
Introduction:
Outsourced Bioanalysis Services help biotech startups reach the clinic faster by providing ready-to-deploy analytical expertise, infrastructure, and regulatory support—without the burden of building in-house labs.
For early-stage biotech companies, speed is survival. Every month saved before filing an IND (Investigational New Drug application) can significantly improve investor confidence, valuation, and competitive positioning. However, establishing internal bioanalytical capabilities requires:
- Capital-intensive equipment such as LC-MS/MS bioanalytical services
- Specialized bioanalytical method development
- GLP-compliant infrastructure and regulated bioanalytical services
- Quality systems aligned with regulatory agencies
- Deep understanding of why bioanalysis is important in drug development
This is where Bioanalytical CRO services become a strategic advantage rather than a simple vendor relationship.
Share via:
Summary:
- Outsourced bioanalysis for drug development reduces time-to-clinic by eliminating infrastructure setup and hiring delays.
- Startups gain immediate access to validated platforms and high-sensitivity bioanalysis capabilities.
- Early-phase work including PK/PD bioanalysis, ADA, and biomarker bioanalytical services is executed faster.
- Established CRO partners provide bioanalytical services for IND/NDA submissions.
- Scalable clinical bioanalytical services support seamless transition to Phase I.
- Robust data integrity in bioanalytical studies reduces compliance risk. GLP/GCP standards.
1: How Do Outsourced Bioanalysis Services Improve Speed-to-Clinic?
Outsourced Bioanalysis Services improve speed-to-clinic by eliminating infrastructure delays and providing immediate access to validated methods, experienced scientists, and regulatory-ready systems.
For startups, time is one of the most valuable assets. Building internal bioanalytical capabilities can take 12–24 months due to equipment procurement, lab setup, quality system development, and hiring specialized staff. Outsourcing removes these barriers and allows programs to move forward immediately.
Instead of spending 12–24 months building in-house labs, startups leveraging bioanalytical outsourcing can:
- Initiate rapid bioanalytical method development within weeks
- Begin bioanalytical quantification immediately after study start
- Align strategy with IND-enabling bioanalytical studies expectations early
- Access validated SOPs and instrumentation
- Utilize regulatory documentation teams
Immediate Timeline Acceleration
Instead of waiting to establish in-house capacity, startups can:
- Initiate method development within weeks of project kickoff
- Begin sample analysis immediately after study initiation
- Align bioanalytical strategy early with IND expectations
- Access pre-qualified instrumentation and SOPs
- Leverage experienced regulatory documentation teams
Where Timeline Compression Occurs
A structured outsourcing model accelerates progress across multiple development stages:
1. Preclinical Pharmacokinetics (PK)
- Rapid assay development and validation
- High-throughput sample analysis
- PK/TK bioanalysis integration
- Reliable exposure data to guide dose selection
2. Toxicokinetic (TK) Studies
- Timely quantification during GLP toxicology studies
- Integrated reporting for regulatory submission
- Reduced risk of delayed toxicology packages
- Timely toxicokinetic bioanalysis
3. Biomarker Analysis
- Early integration of pharmacodynamic endpoints
- Integrated biomarker bioanalytical services
- Faster decision-making based on translational data
- Improved clinical trial design readiness
4. Immunogenicity (ADA) Testing
- Validated anti-drug antibody assays
- Risk mitigation before clinical exposure
- Regulatory-aligned immunogenicity assessments
5. Early Clinical Phase I Bioanalysis
- Dedicated clinical bioanalytical services teams
- Real-time data reporting
- IND-ready documentation packages
A well-structured outsourcing model compresses development timelines across:
- Preclinical pharmacokinetics (PK)
- Toxicokinetic (TK) studies
- Biomarker analysis
- Immunogenicity (ADA) testing
- Early clinical Phase I bioanalysis
Outsourced Bioanalysis Services are not just operational support—they are a strategic accelerator. By removing internal bottlenecks and embedding regulatory expertise from day one, startups can significantly reduce the time required to move from discovery to Phase I clinical trials.
