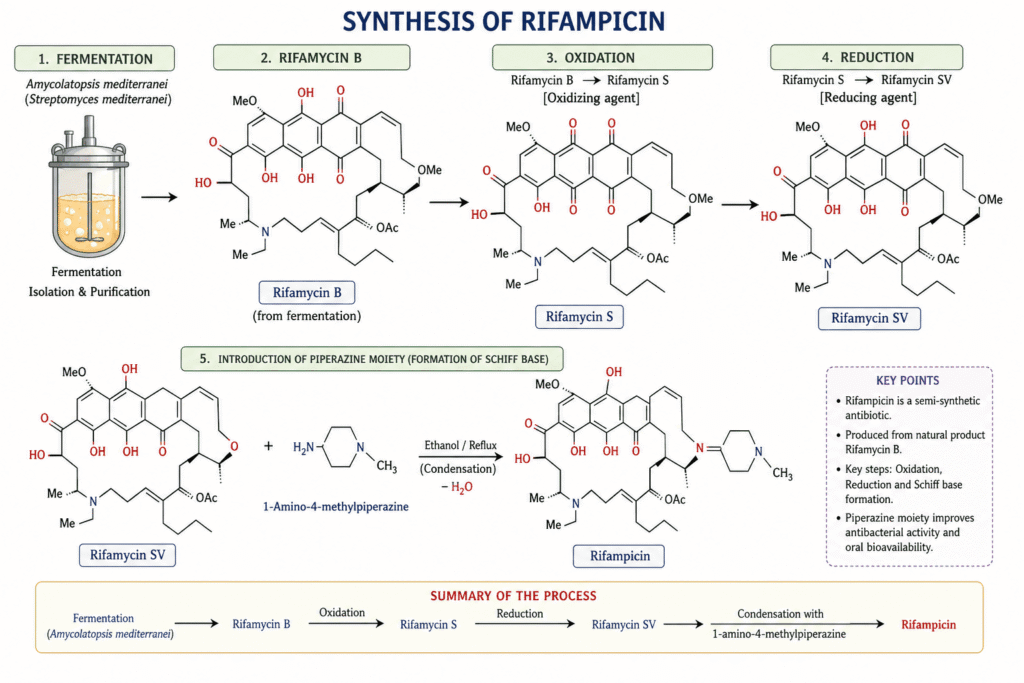
Nitrosamine Testing for Rifampicin requires a detailed evaluation of the API synthesis pathway, as multiple steps introduce potential contamination sources. The standard manufacturing process involves fermentation, oxidation, reduction, and final chemical condensation. Each stage can introduce reactive intermediates or impurities that contribute to nitrosamine formation. A thorough process understanding is necessary to identify critical control points.
Analysis of Starting Materials, Reagents, and Solvents
The synthesis begins with fermentation using Amycolatopsis mediterranei to produce Rifamycin B, followed by oxidation to Rifamycin S and reduction to Rifamycin SV. The introduction of a formyl group at the 3-position creates 3-formylrifamycin SV, typically through a Mannich reaction or similar chemical strategy. These transformations involve multiple reagents that may carry impurity risks. Careful selection and qualification of raw materials are therefore essential.
Common solvents such as DMF, DMA, and NMP can degrade into secondary amines like dimethylamine. These amines can react with nitrosating agents to form small nitrosamines such as NDMA. Additionally, catalysts like quaternary ammonium salts may degrade into tertiary amines, further increasing nitrosation risk. Even trace impurities in solvents can contribute significantly due to cumulative exposure during processing.
Explore mitigation strategies: Nitrosamine Solvent and Catalyst Mitigation Techniques
Nitrosating Agents and the Role of 1-Amino-4-Methylpiperazine
The final condensation step, involving 1-AMP, represents a major risk factor. This compound behaves as a “vulnerable amine” and is highly reactive in the presence of nitrosating agents. Organic nitrites such as isoamyl nitrite may either be used intentionally or appear as impurities, directly converting 1-AMP into MeNP. This step requires strict monitoring and control of reagent purity.
