
Introduction:
Orthogonal Analytical Techniques for ANDA Sameness Evaluation play a crucial role in demonstrating that a generic drug product is equivalent to its Reference Listed Drug (RLD). These techniques use different scientific principles to analyze the same attribute, ensuring that results are not method-dependent and are scientifically reliable.
In Abbreviated New Drug Application (ANDA) submissions, proving sameness goes beyond basic testing—it requires multi-dimensional characterization of the active pharmaceutical ingredient (API) and formulation. Advanced approaches such as sameness evaluation in ANDA and active ingredient sameness in ANDA highlight the importance of orthogonal strategies. These approaches provide the analytical depth needed for confident regulatory submissions while minimizing approval risks.
Summary:
- Orthogonal Analytical Techniques for ANDA Sameness Evaluation ensure comprehensive comparison of APIs and formulations using independent methods.
- They improve accuracy, reliability, and regulatory confidence by confirming results across multiple analytical principles.
- Critical for detecting impurities, structural differences, and subtle variations in complex molecules like peptides and biologics.
- Common orthogonal tools include LC-MS, NMR, FTIR, XRD, DSC, and chromatography techniques.
- Regulatory agencies expect robust, multi-technique evidence to demonstrate sameness in ANDA submissions.
1: What Are Orthogonal Analytical Techniques?
Orthogonal analytical techniques are independent analytical methods that evaluate the same property using different scientific principles. This ensures that the results are reliable, reproducible, and not dependent on a single method.
For example, molecular structure can be assessed using NMR (Nuclear Magnetic Resonance) and mass spectrometry. These approaches are widely applied in studies such as Lanreotide sameness study and Liraglutide sameness study, where complementary analytical insights are essential.
Key Characteristics
- Based on different principles: Each technique relies on distinct physical or chemical mechanisms
- Provide complementary data: Multiple perspectives improve overall understanding
- Reduce analytical bias: Minimizes dependence on a single method
- Enhance confidence: Cross-verification strengthens data reliability
In ANDA sameness evaluation, these characteristics are essential for generating robust, defensible analytical evidence that meets regulatory expectations.
2: Why Are Orthogonal Analytical Techniques Important in ANDA Sameness Evaluation?
Orthogonal analytical techniques are important because a single analytical method cannot fully characterize complex pharmaceutical products. Using multiple independent methods ensures comprehensive, reliable, and regulator-ready data to demonstrate sameness with the Reference Listed Drug (RLD).
This is especially critical in peptide sameness study for ANDA and FDA peptide sameness study requirements, where regulatory expectations demand multi-technique validation.
Key Reasons
- Regulatory Expectation: Health authorities expect robust evidence supported by multiple analytical techniques
- Data Confirmation: Cross-verification across methods improves accuracy and reproducibility
- Complex Molecules: Peptides, liposomes, and injectables require multi-dimensional characterization
- Impurity Detection: Enables identification of trace-level and structurally similar impurities
- Risk Mitigation: Minimizes the likelihood of regulatory queries, delays, or rejection
In ANDA submissions, orthogonal approaches provide high-confidence analytical proof, making them essential for successful approval.
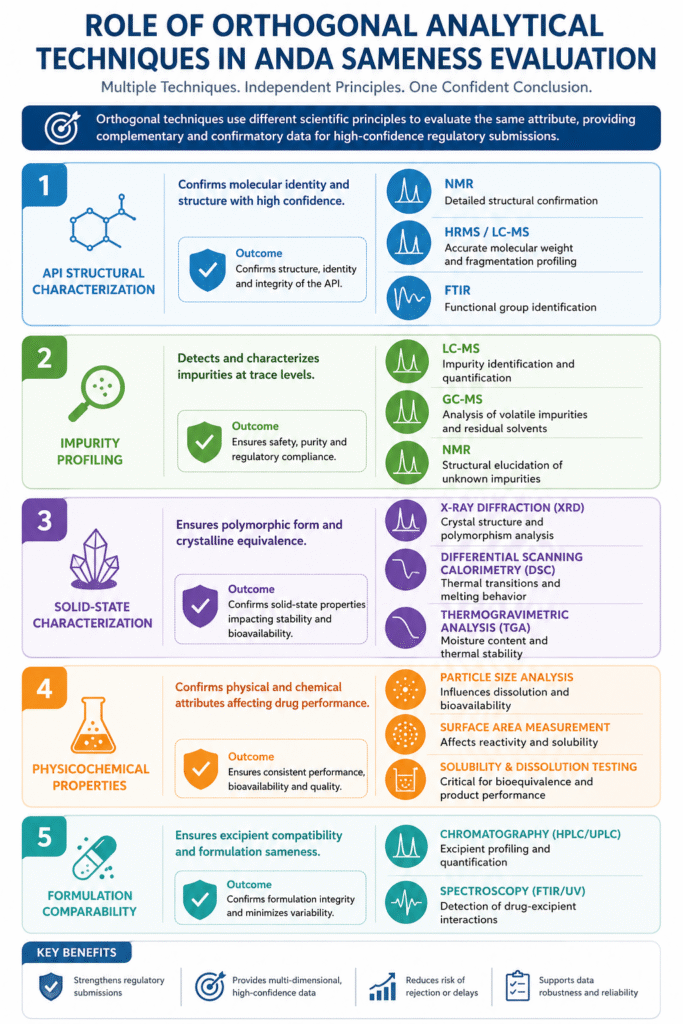
3: Core Areas Where Orthogonal Techniques Are Applied
Orthogonal analytical techniques are applied across multiple critical areas in ANDA sameness evaluation to ensure comprehensive and scientifically robust comparison of the generic product with the RLD.