2: The Startup Bottleneck: Why In-House Bioanalysis Slows Progress
Building internal labs requires major investment in:
- Triple quadrupole systems for LC-MS/MS bioanalysis of xenobiotics
- Infrastructure to manage bioanalytical matrix effects
- Complex bioanalytical stability testing
- Expertise to avoid common bioanalytical mistakes
In contrast, affordable bioanalytical services for biotech startups eliminate capital barriers while maintaining regulatory compliance.
| Challenge | In-House Model | Outsourced Bioanalysis Services |
|---|---|---|
| Equipment Procurement | 6–12 months | Immediately available |
| Method Validation | Depends on internal expertise | Established SOP-driven validation |
| Regulatory Compliance | Build from scratch | Pre-audited GLP-compliant systems |
| Staffing | Recruitment + training delays | Experienced PhD/MS scientists onboard |
| Quality Systems | Develop & implement | Already operational |
For startups operating on venture funding cycles, these delays can be critical.
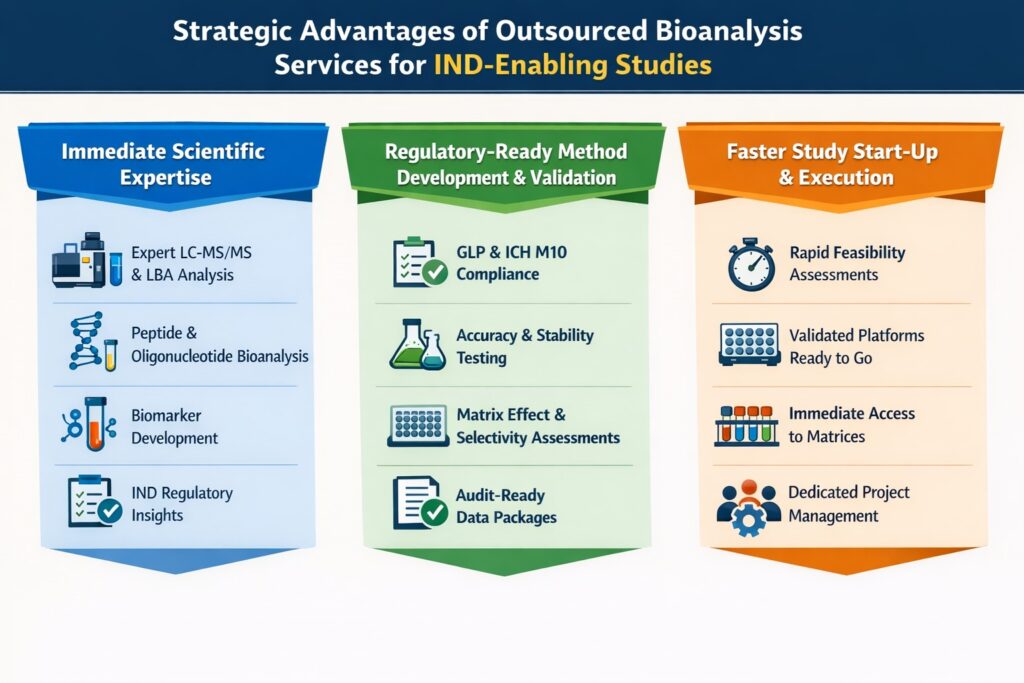
3: Strategic Advantages of Outsourced Bioanalysis Services for IND-Enabling Studies
1. Immediate Scientific Expertise
Outsourced Bioanalysis Services provide access to senior scientists with deep experience in regulatory bioanalysis.
Experienced teams provide:
- Small molecule quantification
- LC-MS bioanalysis for oligonucleotides
- Antibody-drug conjugate bioanalytical services
- Cell and gene therapy bioanalysis
- Hybrid LBA–LC-MS assays
- Proteomics bioanalytical services
Complex modalities benefit from advanced bioanalytical strategies for complex drug modalities.
2. Regulatory-Ready Method Development & Validation
Regulatory-aligned method validation prevents rework and IND delays.
Regulatory success requires:
- Bioanalytical method validation
- Addressing challenges in bioanalytical method development
- Managing matrix variability
- Strong documentation systems
Well-structured quality bioanalysis ensures generation of robust bioanalytical data.
Bioanalytical methods must meet:
- U.S. Food and Drug Administration guidance
- Health Canada requirements
- International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH M10 Bioanalytical Method Validation)
Outsourced labs experienced with these frameworks ensure:
- Accuracy & precision validation
- Stability assessments
- Matrix effect evaluation
- Selectivity and sensitivity validation
This minimizes the risk of regulatory queries during IND review.
