
Introduction:
Outsourcing generic drug development Canada has become a strategic approach for pharmaceutical companies looking to accelerate market entry, reduce operational costs, and leverage specialized scientific expertise. As regulatory expectations and analytical complexities increase, many pharma and biotech companies choose to collaborate with experienced Contract Development and Manufacturing Organizations (CDMOs).
In Canada, CDMOs provide integrated services such as analytical method development, bioanalysis, impurity testing, stability studies, and regulatory support. These services are often supported by strong scientific planning through a bioanalytical strategy in drug development that ensures accurate data generation and regulatory compliance.
This article explains why outsourcing generic drug development in Canada is growing rapidly and how CDMO partnerships help companies successfully bring generic medicines to market.
Share via:
Summary:
- Outsourcing generic drug development Canada allows pharmaceutical companies to reduce development timelines and costs.
- Canadian CDMOs (Contract Development and Manufacturing Organizations) offer regulatory expertise aligned with Health Canada requirements.
- Outsourcing provides access to specialized analytical technologies such as bioanalysis services in Canada and impurity testing.
- Pharma companies benefit from flexible scalability without building internal infrastructure.
- Experienced CDMOs help streamline ANDA/NDS submissions and regulatory approvals in Canada.
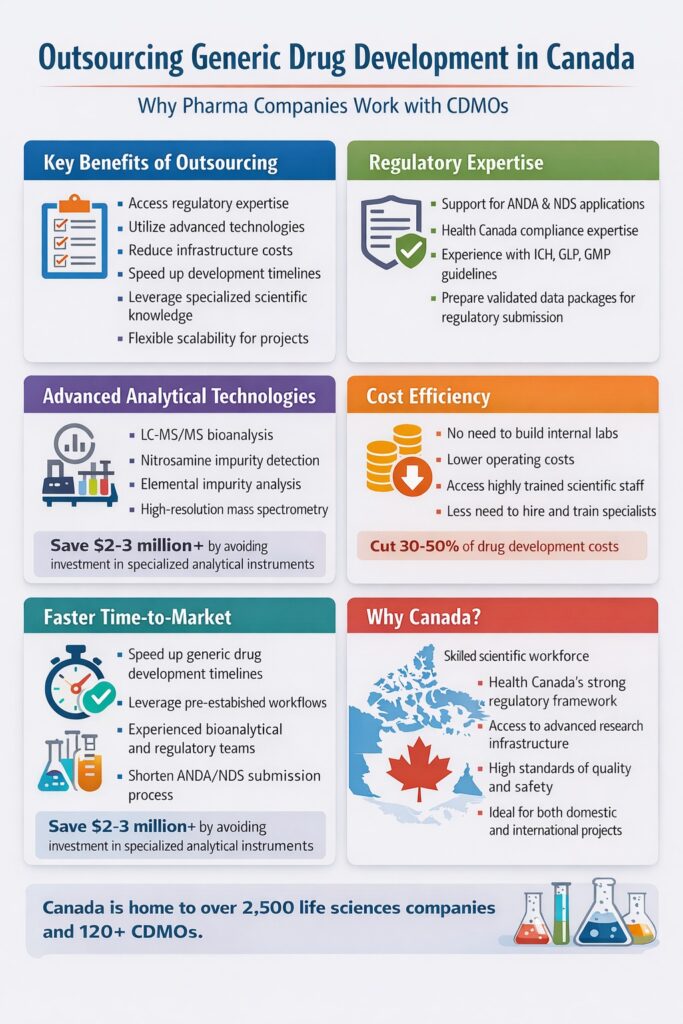
1: Why Pharma Companies Prefer Outsourcing Generic Drug Development Canada
Pharmaceutical companies outsource drug development in Canada primarily to gain regulatory expertise, reduce infrastructure costs, and access specialized analytical technologies.
Developing generic drugs requires extensive analytical work, regulatory documentation, and compliance with Health Canada guidelines. Building this capability internally can be expensive and time-consuming.
Companies often collaborate with specialized partners such as a generic pharmaceutical CDMO in Canada that provides end-to-end development support.