These approaches are fundamental to analytical strategies for sameness study and complex peptide evaluations.
1. API Structural Characterization
Orthogonal techniques confirm molecular identity and structure with high confidence.
Used extensively in studies like Semaglutide sameness study and Octreotide sameness study for ANDA submission.
- NMR (Nuclear Magnetic Resonance): Detailed structural confirmation
- HRMS / LC-MS: Accurate molecular weight and fragmentation profiling
- FTIR (Fourier Transform Infrared Spectroscopy): Functional group identification
2. Impurity Profiling
Multiple orthogonal techniques detect and characterize impurities at trace levels.
Critical for identifying issues highlighted in peptide sameness study deficiencies.
- LC-MS: Identification and quantification of impurities
- GC-MS: Analysis of volatile impurities and residual solvents
- NMR: Structural elucidation of unknown impurities
3. Solid-State Characterization
Orthogonal methods ensure polymorphic form and crystalline equivalence.
- X-ray Diffraction (XRD): Crystal structure and polymorphism analysis
- Differential Scanning Calorimetry (DSC): Thermal transitions and melting behavior
- Thermogravimetric Analysis (TGA): Moisture and thermal stability assessment
4. Physicochemical Properties
Techniques confirm physical and chemical attributes that impact drug performance.
- Particle Size Analysis: Influences dissolution and bioavailability
- Surface Area Measurement: Affects reactivity and solubility
- Solubility & Dissolution Testing: Critical for bioequivalence
5. Formulation Comparability
Orthogonal approaches ensure excipient compatibility and formulation sameness.
- Chromatography (HPLC/UPLC): Excipient profiling and quantification
- Spectroscopy (FTIR/UV): Detection of drug-excipient interactions
By applying orthogonal techniques across these core areas, pharmaceutical companies can generate multi-dimensional, high-confidence data, which is essential for successful ANDA submissions and regulatory approval.
4: Common Orthogonal Analytical Techniques Used
These techniques are widely applied across global analytical programs including peptide sameness study services in United States and peptide sameness study services in Canada
| Analytical Technique | Principle | Application in ANDA Sameness |
|---|---|---|
| LC-MS / HRMS | Mass-to-charge ratio | Molecular weight, impurity profiling |
| NMR (1D & 2D) | Nuclear spin interactions | Structural elucidation |
| FTIR | Infrared absorption | Functional group identification |
| XRD | Crystal diffraction | Polymorphism analysis |
| DSC | Heat flow measurement | Thermal properties |
| HPLC / UPLC | Chromatographic separation | Assay, impurities |
| GC-MS | Volatile analysis | Residual solvents |
| ICP-MS | Elemental analysis | Trace metals |
5: How Orthogonal Techniques Strengthen Regulatory Submissions
Orthogonal Analytical Techniques for ANDA Sameness Evaluation strengthen regulatory submissions by ensuring data robustness, reproducibility, and scientific credibility. Using multiple independent methods provides regulators with high-confidence evidence that supports product equivalence.
These approaches are especially critical in targeted submissions such as Liraglutide sameness study for ANDA submission.
Regulatory Advantages
- Multi-layered Evidence: Confirms sameness through complementary analytical approaches
- Reduced Review Cycles: Minimizes regulatory queries and deficiency letters
- Scientific Rigor: Demonstrates strong analytical design and method validation
- Regulatory Alignment: Meets expectations from agencies such as the USFDA and EMA
By integrating orthogonal techniques into the analytical strategy, companies can significantly improve approval timelines, submission quality, and regulatory confidence in ANDA filings.
