
Introduction:
In today’s competitive pharmaceutical landscape, companies increasingly Outsource GLP-1 Peptide Sequencing Services to specialized analytical laboratories to accelerate development timelines and ensure high-quality data. GLP-1 peptides, such as liraglutide and semaglutide, are complex molecules requiring advanced analytical techniques like LC-MS characterization of GLP-1 peptides for accurate sequencing and characterization.
A well-defined GLP-1 analog peptide sequencing workflow is essential to ensure reproducibility and regulatory acceptance.
Outsourcing not only reduces operational burden but also provides access to cutting-edge instrumentation, experienced scientists, and regulatory expertise—factors that are essential for successful drug development and approval. Leading providers offering GLP-1 peptide sequencing CRO services enable companies to streamline this process efficiently.
Share via:
Summary:
- Outsource GLP-1 Peptide Sequencing Services helps pharmaceutical companies access advanced analytical expertise without heavy capital investment.
- Cost depends on project complexity, analytical depth, and regulatory requirements.
- Timeline ranges from a few days to several weeks depending on sequencing approach and validation needs.
- Data quality is critical for regulatory submissions and depends on instrumentation, expertise, and method robustness.
- Choosing the right CRO/CDMO partner ensures accurate sequencing, impurity profiling, and compliance-ready data.
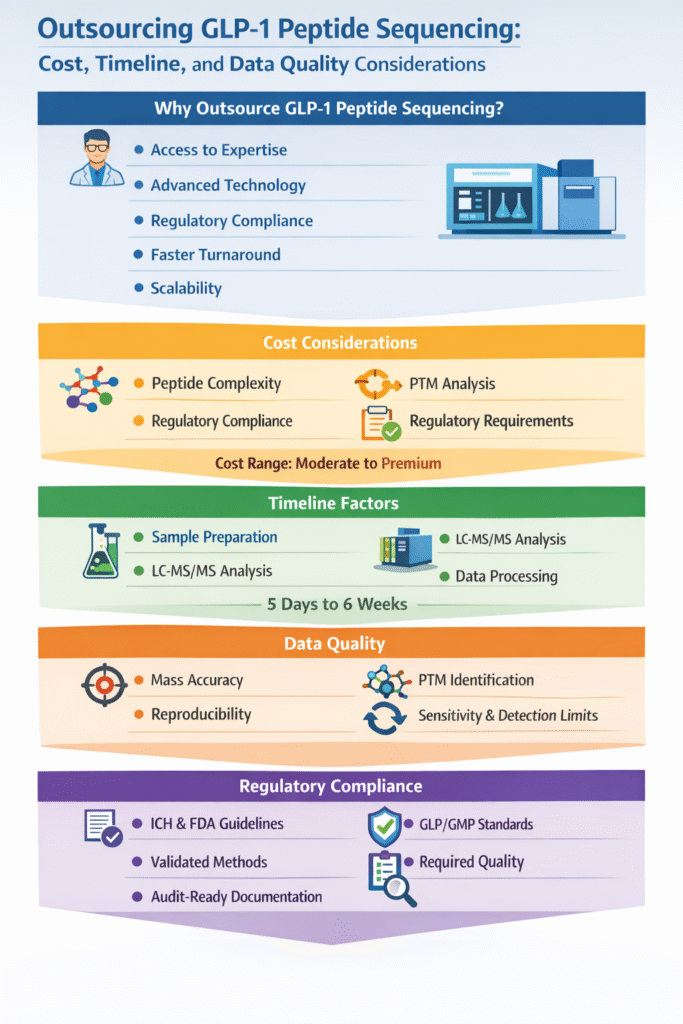
1: Why Do Companies Outsource GLP-1 Peptide Sequencing Services?
Companies outsource to reduce cost, gain expertise, and ensure regulatory-compliant high-quality data.
GLP-1 peptide sequencing is technically demanding and requires advanced approaches such as peptide sequencing of GLP-1 peptide and peptide sequencing of GLP-1 drugs.
