
Introduction:
The Octreotide Sameness Study for ANDA submission is a critical regulatory requirement when filing a generic octreotide product with the USFDA. In this case study, ResolveMass Laboratories Inc. presents a detailed analytical roadmap used for the Sameness Evaluation of an Octreotide Generic Project submission, demonstrating structural identity, impurity equivalence, and physicochemical comparability with the Reference Listed Drug (RLD).
Octreotide is a synthetic cyclic octapeptide with a disulfide bridge and complex impurity profile. Due to its peptide nature, regulatory authorities demand rigorous analytical proof of sameness rather than simple chemical equivalence.
This case study outlines:
- Analytical strategy
- Instrumentation workflow (peptide mass spectrometry experts)
- Regulatory alignment
- Data interpretation approach
- Key regulatory considerations for ANDA success
Share via:
Summary:
- The Octreotide Sameness Study for ANDA submission is a full analytical demonstration that a generic octreotide product is structurally and functionally identical to the Reference Listed Drug (RLD).
- USFDA requires confirmation of primary structure, cyclic integrity, disulfide bridge configuration, impurity profile, related substances, and physicochemical comparability.
- High-resolution LC-MS/MS, peptide mapping, HRMS, and orthogonal analytical tools are mandatory for regulatory acceptance.
- ResolveMass Laboratories Inc. conducted a complete analytical characterization aligned with USFDA peptide characterization requirements.
- The study ensured regulatory-ready documentation, reproducible data, and defensible analytical validation.
1: What Is Required in an Octreotide Sameness Study for ANDA Submission?
In simple terms, USFDA requires complete analytical evidence that the generic octreotide is identical in structure, purity, and quality to the RLD.
For peptide generics like octreotide, sameness evaluation includes:
| Parameter | Requirement for ANDA |
|---|---|
| Primary structure | Exact amino acid sequence confirmation (peptide sequencing and mapping) |
| Molecular weight | Accurate mass verification via HRMS |
| Disulfide bridge | Correct cysteine linkage confirmation (peptide characterization techniques) |
| Impurity profile | Comparable related substances profile (impurity profiling in peptides) |
| Degradation pattern | Stress condition comparability (peptide degradation product characterization) |
| Physicochemical properties | HPLC retention, pH, solubility match (peptide purity testing) |
The Octreotide Sameness Study for ANDA submission requires orthogonal analytical validation supported by reproducible datasets (peptide characterization in drug development).
2: Project Overview: Octreotide Generic Sameness Evaluation
Demonstrate analytical sameness between the generic octreotide formulation and the RLD for ANDA filing.
Project Scope
ResolveMass Laboratories Inc. followed a USFDA-aligned analytical protocol to ensure the Octreotide Sameness Study for ANDA submission meets regulatory expectations. The study included:
- Primary sequence confirmation
- Cyclic structure verification
- Disulfide bond mapping
- Impurity profiling
- Comparative LC chromatographic assessment
- Forced degradation comparability (peptide degradation product characterization)
- Regulatory documentation preparation
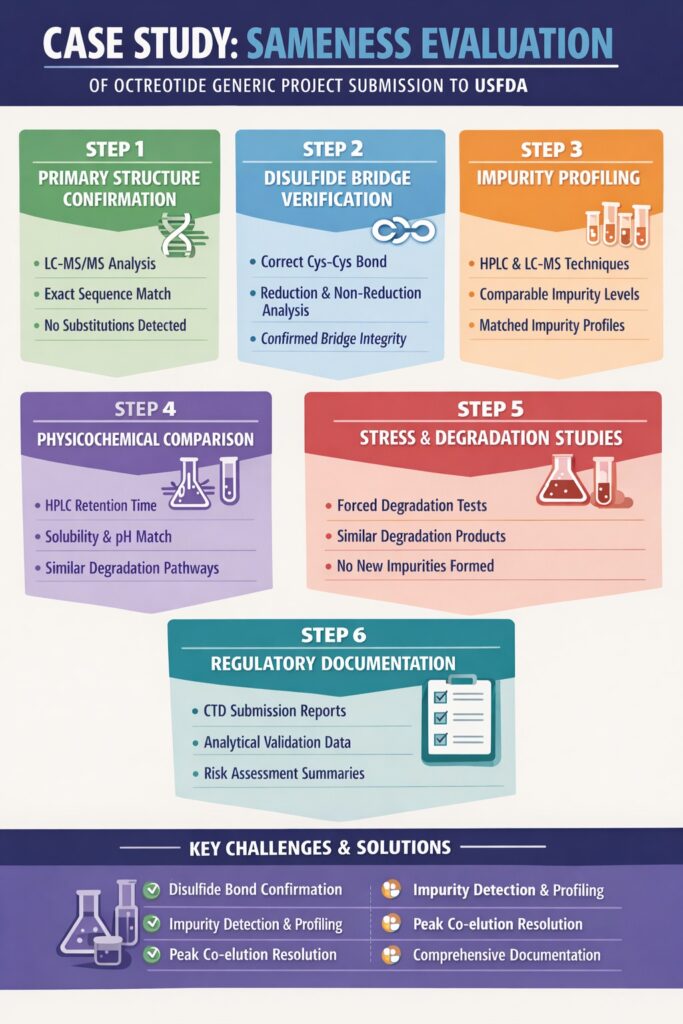
Step 1: Primary Structure Confirmation in Octreotide Sameness Study for ANDA Submission
We confirmed the exact amino acid sequence using high-resolution LC-MS/MS and peptide fragmentation analysis.