6: Challenges in Implementing Orthogonal Analytical Techniques
While Orthogonal Analytical Techniques for ANDA Sameness Evaluation are highly powerful, they come with operational, technical, and resource-related challenges. Proper planning and strategy are essential to fully leverage their benefits.
Insights from peptide sameness study deficiencies highlight common analytical gaps and mitigation needs.
Common Challenges
- High Instrumentation Cost: Advanced tools like HRMS, NMR, and XRD require significant capital investment
- Specialized Expertise: Skilled analysts are needed for method development, execution, and interpretation
- Complex Data Interpretation: Multi-technique datasets can be difficult to correlate and analyze
- Time-Intensive Method Development: Developing and validating multiple methods increases timelines
Mitigation Strategies
- Use Validated & Standardized Methods: Ensures consistency, reliability, and regulatory acceptance
- Leverage Advanced Data Integration Tools: Software solutions help correlate multi-technique data efficiently
- Collaborate with Expert Analytical Labs: Access to experienced scientists and state-of-the-art infrastructure
- Adopt Risk-Based Strategies: Focus efforts on critical quality attributes (CQAs) to optimize resources
By proactively addressing these challenges, organizations can successfully implement orthogonal approaches and generate robust, high-quality data that strengthens ANDA submissions and regulatory outcomes.
7: Best Practices for Effective Orthogonal Analysis
A structured and strategic approach ensures maximum benefit from Orthogonal Analytical Techniques for ANDA Sameness Evaluation by improving data quality, reliability, and regulatory acceptance.
A structured approach ensures maximum benefit and aligns with analytical strategies for sameness study.
Recommended Approach
- Select Techniques Based on API Complexity: Choose orthogonal methods aligned with the molecular nature (small molecule, peptide, complex injectable)
- Use Multiple Independent Methods: Apply at least two orthogonal techniques for each critical quality attribute (CQA)
- Perform Forced Degradation Studies: Understand degradation pathways and confirm method specificity
- Ensure Method Validation & Reproducibility: Validate all analytical methods as per regulatory guidelines to ensure consistency
- Maintain Comprehensive Documentation: Record methodologies, results, and interpretations to support regulatory review
Implementing these best practices helps generate robust, reproducible, and defensible analytical data, significantly improving the success rate of ANDA submissions.
8: Case Example: Orthogonal Approach in Peptide Sameness
Peptide APIs require multiple orthogonal analytical techniques to confirm structural integrity, sequence accuracy, and impurity equivalence with high confidence. A single method is insufficient due to the inherent complexity of peptide molecules.
Peptide APIs require multi-technique validation as demonstrated in:
Typical Workflow
- LC-MS: Peptide mapping and impurity detection
- HRMS: Accurate mass measurement and confirmation of molecular composition
- 2D NMR: Detailed sequence verification and structural elucidation
- HPLC: Purity assessment and quantitative assay
Why This Approach Works
- Comprehensive Characterization: Covers identity, structure, and purity from multiple analytical perspectives
- High Sensitivity & Specificity: Detects even trace-level impurities and minor sequence variations
- Cross-Verification: Confirms results across independent techniques, reducing uncertainty
- Regulatory Confidence: Meets stringent expectations for complex generic approvals
This multi-technique orthogonal strategy ensures complete molecular characterization, making it essential for successful ANDA approval of peptide-based generics.
9: Future Trends in Orthogonal Analytical Techniques
Emerging technologies are making Orthogonal Analytical Techniques for ANDA Sameness Evaluation more efficient, precise, and data-driven. These advancements are enhancing analytical depth while reducing time and complexity.
Key Trends
- Integration of AI-Driven Data Analysis:
Artificial intelligence and machine learning are enabling faster interpretation of complex, multi-technique datasets, improving accuracy and decision-making. - Advanced Hyphenated Techniques (LC-MS/MS, GC-MS/MS):
Next-generation hyphenated systems provide higher sensitivity, selectivity, and structural insight, especially for trace-level impurities. - Increased Use of Automation and Robotics:
Automated sample preparation and analysis improve reproducibility, reduce human error, and enhance laboratory efficiency. - Real-Time Analytical Monitoring:
Process Analytical Technology (PAT) and real-time monitoring tools allow continuous assessment of critical quality attributes during manufacturing.