Key Advantages of Outsourcing
- Access to experienced scientists and analytical experts
- Faster development timelines
- Reduced capital investment in laboratories and equipment
- Improved regulatory compliance
- Flexible project scalability
Many companies find that outsourcing drug development Canada allows them to remain competitive while ensuring high-quality development processes.
2: Outsourcing Generic Drug Development Canada for Regulatory Expertise
One of the main reasons companies pursue outsourcing generic drug development Canada is to navigate complex regulatory requirements efficiently.
Generic drug approvals in Canada require compliance with multiple regulatory frameworks, including:
- Health Canada guidance documents
- Good Laboratory Practices (GLP)
- Bioanalytical method validation standards
- Stability testing and impurity profiling
Experienced CDMOs already operate within these regulatory frameworks and understand the documentation required for successful submissions.
These organizations also support analytical method development for generic drugs to generate validated data required for regulatory submissions.
Regulatory Responsibilities Handled by CDMOs
| Development Area | CDMO Role |
|---|---|
| Method Development | Create validated analytical methods |
| Bioanalysis | Quantify drugs and metabolites in biological samples |
| Stability Studies | Evaluate product stability under various conditions |
| Impurity Testing | Identify and quantify impurities including nitrosamines |
| Regulatory Documentation | Prepare reports for Health Canada submission |
Through outsourcing drug development Canada, pharmaceutical companies significantly reduce regulatory risk.
3: Access to Advanced Analytical Technologies Through Outsourcing Generic Drug Development Canada
Outsourcing drug development Canada provides companies access to specialized technologies that are expensive to build internally.
Modern drug development requires sophisticated analytical platforms such as:
- LC-MS/MS bioanalysis
- High-resolution mass spectrometry
- Nitrosamine impurity detection
- Elemental impurity analysis
- Extractables and leachables studies
These technologies are often managed by mass spectrometry experts in drug development who specialize in high-sensitivity analytical testing.
In addition, complex molecules like peptides require specialized testing such as peptide characterization in drug development and peptide testing services to ensure accurate structural and impurity analysis.
CDMOs that specialize in analytical services often maintain cutting-edge instrumentation and experienced scientists capable of developing highly sensitive methods.
4: Cost Efficiency of Outsourcing Generic Drug Development Canada
Outsourcing drug development Canada reduces capital expenditure by eliminating the need to build internal laboratories and hire specialized teams.
Establishing a complete drug development infrastructure requires:
- Mass spectrometry instruments
- Analytical laboratories
- Highly trained scientific staff
- Regulatory compliance systems
These investments can cost millions of dollars.
Many pharmaceutical companies also utilize outsourced chemistry in drug discovery services to further reduce internal resource requirements.
Cost Comparison: In-House vs Outsourced Development
| Factor | In-House Development | Outsourced Development |
|---|---|---|
| Infrastructure cost | Very high | Minimal |
| Staffing requirements | Large internal team | Managed by CDMO |
| Regulatory expertise | Must be built internally | Already available |
| Project scalability | Limited | Flexible |
This cost advantage is a major driver behind outsourcing generic drug development Canada.
5: Faster Time-to-Market Through Outsourcing Generic Drug Development Canada
Working with experienced CDMOs accelerates timelines for drug development and regulatory approval.
Speed is critical in the generic drug industry because the first companies to launch generics often capture the largest market share.
CDMOs streamline development by offering:
- Established workflows
- Experienced regulatory teams
- Validated analytical platforms
- Efficient project management
Many development programs also include reverse engineering for development of generic drugs to understand reference listed drug formulations and accelerate development.
These factors significantly reduce development timelines.
6: Scientific Expertise Available Through Outsourcing Generic Drug Development Canada
Another major benefit of outsourcing drug development Canada is access to multidisciplinary scientific expertise.
Generic drug development requires expertise in multiple areas including:
- Analytical chemistry
- Bioanalysis
- Pharmaceutical formulation
- Regulatory science
- Mass spectrometry
Partnering with experienced CDMOs such as those offering CDMO for generic drug development in Canada ensures that pharmaceutical companies have access to highly specialized scientific teams.
Building this expertise internally can take years. Partnering with a CDMO provides immediate access to experienced professionals who understand the technical challenges of generic drug development.