GLP-1 peptide sequencing is technically demanding and requires:
- High-resolution mass spectrometry
- Advanced peptide mapping techniques
- Expertise in post-translational modification (PTM) analysis
- Regulatory-compliant documentation
Key Reasons for Outsourcing
- Access to Expertise: Skilled scientists with deep knowledge of peptide chemistry and analytical characterization of GLP-1 peptide drugs
- Advanced Technology: High-end LC-MS/MS systems without capital investment
- Regulatory Compliance: Data aligned with regulatory requirements for GLP-1 peptide characterization
- Faster Turnaround: Optimized workflows reduce project timelines
- Scalability: Ability to handle small research projects to large regulatory studies via specialized CRO for GLP-1 peptide characterization
2: Cost Considerations in Outsourcing GLP-1 Peptide Sequencing
Costs vary based on project complexity, analytical depth, and regulatory requirements.
Outsourcing is often more cost-effective than building in-house capabilities, especially when using advanced techniques like de novo GLP-1 peptide sequencing accuracy approaches.
Key Cost Drivers
| Factor | Impact on Cost |
|---|---|
| Peptide complexity | Higher complexity increases analysis time |
| Type of sequencing | De novo sequencing costs more than targeted |
| PTM analysis | Additional cost for modification mapping |
| Sample number | Bulk samples reduce per-sample cost |
| Regulatory requirements | GLP/GMP compliance increases cost |
| Turnaround time | Faster delivery may incur premium charges |
Estimated Cost Range
- Basic sequencing: Moderate cost
- Advanced LC-MS/MS with PTM mapping: Higher cost
- Regulatory-grade studies: Premium pricing
Cost Optimization Tips
- Clearly define project scope
- Avoid unnecessary analyses
- Choose experienced providers to reduce rework
3: Timeline Considerations
Typical timelines range from a few days to several weeks depending on analysis depth.
Efficient timelines depend on structured workflows and proven methodologies such as GLP-1 peptide sequencing analytical technique.
Time is critical in drug development, and outsourcing helps streamline workflows.
Typical Timeline Breakdown
| Stage | Time Required |
|---|---|
| Sample preparation | 1–3 days |
| LC-MS/MS analysis | 2–7 days |
| Data processing | 3–10 days |
| Reporting | 2–5 days |
Total Estimated Timeline
- Simple projects: 5–10 days
- Complex sequencing with PTMs: 2–4 weeks
- Regulatory submissions: 3–6 weeks
Factors Affecting Timeline
- Sample quality and preparation
- Complexity of peptide structure
- Required depth of analysis
- Number of samples
- Regulatory documentation needs
4: Data Quality Considerations
Data quality depends on instrumentation, method validation, and scientific expertise.
For GLP-1 peptides, data accuracy is critical because even minor errors can impact drug safety and efficacy. High-quality outputs are achieved through robust GLP-1 peptide impurity characterization and stability evaluation.
For GLP-1 peptides, data accuracy is critical because even minor errors can impact drug safety and efficacy.
Key Data Quality Parameters
- Mass accuracy (m/z precision)
- Sequence coverage (%)
- Reproducibility
- Sensitivity and detection limits
- PTM identification accuracy
Best Practices for High-Quality Data
- Use high-resolution LC-MS/MS systems
- Validate analytical methods
- Ensure proper calibration and controls
- Maintain detailed documentation
- Follow GLP-1 peptide characterization regulatory requirements
Common Data Outputs
- Fragment ion spectra
- Peptide sequence confirmation
- Impurity profiling
- Modification mapping
Additionally, applying GLP-1 peptide stability analytical methods ensures long-term product reliability.
5: Regulatory Considerations
Regulatory compliance is essential for drug approval and requires validated, traceable data.
GLP-1 peptide sequencing data must align with global expectations and follow strict regulatory requirements for GLP-1 peptide characterization.
GLP-1 peptide sequencing data must meet strict regulatory standards.
Key Regulatory Requirements
- ICH guidelines (Q2, Q6B)
- FDA and EMA expectations
- GLP/GMP compliance
- Data integrity (ALCOA principles)
Documentation Requirements
- Method validation reports
- Analytical protocols
- Raw data and processed results
- Audit-ready documentation
6: Challenges in Outsourcing GLP-1 Peptide Sequencing
Common challenges include communication gaps, data inconsistency, and vendor selection.