Octreotide is a cyclic octapeptide with the following sequence:
D-Phe-Cys-Phe-D-Trp-Lys-Thr-Cys-Thr-ol
Analytical Approach
- High-Resolution Mass Spectrometry (HRMS) (analytical support in peptide synthesis)
- MS/MS fragmentation pattern confirmation
- Theoretical vs observed mass comparison
- Sequence ion mapping (b/y ions)
Outcome
- Exact molecular mass match within ppm accuracy
- Complete sequence ion coverage
- No amino acid substitution detected
This confirms structural identity — a foundational requirement in the Octreotide Sameness Study for ANDA submission.
Step 2: Disulfide Bridge Verification
The correct Cys–Cys disulfide bond was confirmed using reduction and non-reduction comparative analysis.
Octreotide contains a crucial disulfide bond forming its cyclic structure. Any mismatch impacts biological activity.
Methodology
- Non-reduced LC-MS analysis
- DTT reduction studies
- Mass shift confirmation (+2H change)
- Comparative fragmentation studies
Regulatory Significance
USFDA requires confirmation of:
- Correct cysteine connectivity
- Absence of incorrect disulfide scrambling
- No open-chain peptide impurity
This step strengthens the robustness of the Octreotide Sameness Study for ANDA submission.
Step 3: Impurity Profiling and Related Substances Evaluation
Impurity profiles of the generic and RLD were compared using validated HPLC and LC-MS (impurity profiling in peptides).
Peptide impurities may include:
- Deamidation products
- Oxidation variants
- Epimerization products
- Truncated sequences
- Disulfide scrambling variants
Analytical Workflow
- Gradient RP-HPLC method development
- Impurity peak identification by LC-MS
- Relative retention time comparison
- Quantitative impurity comparison
Comparative Results
| Parameter | RLD | Generic | Status |
|---|---|---|---|
| Total impurities | Comparable | Comparable | Acceptable |
| Major impurity | Identified | Identified | Matched |
| Unknown peaks | None significant | None significant | Matched |
The impurity similarity supports regulatory acceptance of the Octreotide Sameness Study for ANDA submission.
Step 4: Physicochemical Comparability Assessment
Chromatographic behavior, solubility, and physicochemical properties were matched (peptide characterization techniques).
Evaluated Parameters
- HPLC retention time
- UV absorbance pattern
- pH compatibility
- Solubility characteristics
- Stability under stress conditions
Observations
- Identical retention window
- Comparable UV λmax
- Similar degradation pathways under acid/base stress
Such orthogonal data reinforces scientific credibility in the Octreotide Sameness Study for ANDA submission.
Step 5: Stress Testing and Degradation Studies
Forced degradation studies confirmed the generic behaves similarly to the RLD (peptide degradation product characterization).
Stress Conditions Applied
- Acid hydrolysis
- Base hydrolysis
- Oxidative stress
- Thermal exposure
- Photolytic degradation
Key Outcome
- Comparable degradation products
- No novel impurity formation
- Similar degradation kinetics
USFDA evaluates degradation behavior as part of impurity risk assessment in the Octreotide Sameness Study for ANDA submission.
Step 6: Regulatory Documentation Prepared for ANDA Filing
Comprehensive analytical reports were compiled in CTD-ready format (peptide characterization for IND and ANDA).
Included Documentation
- Method validation reports
- Chromatograms
- Mass spectra
- Comparative impurity tables
- Justification summaries
- Risk assessment statements
Octreotide’s cyclic structure, disulfide bond, and complex impurity pathways demand advanced peptide analytical expertise (top peptide testing labs, peptide characterization for FDA).