7: How to Choose the Right Partner for Outsourcing Generic Drug Development Canada
Selecting the right CDMO is essential for successful outsourcing drug development Canada.
Pharmaceutical companies should evaluate potential partners based on the following criteria:
Key Factors to Consider
- Regulatory experience in Canada
- Advanced analytical capabilities
- Track record with generic drug projects
- Data integrity and quality systems
- Communication and project management
Many companies choose experienced partners providing CDMO services for generic projects in Canada to ensure efficient project execution.
Questions to Ask a CDMO
- Do they have experience supporting Health Canada submissions?
- Do they specialize in bioanalysis and mass spectrometry?
- Can they handle complex impurity and nitrosamine testing?
- Do they provide flexible project support?
Choosing an experienced CDMO significantly improves the success rate of outsourced projects.
8: Role of Specialized Analytical CDMOs in Outsourcing Generic Drug Development Canada
Specialized analytical CDMOs play a critical role in outsourcing drug development Canada by providing high-quality data required for regulatory submissions.
These organizations focus on:
- Analytical method development and validation
- Bioanalytical testing for pharmacokinetic studies
- Impurity profiling and stability studies
- Regulatory documentation support
Because of their focused expertise, specialized CDMOs often deliver more reliable and accurate results compared to generalized service providers.
9: Why Canada Is an Attractive Location for Outsourced Generic Drug Development
Canada has become a preferred location for outsourcing drug development due to its strong regulatory framework and scientific ecosystem.
Key advantages include:
- Highly trained scientific workforce
- Strong regulatory oversight by Health Canada
- Advanced research infrastructure
- Growing biotech and pharmaceutical sector
These factors make outsourcing generic drug development Canada an attractive option for both domestic and international pharmaceutical companies.
Conclusion
Outsourcing drug development Canada offers pharmaceutical companies a strategic advantage by providing access to regulatory expertise, advanced analytical technologies, and cost-efficient development pathways.
By partnering with experienced CDMOs, companies can accelerate drug development timelines, reduce operational costs, and ensure compliance with Health Canada regulations.
As the complexity of generic drug development continues to increase, outsourcing drug development Canada will remain a key strategy for pharmaceutical companies aiming to bring high-quality generic medicines to market efficiently.
Frequently Asked Questions:
Outsourcing generic drug development Canada refers to partnering with specialized CDMOs to handle analytical testing, bioanalysis, regulatory support, and development activities for generic drugs.
Pharmaceutical companies outsource these services to access advanced scientific expertise, reduce development costs, and accelerate regulatory approvals under Health Canada guidelines.
Outsourcing generic drug development Canada typically includes analytical testing, bioanalysis, regulatory support, and method development.
Common services include:
• Analytical method development and validation
• Bioanalytical testing for pharmacokinetic studies
• Impurity and nitrosamine testing
• Stability studies
• Reverse engineering of reference drugs
• Regulatory documentation support
Outsourcing generic drug development Canada accelerates time-to-market by using established workflows, experienced regulatory teams, and advanced analytical technologies.
CDMOs already have validated methods, regulatory experience, and trained scientists, which helps reduce development delays and speed up regulatory submissions.
Generic drugs in Canada must comply with Health Canada regulatory requirements, including bioequivalence data and validated analytical methods.
Companies must provide:
• Bioanalytical data supporting pharmacokinetics
• Stability studies
• Impurity and safety data
• Validated analytical methods
• Complete regulatory documentation
Experienced CDMOs help ensure these requirements are met.
Reference
- Investigating Outsourcing. https://www.biopharminternational.com/view/investigating-outsourcing
- The Rise of Biopharmaceutical Outsourcing to Indian CDMOs.https://eu-assets.contentstack.com/v3/assets/blt0a48a1f3edca9eb0/bltbf57a24293b0039e/658c2acb4fc588040aed1c7d/BPI_A_141206SUPAR06_O.pdf
- The Outsourcing Success Story.https://www.pharmtech.com/view/the-outsourcing-success-story
- Selection and Oversight of Domestic and International CMOs.https://eu-assets.contentstack.com/v3/assets/blt0a48a1f3edca9eb0/blt02414e2aa377513b/658c3b00e5473f040a510009/BPI_A_090703AR04_O_78234a.pdf