Understanding GLP-1 peptide sequencing challenges helps organizations proactively mitigate risks.
Major Challenges
- Misaligned project expectations
- Delays due to unclear requirements
- Inconsistent data quality from inexperienced providers
- Limited transparency in analytical methods
How to Overcome These Challenges
- Define clear project scope
- Choose experienced and specialized CROs
- Maintain regular communication
- Request sample reports before project initiation
7: How to Choose the Right Partner
Select a partner with proven expertise, advanced technology, and regulatory experience.’
Choosing a partner offering comprehensive analytical characterization of GLP-1 peptide drugs is essential.
Key Selection Criteria
- Experience in GLP-1 peptide analysis
- Advanced LC-MS/MS capabilities
- Strong regulatory track record
- Transparent communication
- End-to-end analytical support
Why Expertise Matters
An experienced provider ensures:
- Accurate sequence confirmation
- Reliable impurity detection
- Faster turnaround times
- Regulatory-ready data
8: Why ResolveMass Laboratories Inc. is a Trusted Partner
ResolveMass Laboratories Inc. stands out as a leading provider of Outsource GLP-1 Peptide Sequencing Services by combining scientific expertise with advanced analytical capabilities.
Core Strengths
- Advanced mass spectrometry platforms
- Deep expertise in peptide sequencing and characterization
- Strong regulatory compliance (FDA, EMA, ICH)
- High data accuracy and reproducibility
- Customized solutions for each project
What Sets ResolveMass Apart
- Proven experience in GLP-1 peptide analysis
- Comprehensive impurity profiling and PTM mapping
- Fast and reliable turnaround times
- Client-focused approach with transparent communication
Conclusion
Outsourcing is a strategic decision that enables pharmaceutical companies to efficiently manage cost, timeline, and data quality. By choosing to Outsource GLP-1 Peptide Sequencing Services, organizations gain access to specialized expertise, advanced instrumentation, and regulatory-compliant data—critical components for successful drug development.
Selecting the right partner, such as ResolveMass Laboratories Inc., ensures not only accurate sequencing results but also confidence in regulatory submissions and product quality.
Frequently Asked Questions:
Reference
- Hach M, Engelund DK, Mysling S, Mogensen JE, Schelde O, Haselmann KF, Lamberth K, Vilhelmsen TK, Malmstrøm J, Højlys-Larsen KB, Rasmussen TS. Impact of manufacturing process and compounding on properties and quality of follow-On GLP-1 polypeptide drugs. Pharmaceutical Research. 2024 Oct;41(10):1991-2014.https://link.springer.com/article/10.1007/s11095-024-03771-6
- Wen J, Razick A, How‐Volkman C, Bernstein E, Nadora D, Truong A, Razick D, Akhtar M, Karabala M, Frezza E. An exploratory analysis of glucagon‐like peptide‐1 (GLP‐1) agonists and biosimilars: A literature review. Diabetes, Obesity and Metabolism. 2025 Mar;27(3):1113-22.https://dom-pubs.onlinelibrary.wiley.com/doi/abs/10.1111/dom.16110
- CREMONESE J. GLP-1 Receptor Agonists in the Pharmaceutical Landscape: An Analysis of Current Applications, Market Barriers, and Future Developments.https://thesis.unipd.it/handle/20.500.12608/76821
- Bursick A. An Analysis of Safety Risks Associated with GLP-1 RA Use in the Direct-to-Consumer Telehealth Model Compared to Traditional Care Model (Doctoral dissertation, Northeastern University).https://search.proquest.com/openview/6bc47d21ca7ebd09b4b0a6eace6e93f9/1?pq-origsite=gscholar&cbl=18750&diss=y
- Walter-Evans, A., 2025. Advancements in GLP-1 Receptor Agonists for Treating Eating Disorders-Advancements in GLP-1 Receptor Agonists for Treating Eating Disorders: Mechanisms, Clinical Successes, Genetic Insights, and Broader Economic and Public Health Impacts. Clinical Successes, Genetic Insights, and Broader Economic and Public Health Impacts (December 23, 2025).https://papers.ssrn.com/sol3/papers.cfm?abstract_id=5964739