3. Faster Study Start-Up and Execution
Outsourced Bioanalysis Services reduce study initiation timelines by leveraging established SOPs and validated workflows.
Typical acceleration areas:
- Rapid feasibility assessment
- Pre-existing validated bioanalytical platforms
- Immediate access to control matrices
- Dedicated project management
For startups racing toward first-in-human trials, shaving even 3–6 months off development is transformational.
4: How Outsourced Bioanalysis Services Improve Capital Efficiency
They convert fixed capital expenses into variable operational costs.
Instead of investing heavily in infrastructure, startups leverage:
- Cost-effective bioanalytical services
- Transparent evaluation of bioanalytical testing services cost
- Flexible capacity models
This aligns with lean operational models and investor expectations.
Instead of investing millions in:
- Triple quadrupole mass spectrometers
- Cleanroom facilities
- Data integrity systems
Startups can allocate capital toward:
- Clinical trial execution
- Manufacturing scale-up
- Pipeline expansion
This lean operational model aligns with investor expectations for capital efficiency.
5: Risk Mitigation Through Experienced Outsourced Bioanalysis Services
Experienced CRO partners reduce technical and regulatory risk through proactive oversight.
Risk reduction mechanisms include:
- Early gap analysis
- Stability trend monitoring
- Pre-IND data readiness reviews
- Mock audits
Startups benefit from teams who have supported:
- Multiple IND submissions
- FDA inspections
- Global regulatory audits
Experienced partners support:
- Managing bioanalytical CRO projects
- Structured bioanalytical method transfer
- AI-supported analytics via AI in bioanalysis
- Collaboration with bioanalytical CRO for AI drug discovery
Integrated systems ensure audit-ready data integrity.
This institutional experience dramatically reduces compliance-related delays.
6: Integrated Data Integrity & Quality Systems
Regulatory-compliant data integrity systems ensure that bioanalytical results are submission-ready.
Modern outsourced labs implement:
- 21 CFR Part 11-compliant systems
- Validated LIMS platforms
- Audit trails and secure backups
- Independent QA oversight
For startups, this avoids costly remediation later.
Scalable partners provide:
- High-throughput bioanalysis
- Seamless progression from discovery vs regulated bioanalysis
- Integrated chemistry through integrated chemistry and bioanalytical CRO
Continuity avoids disruptions and eliminates re-validation delays.
7: Case-Based Example: Timeline Impact Comparison
| Development Phase | In-House Setup | Outsourced Bioanalysis Services |
|---|---|---|
| Lab Establishment | 12 months | 0 months |
| Method Development | 4–6 months | 2–3 months |
| Validation | 3 months | 1–2 months |
| IND Submission Readiness | 18–24 months total | 9–12 months total |
While actual timelines vary by molecule type, outsourcing often reduces early development time by 30–50%.
Startups should evaluate:
- Track record in bioanalytical CRO services for PK and TK
- Experience as a bioanalytical CRO for drug discovery
- Strength in virtual bioanalytical strategy
- Proven expertise in bioanalytical laboratory services
8: Scalability: Supporting Growth from Preclinical to Phase I
Outsourced Bioanalysis Services enable seamless scalability from preclinical development to Phase I clinical trials without operational disruption.
For startups, growth often means rapidly increasing study complexity, sample volume, regulatory expectations, and data reporting requirements. A scalable outsourcing partner ensures this transition happens smoothly—without changing vendors or rebuilding analytical strategies mid-program.
Key Advantages of Scalable Outsourced Bioanalysis Services
1. Capacity for Increased Sample Volumes
As studies progress, sample numbers can multiply significantly.
Phase I trials may involve hundreds to thousands of samples compared to smaller preclinical studies. Established bioanalytical laboratories offer:
- High-throughput LC-MS/MS platforms
- Validated batch processing workflows
- Automated sample preparation systems
- Redundant instrumentation to prevent downtime
This ensures timelines are maintained even as volume expands.
2. Dedicated Clinical Bioanalysis Teams
Clinical-phase studies require specialized oversight and documentation standards.
Scalable partners provide:
- Teams trained in GCP-aligned clinical bioanalysis
- Structured communication with clinical operations teams
- Protocol-specific analytical planning
- Real-time data tracking and reporting
This dedicated structure minimizes errors and supports tight clinical timelines.
3. Global Sample Logistics Support
Phase I trials often involve multi-site sample collection and international logistics.