These innovations are transforming orthogonal analysis into a more integrated, intelligent, and high-throughput approach, strengthening its role in future ANDA submissions and regulatory success.
Conclusion:
Orthogonal Analytical Techniques for ANDA Sameness Evaluation are indispensable for demonstrating pharmaceutical equivalence with scientific confidence. By leveraging multiple independent analytical methods, pharmaceutical companies can ensure accuracy, regulatory compliance, and faster approvals.
By integrating approaches used in sameness evaluation in ANDA and advanced peptide studies, companies can ensure accuracy, compliance, and faster approvals.
A well-designed orthogonal strategy not only strengthens ANDA submissions but also builds credibility, reliability, and long-term regulatory success. As drug complexity increases, the importance of orthogonal approaches will continue to grow, making them a cornerstone of modern pharmaceutical analysis.
Frequently Asked Questions:
Orthogonal analytical techniques are independent methods used to analyze the same quality attribute using different scientific principles.
They help confirm results through multiple scientific approaches like spectroscopy, chromatography, and mass spectrometry.
This reduces analytical bias and improves confidence in data.
They are widely used in ANDA submissions to demonstrate drug sameness with the RLD.
Overall, they ensure robust and scientifically reliable characterization of pharmaceutical products.
They are important because a single method cannot fully characterize complex drug products.
Orthogonal techniques provide multi-layered and confirmatory evidence of sameness.
They improve regulatory confidence by ensuring accurate and reproducible results.
They also help reduce review queries and approval delays.
This makes submissions more scientifically strong and regulator-ready.
Common techniques include LC-MS/HRMS, NMR, FTIR, HPLC/UPLC, XRD, DSC, and GC-MS.
Each technique provides different but complementary information about the sample.
For example, LC-MS helps with mass and impurities while NMR confirms structure.
Together, they provide a complete analytical profile of the drug.
This combination ensures strong scientific validation of sameness.
They detect and confirm impurities using multiple independent methods.
LC-MS identifies impurity profiles, while NMR helps in structural confirmation.
GC-MS is useful for volatile impurities and residual solvents.
Using multiple techniques ensures no impurity is overlooked.
This improves accuracy and regulatory acceptance of impurity data.
Yes, they are essential for peptide-based drugs due to high structural complexity.
Techniques like LC-MS, HRMS, and 2D NMR are commonly used together.
They help confirm sequence, molecular weight, and impurity profiles.
This ensures accurate comparison with the reference product.
Regulatory agencies strongly expect orthogonal evidence for peptide generics.
They require high-cost advanced instruments and skilled professionals.
Data interpretation can be complex due to multi-technique outputs.
Method development and validation can be time-consuming.
Integration of results from different techniques can be challenging.
However, these challenges can be managed with proper strategy and expertise.
Companies should select techniques based on API complexity and risk level.
Using validated methods ensures consistency and regulatory acceptance.
Data integration tools can help manage complex datasets efficiently.
Collaboration with experienced analytical labs improves reliability.
A risk-based approach ensures focused and cost-effective analysis.
Reference
- Jáčová J, Gardlo A, Dimandja JM, Adam T, Friedecký D. Impact of sample dimensionality on orthogonality metrics in comprehensive two-dimensional separations. Analytica chimica acta. 2019 Aug 8;1064:138-49.https://www.sciencedirect.com/science/article/pii/S0003267019302910
- Nupur N, Joshi S, Gulliarme D, Rathore AS. Analytical similarity assessment of biosimilars: global regulatory landscape, recent studies and major advancements in orthogonal platforms. Frontiers in bioengineering and biotechnology. 2022 Feb 9;10:832059.https://www.frontiersin.org/journals/bioengineering-and-biotechnology/articles/10.3389/fbioe.2022.832059/full
- Al Bakain R, Rivals I, Sassiat P, Thiébaut D, Hennion MC, Euvrard G, Vial J. Comparison of different statistical approaches to evaluate the orthogonality of chromatographic separations: application to reverse phase systems. Journal of Chromatography a. 2011 May 20;1218(20):2963-75.https://www.sciencedirect.com/science/article/pii/S0021967311003797
- Simon Jr CG, Borgos SE, Calzolai L, Nelson BC, Parot J, Petersen EJ, Roesslein M, Xu X, Caputo F. Orthogonal and complementary measurements of properties of drug products containing nanomaterials. Journal of Controlled Release. 2023 Feb 1;354:120-7.https://www.sciencedirect.com/science/article/pii/S016836592200863X