Oxidation of Hydrazine and Hydrazone Intermediates
Oxidation is a well-known pathway for converting hydrazines into nitrosamines. Exposure to atmospheric oxygen during drying, milling, or storage can lead to the formation of MeNP from residual 1-AMP or hydrazone intermediates. The presence of oxidants like peroxides, ozone, or hypochlorite further accelerates this process. Controlling oxygen exposure and minimizing oxidative stress are therefore critical.
Case Study Analysis: NDMA Root Cause Investigation for Complex Intermediates
Impact of Recovered and Recycled Materials
The use of recycled solvents introduces a systemic contamination risk. Cross-contamination from other production lines can introduce nitrites or amines into the process. Given the strict limit of 0.16 ppm for MeNP, even minimal carryover can result in non-compliance. Proper segregation and validation of recovery systems are essential to mitigate this risk.
| Synthesis Risk Factor | Mechanism | Associated Impurities |
|---|---|---|
| DMF/DMA/NMP | Secondary amine formation + nitrites | NDMA, NDEA |
| Alkyl Nitrites | Direct nitrosation of 1-AMP | MeNP |
| Atmospheric Oxygen | Oxidation of hydrazine intermediates | MeNP |
| Quaternary Salts | Dealkylation to amines | Various nitrosamines |
| Recycled Solvents | Cross-contamination | MeNP, NDMA |
Evaluation of Nitrosamine Risk in Finished Product Manufacturing
Nitrosamine Testing for Rifampicin must also consider the finished dosage form, where interactions between the API and excipients can lead to impurity formation during storage. The microenvironment within tablets and capsules plays a critical role in chemical stability. Factors such as humidity, temperature, and packaging conditions influence reaction kinetics. These variables must be carefully controlled throughout the product lifecycle.
Nitrosatable Functionality and Inherent API Reactivity
Rifampicin contains multiple reactive sites capable of undergoing nitrosation. The secondary-like nitrogen in 1-AMP is particularly susceptible, while tertiary amines in the piperazine ring contribute through indirect pathways. Degradation processes continue throughout shelf life, especially in the presence of moisture and oxygen. This makes long-term stability studies essential.
Excipient-Derived Nitrites as a Primary Root Cause
Nitrites present in excipients are a major source of nitrosamine formation. These impurities may originate from raw materials, process water, or environmental exposure. Their concentration varies widely depending on supplier and manufacturing conditions. Even low levels can drive significant impurity formation in high-dose formulations.
Advanced Strategies: The CPCA Approach for Nitrosamine Drug Substance Related Impurities (NDSRIs)
| Excipient Category | Nitrite Range (ppm) | Risk |
|---|---|---|
| Superdisintegrants | 0.1 – 285.6 | Very high |
| Fillers | 0.04 – 2.4 | High |
| Lubricants | 0.1 – 1.5 | Moderate |
| Glidants | 0.05 – 1.2 | Low |
| Binders | 0.5 – 10 | Moderate |
Manufacturing Processes and Environmental Stress
Processes like wet granulation increase exposure to moisture and heat, accelerating nitrosation reactions. Mechanical stress during milling or compression enhances contact between reactive components. Drying under atmospheric conditions further promotes oxidation. Switching to inert atmospheres and optimizing process parameters can significantly reduce risk.
NDSRIs and Regulatory Guidelines: Health Canada, FDA, and EMA
The detection of NDSRIs in rifampicin has led to coordinated regulatory actions worldwide. Nitrosamine Testing for Rifampicin is now governed by strict impurity limits based on toxicological risk assessments. These regulations aim to balance patient safety with continued access to essential medicines. Ongoing updates reflect evolving scientific understanding.
Published NDSRIs and Acceptable Intake Limits
Regulatory agencies have defined acceptable intake limits for MeNP and related compounds. Interim limits allow continued drug availability while long-term solutions are implemented. These limits are based on lifetime exposure models and cancer risk assessments.
| Impurity | Drug | AI (ng/day) | AI (ppm) | Status |
|---|---|---|---|---|
| MeNP | Rifampicin | 96 | 0.16 | Lifetime |
| MeNP | Rifampicin | 3000 | 5.0 | Interim |
| CPNP | Rifapentine | 96 | 0.1 | Lifetime |
| CPNP | Rifapentine | 12000 | 20.0 | Interim |
Regulatory Compliance: Impact of ICH M7(R2) Updates on Nitrosamine Risk Assessment
Implementation Timelines and CAPA Requirements
Manufacturers must follow a structured process including risk assessment, confirmatory testing, and corrective actions. Deadlines have been extended to allow adequate time for compliance. Continuous monitoring and documentation are required to demonstrate regulatory adherence. These measures ensure long-term product safety.