3: Regulatory Documentation Prepared for ANDA Filing
Comprehensive analytical reports were compiled in CTD-ready format for regulatory submission.
Included Documentation
- Method validation reports
- Chromatograms
- Mass spectra
- Comparative impurity tables
- Justification summaries
- Risk assessment statements
ResolveMass Laboratories Inc. ensures documentation meets USFDA expectations for peptide generics.
4: Why Octreotide Sameness Study for ANDA Submission Requires Specialized Expertise
Short answer: Because octreotide is a cyclic peptide with complex impurity pathways and disulfide chemistry.
Unlike small molecules, peptide generics demand:
- Advanced mass spectrometry expertise
- Deep knowledge of peptide degradation chemistry
- Regulatory understanding of ANDA pathways
- Orthogonal analytical strategies
ResolveMass Laboratories Inc. has extensive experience in peptide characterization, supporting multiple generic ANDA programs.
5: Key Regulatory Considerations for Octreotide ANDA Submission
- Exact sequence identity
- No structural variants
- Comparable impurity levels
- Equivalent degradation pathways
- Validated analytical methods
- Data reproducibility
- Complete CTD documentation
The Octreotide Sameness Study for ANDA submission is successful only when all these components align.
6: Challenges Faced and How They Were Addressed
| Challenge | Resolution |
|---|---|
| Disulfide bond confirmation | Reduction and MS/MS orthogonal validation |
| Low-level impurity detection | High sensitivity LC-MS methods |
| Peak co-elution | Optimized gradient separation |
| Regulatory defensibility | Comprehensive documentation |
Conclusion:
The Octreotide Sameness Study for ANDA submission is a scientifically rigorous process requiring advanced peptide characterization, impurity profiling, and regulatory documentation expertise (peptide sameness services in US, peptide sameness vs biosimilar).
Through orthogonal mass spectrometry, validated chromatographic methods, and CTD-ready reporting, this project exemplifies how analytical precision drives regulatory success.
For companies preparing an ANDA for peptide generics, a comprehensive Octreotide Sameness Study for ANDA submission is not optional — it is the foundation of approval.
Frequently Asked Questions:
A Sameness Study demonstrates that a generic octreotide product is structurally and functionally identical to the Reference Listed Drug (RLD). It includes analytical confirmation of primary structure, disulfide bonds, impurity profiles, and physicochemical properties to meet USFDA regulatory requirements.
Peptides like octreotide are complex, with cyclic structures and disulfide bridges. Accurate characterization ensures the generic maintains the same sequence, folding, and impurity profile as the RLD, supporting regulatory approval (peptide characterization techniques).
Common methods include high-resolution LC-MS/MS, HRMS, peptide mapping, RP-HPLC for impurity profiling, disulfide bond verification, and forced degradation studies. Orthogonal approaches ensure robust data and reproducibility (peptide mass spectrometry experts).
Disulfide connectivity is verified using reduction and non-reduction LC-MS, monitoring mass shifts and fragment ions to confirm correct Cys–Cys bond formation, preventing structural or functional errors in the generic peptide.
Comprehensive CTD-ready reports including method validation, chromatograms, mass spectra, impurity tables, risk assessments, and analytical justification are mandatory for regulatory submission (peptide characterization for IND and ANDA).
Reference
- Jessica A. Rogers-Crovak, Edward J. Delaney & David J. Detlefsen. Recommendation for Clarifying FDA Policy in Evaluating “Sameness” of Higher Order Structure for Generic Peptide Therapeutics.https://link.springer.com/article/10.1208/s12248-024-00994-8
- Le Wang, Shenglin Chen, Mengying Wu & Lijuan Zhou. Comparative analysis of adverse event profiles of lanreotide and octreotide in somatostatin-responsive endocrine and neoplastic diseases.https://www.nature.com/articles/s41598-025-03850-7
- Giuseppina Sabatino, Ivan Guryanov, Andrea Rombecchi, Jacopo Zanon, Antonio Ricci, Walter Cabri. Production of peptides as generic drugs: a patent landscape of octreotide.https://www.tandfonline.com/doi/abs/10.1517/13543776.2016.1158810
- David J. Brayden & Sam Maher. Transient Permeation Enhancer® (TPE®) technology for oral delivery of octreotide: a technological evaluation.https://www.tandfonline.com/doi/full/10.1080/17425247.2021.1942838