Experienced outsourced providers manage:
- Cold-chain shipping coordination
- Chain-of-custody documentation
- Time-sensitive plasma/serum handling
- Import/export regulatory compliance
This logistical expertise reduces risk of sample degradation and protocol deviations.
4. Biomarker Integration
As programs mature, pharmacodynamic and biomarker endpoints become critical.
Scalable bioanalytical services integrate:
- PK and biomarker assays
- Exploratory translational endpoints
- Hybrid LBA–LC-MS approaches
- Multi-analyte panels
This integrated strategy enhances data interpretation and strengthens early clinical decision-making.
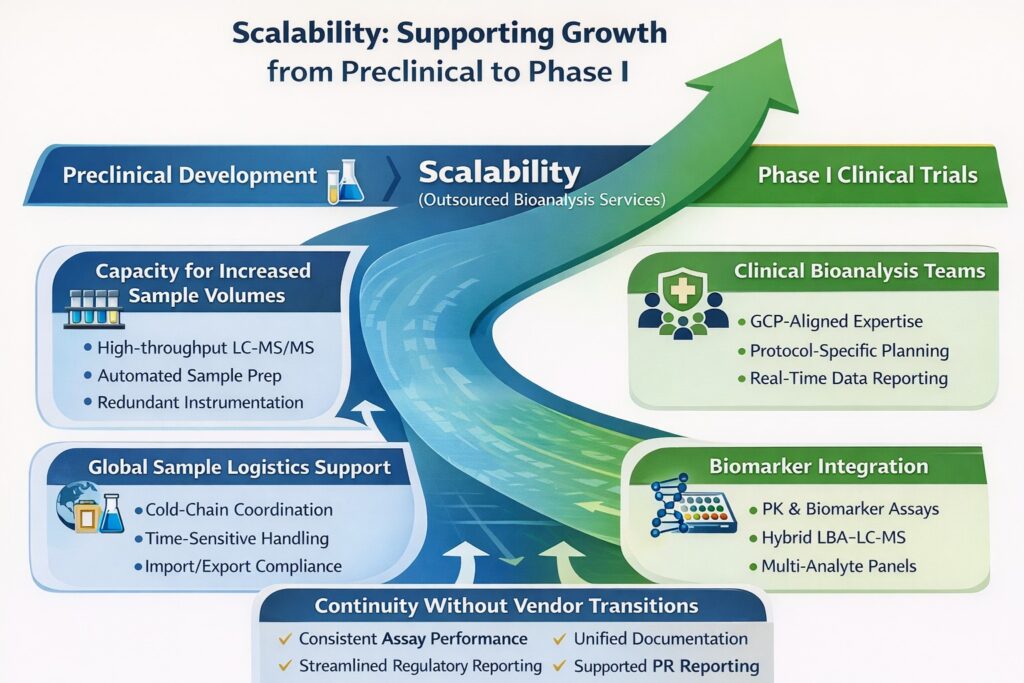
9: Continuity Without Vendor Transitions
One of the most important advantages of scalable Outsourced Bioanalysis Services is continuity.
Instead of transitioning between separate preclinical and clinical vendors—risking data inconsistencies and method transfer delays—startups benefit from:
- Consistent assay performance
- Unified documentation standards
- Historical method knowledge retention
- Streamlined regulatory reporting
Continuity reduces technical risk and preserves momentum as programs move toward first-in-human studies.
10: Why Specialized Bioanalytical CROs Are Critical for Complex Molecules
Complex modalities require advanced analytical capabilities not easily replicated in-house.
This includes:
- Peptides and cyclic peptides
- Oligonucleotides
- Antibody-drug conjugates
- Highly potent compounds
Advanced LC-MS/MS and HRMS platforms, combined with experienced scientists, are essential for reliable quantification and regulatory acceptance.
11: How Outsourced Bioanalysis Services Improve Investor Confidence
Faster milestones and reduced technical risk increase valuation and funding success.
Investors assess:
- IND timeline feasibility
- Data robustness
- Regulatory preparedness
- Operational efficiency
Partnering with a reputable bioanalytical provider signals structured, risk-aware execution.
12: Choosing the Right Outsourced Bioanalysis Services Partner
The right partner combines scientific depth, regulatory credibility, and operational agility.
Key evaluation criteria:
- Proven IND support track record
- Experience across molecule classes
- Transparent communication
- QA independence
- Scalable infrastructure
Startups should seek partners that operate as strategic collaborators—not transactional vendors.