Submission Support: Nitrosamine Risk Assessment for ANDA Submissions
Advanced Analytical Strategies for Nitrosamine Testing for Rifampicin
Detecting trace levels of MeNP requires highly sensitive analytical techniques such as LC-MS/MS. These methods must overcome complex matrix effects and achieve very low detection limits. Method development requires significant expertise and validation. Advanced instrumentation ensures reliable and reproducible results.
Overcoming Matrix Interference and Sensitivity Hurdles
Rifampicin’s strong color and complex structure interfere with analysis. APCI is preferred over ESI due to reduced ion suppression. Careful optimization of instrument conditions is necessary. This improves accuracy and detection sensitivity.
Deep Dive: High-Resolution Mass Spectrometry (HRMS) for Nitrosamine Testing
Mandatory LC-MS/MS Method Parameters
Validated methods include specific chromatographic and mass spectrometric conditions. Internal standards such as MeNP-d4 improve quantification accuracy. Proper calibration and system suitability testing are essential. These ensure consistent performance across batches.
Specialized Sample Preparation
Sample preparation must prevent artificial formation of MeNP during analysis. Methanol-based extraction and pH control are commonly used. Stabilizing the API is critical to avoid false positives. Proper handling techniques significantly improve data reliability.
Analytical Excellence: Achieving Ultra-Low Limit of Quantitation (LOQ) in Testing
E-E-A-T: Demonstrating Expertise in Nitrosamine Testing for Rifampicin
Managing nitrosamine risks requires high levels of expertise and technical capability. Laboratories must demonstrate strong analytical proficiency and regulatory knowledge. Advanced method development and validation are key indicators of competence. This ensures accurate detection and compliance.
Organizations with specialized infrastructure can handle complex impurity profiling. Their ability to manage interferences and meet regulatory standards reflects their reliability. Following updated pharmacopeial guidelines further strengthens credibility. This builds trust with manufacturers and regulators.
Conclusion: Ensuring Safety through Nitrosamine Testing for Rifampicin
Nitrosamine Testing for Rifampicin is one of the most challenging areas in pharmaceutical quality control. It requires a deep understanding of chemical behavior, manufacturing processes, and analytical science. Identifying risks across the product lifecycle enables effective control strategies. This ensures compliance with global regulatory standards.
Advanced LC-MS/MS methods, combined with robust process controls, provide reliable detection of genotoxic impurities. These approaches help maintain the safety and effectiveness of rifampicin-based therapies. Continuous monitoring and improvement remain essential. This supports the global effort to combat tuberculosis safely.
Partner with Experts: Selecting the Right CRO for Nitrosamine Testing
For detailed technical consultations on risk assessments or to schedule validated nitrosamine testing for your products, please contact our laboratory experts:
https://resolvemass.ca/contact/
Frequently Asked Questions (FAQs)
The main origin of MeNP is the transformation of 1-amino-4-methylpiperazine (1-AMP), a reagent used in the final stage of rifampicin synthesis. This compound can undergo oxidation or react with nitrosating agents to form MeNP. Additionally, when rifampicin degrades, especially through hydrolysis of the azomethine linkage, it releases 1-AMP, which can further convert into MeNP. This makes both synthesis and degradation pathways important contributors.
Health authorities such as the FDA and EMA have defined a long-term acceptable limit of 0.16 ppm for MeNP to minimize cancer risk. However, a higher temporary limit of 5 ppm is permitted to prevent disruption in treatment availability. This approach ensures that patients continue receiving essential therapy for serious infections like tuberculosis. The decision reflects a balance between theoretical impurity risks and the immediate danger of untreated disease.