13: Why ResolveMass Laboratories Inc. Is Positioned to Support Startup Speed-to-Clinic
ResolveMass Laboratories Inc. specializes in advanced LC-MS/MS-based bioanalysis, supporting biotech innovators from preclinical through early clinical development.
ResolveMass bioanalytical services overview highlights capabilities spanning:
- Small molecules
- Complex modalities
- Regulatory-compliant validation
- IND-support documentation
As a specialized bioanalytical CRO, ResolveMass integrates scientific rigor with operational agility to help startups reach first-in-human studies faster.
Core strengths include:
- Deep expertise in small molecules and complex modalities
- Regulatory-aligned method development and validation
- Robust quality systems
- IND-supporting documentation packages
- Scientific leadership with extensive regulatory experience
By integrating scientific rigor with operational agility, ResolveMass helps startups move from discovery to clinic efficiently and compliantly.
Conclusion:
Outsourced Bioanalysis Services significantly improve speed-to-clinic for startups by eliminating infrastructure delays, reducing regulatory risk, and enabling capital-efficient development.
For emerging biotech companies, outsourcing is not merely a cost decision—it is a strategic pathway to:
- Faster IND submissions
- Stronger regulatory positioning
- Reduced technical risk
- Improved investor confidence
- Accelerated entry into Phase I trials
Outsourced Bioanalysis Services significantly improve speed-to-clinic by:
- Eliminating infrastructure delays
- Reducing regulatory risk
- Enhancing capital efficiency
- Accelerating PK, TK, biomarker, and clinical bioanalysis
For biotech startups, strategic outsource bioanalysis for biotech startups partnerships are not just operational decisions—they are competitive advantages.
In today’s high-pressure biotech landscape, leveraging expert bioanalytical services can mean the difference between delayed development and successful Phase I entry.
Frequently Asked Questions:
They eliminate the 12–24 month delay required to:
-Purchase and qualify mass spectrometry equipment
-Hire and train specialized scientists
-Establish GLP-compliant infrastructure
-Develop and validate analytical methods
Instead, startups gain immediate access to operational laboratories, which can reduce early development timelines by 30–50%.
Outsourced bioanalysis supports:
-Preclinical pharmacokinetics (PK)
-Toxicokinetic (TK) studies
-IND-enabling studies
-Biomarker bioanalysis
-Immunogenicity (ADA) testing
-Phase I clinical bioanalysis
-Stability testing
-Method transfer
Most programs can be supported from discovery through early clinical development.
Yes. Established bioanalytical CROs operate under:
-GLP-compliant systems
-21 CFR Part 11 data integrity requirements
-ICH M10 Bioanalytical Method Validation guidance
-FDA and Health Canada expectations
Regulatory-aligned documentation significantly reduces the risk of IND deficiencies.
For startups, yes.
Building an internal bioanalytical lab requires:
-Multi-million-dollar capital investment
-Ongoing equipment maintenance
-Quality assurance infrastructure
-Recruitment and retention of highly specialized scientists
Outsourcing converts fixed capital expenditures into variable operational costs, improving capital efficiency and investor confidence.
Experienced CRO partners can support:
-Small molecules
-Peptides and cyclic peptides
-Oligonucleotides
-Antibody-drug conjugates (ADCs)
-Cell and gene therapies
-Highly potent compounds
Advanced LC-MS/MS and hybrid assay platforms allow accurate quantification of complex modalities.
Outsourced providers reduce risk by:
-Anticipating regulatory questions early
-Conducting stability and matrix effect assessments
-Performing internal QA reviews
-Supporting mock audits
-Preparing submission-ready validation reports
Their experience across multiple IND filings helps prevent costly rework.
Reference
- Andrew Dolman, Xiaojing Yu. Data Driven CRO Benchmarking for Biomarker Analysis.https://www.tandfonline.com/doi/abs/10.4155/bio-2020-0164
- Xiaojing Yu & Arkady I Gusev. CRO Benchmarking for Clinical Biomarker Analysis Outsourcing.https://www.tandfonline.com/doi/full/10.4155/bio-2019-0123
- Manufacturing of Cell-Based Therapies.https://alliancerm.org/wp-content/uploads/2025/08/PROJECT-A-CELL-V2.pdf#page=148