Excipients may carry trace amounts of inorganic nitrites introduced during their manufacturing or storage. In the finished dosage form, these nitrites can react with amine groups present in rifampicin or its degradation products, especially 1-AMP. Environmental factors such as heat, moisture, and pH variations can accelerate this reaction. As a result, excipients become an indirect but significant source of nitrosamine formation.
LC-MS/MS with Atmospheric Pressure Chemical Ionization (APCI) is widely regarded as the most reliable method for detecting nitrosamines in rifampicin. It offers the sensitivity needed to measure impurities at very low levels, such as 0.16 ppm. This technique also handles complex sample matrices effectively, reducing interference from the drug substance. Its accuracy and reproducibility make it the preferred choice in regulatory testing.
1-AMP plays a dual role as both a key intermediate in synthesis and a degradation product of rifampicin. Its chemical structure makes it highly reactive and prone to forming nitrosamines under suitable conditions. Because of this, it is considered a critical risk factor in impurity formation. Monitoring and controlling its presence is essential for maintaining product safety.
Valve switching helps protect the analytical instrument by diverting the main drug compound away from the detector. This prevents contamination and avoids overloading the system with high concentrations of rifampicin. As a result, the instrument remains sensitive enough to detect trace nitrosamines. It also improves method stability and extends the lifespan of the equipment.
Hydrazine-containing compounds like 1-AMP can react with oxygen or oxidizing agents during processing or storage. This reaction converts the hydrazine group into a nitroso functional group, forming a nitrosamine. The process is more likely to occur under conditions such as exposure to air, heat, or peroxide impurities. It is a well-recognized pathway in pharmaceutical impurity formation.
Manufacturers must first conduct a detailed risk assessment to identify possible sources of nitrosamine contamination. If a risk is detected, confirmatory testing is required to quantify impurity levels. Companies must also submit a corrective and preventive action (CAPA) plan outlining how they will reduce impurities to acceptable limits. Regulatory deadlines ensure timely compliance and continuous quality improvement.
Reference:
- O’Brien, C., Thayani, Z., Smith, T., Tran, A. V., Crotty, P., Young, A., Ford, A. I., & Vassar, M. (2025). Evaluating AI guidelines in leading family medicine journals: A cross-sectional study. BMC Primary Care, 26, 368. https://doi.org/10.1186/s12875-025-03044-0
- Boetzel, R., Schlingemann, J., Hickert, S., Korn, C., Kocks, G., Luck, B., Blom, G., Harrison, M., François, M., Allain, L., & others. (2023). Modeling the impact of excipient selection on nitrosamine formation towards risk mitigation. Pharmaceutics, 15(9), 2269. https://doi.org/10.3390/pharmaceutics15092269
- Wohlfart, J., Scherf-Clavel, O., Kinzig, M., Sörgel, F., & Holzgrabe, U. (2021). The nitrosamine contamination of drugs, part 3: Quantification of 4-methyl-1-nitrosopiperazine in rifampicin capsules by LC-MS/HRMS. Journal of Pharmaceutical and Biomedical Analysis, 203, 114205. https://doi.org/10.1016/j.jpba.2021.114205
- Witkowska, A. B., Wołczyńska, A., Lis-Cieplak, A., & Stolarczyk, E. U. (2023). Development and validation of LC-MS/MS method for the determination of 1-methyl-4-nitrosopiperazine (MNP) in multicomponent products with rifampicin—Analytical challenges and degradation studies. Molecules, 28(21), 7405. https://doi.org/10.3390/molecules28217405
- Tricker, A. R., Kumar, R., Siddiqi, M., Khuroo, M. S., & Preussmann, R. (1991). Endogenous formation of N-nitrosamines from piperazine and their urinary excretion following antihelmintic treatment with piperazine citrate. Carcinogenesis, 12(9), 1595–1599. https://doi.org/10.1093/carcin/12.9.1595
- de Souza, G. F. P., Matos, M. F. A. V., Aglio, T. C., Salles, A. G., Jr., & Rath, S. (2023). A comprehensive LC-UHPLC-MS/MS method for the monitoring of N-nitrosamines in lipophilic drugs: A case study with rifampicin. Journal of Pharmaceutical and Biomedical Analysis, 236, 115685. https://doi.org/10.1016/j.jpba.2023.115685



