
Introduction:
Peptide Characterization for ANDA submission is a critical regulatory requirement to demonstrate that a generic peptide drug is equivalent to its reference listed drug (RLD) in terms of structure, purity, and quality.
With the growing number of peptide-based therapeutics entering the market, regulatory agencies such as the FDA demand rigorous analytical characterization to ensure therapeutic equivalence, safety, and efficacy. Unlike small molecules, peptides exhibit structural complexity, sequence variability, and susceptibility to degradation—making their characterization far more challenging.
To understand regulatory expectations in detail, refer to FDA peptide sameness study requirements and sameness evaluation in ANDA.
Summary:
- Peptide Characterization for ANDA submission ensures that a generic peptide drug is identical in structure, purity, and performance to the reference listed drug (RLD).
- Regulatory agencies require comprehensive analytical evidence aligned with active ingredient sameness in ANDA
- Orthogonal analytical techniques (LC-MS, HRMS, NMR, peptide mapping) are essential to demonstrate sameness.
- Impurity profiling and degradation studies are critical for safety and compliance.
- A strong strategy aligned with analytical strategies for sameness study reduces regulatory risk
- Partnering with expert labs like ResolveMass Laboratories Inc. ensures regulatory-ready data and compliance confidence.
1: What does FDA expect in Peptide Characterization for ANDA?
The FDA expects detailed analytical evidence proving structural sameness, impurity equivalence, and physicochemical comparability with the reference product.
For a deeper regulatory framework, explore peptide sameness study for ANDA.
Key Expectations Include:
- Primary Structure Confirmation
- Higher-Order Structure Analysis
- Impurity Profiling
- Physicochemical Property Evaluation
- Comparative Analytical Assessment
Regulatory Focus Areas:
| Characterization Aspect | FDA Expectation |
|---|---|
| Identity | Exact amino acid sequence |
| Purity | Comparable impurity profile |
| Structure | Similar folding and conformation |
| Stability | Matching degradation pathways |
| Functionality | Comparable biological activity |
2: Primary Structure Confirmation
Primary structure confirmation ensures that the amino acid sequence of the generic peptide exactly matches the reference listed drug (RLD). This step provides definitive proof of molecular identity and is a core requirement in Peptide Characterization for ANDA submission.
This aligns with requirements discussed in active ingredient sameness in ANDA and is fundamental to ANDA success.
Techniques Used:
A combination of orthogonal, high-sensitivity analytical techniques is used to confirm sequence accuracy:
- LC-MS/MS (Peptide Sequencing)
Enables precise identification of amino acid sequences through fragmentation patterns. It is the most widely used and regulatory-accepted method for peptide sequencing. - Edman Degradation (where applicable)
A classical method used for stepwise identification of N-terminal amino acids, particularly useful for shorter peptides or confirmation studies. - High-Resolution Mass Spectrometry (HRMS)
Provides accurate molecular weight determination and detects even minimal mass differences, ensuring high-confidence structural confirmation.
What is Evaluated?
Primary structure confirmation focuses on verifying critical molecular attributes:
- Amino Acid Sequence
Confirms the exact order of amino acids and detects any substitutions, deletions, or insertions. - Terminal Modifications
Identifies structural features such as C-terminal amidation or N-terminal acetylation, which can significantly impact biological activity and stability. - Molecular Weight Accuracy
Ensures the observed mass matches the theoretical value within tight tolerance limits, confirming structural integrity.
Even minor sequence deviations can lead to regulatory rejection, making this step foundational.
3: Peptide Mapping: The Cornerstone of Comparability
Peptide mapping confirms sequence integrity and detects even subtle structural variations by combining enzymatic digestion with high-resolution chromatographic and mass spectrometric analysis. It is a critical pillar of Peptide Characterization for ANDA submission, enabling direct comparison between the generic peptide and the reference listed drug (RLD).
For advanced strategies, refer to analytical strategies for sameness study and orthogonal analytical techniques for ANDA sameness evaluation.
Why It Matters:
Peptide mapping provides high-resolution insight into structural integrity and consistency:
- Identifies Sequence Mismatches
Detects even single amino acid substitutions, deletions, or insertions that could impact drug performance. - Detects Post-Translational Modifications (PTMs)
Reveals critical modifications such as oxidation, deamidation, or cyclization that may affect stability and efficacy. - Ensures Batch-to-Batch Consistency
Confirms reproducibility across manufacturing batches—an essential requirement for regulatory approval.
Workflow:
The peptide mapping process follows a well-defined analytical sequence:
- Enzymatic Digestion (e.g., Trypsin)
The peptide is cleaved into predictable fragments based on enzyme specificity. - LC-MS Analysis
The resulting fragments are separated using liquid chromatography and analyzed via mass spectrometry for precise identification. - Comparison with Reference Profile
The generated peptide map is directly compared with that of the RLD to assess structural sameness.
Key Outputs:
Peptide mapping generates multiple layers of analytical data that support regulatory evaluation:
- Retention Time Alignment
Confirms chromatographic consistency between test and reference products. - Fragment Mass Matching
Ensures each peptide fragment corresponds to the expected mass and sequence. - Peak Purity Assessment
Verifies that each chromatographic peak represents a single, well-defined component without co-eluting impurities.
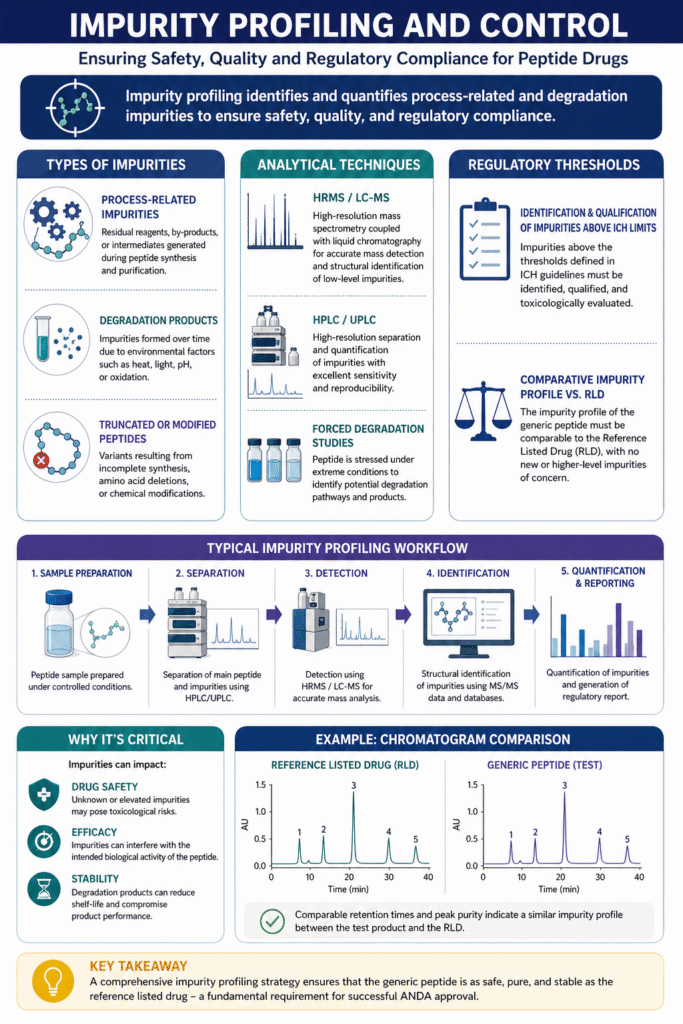
4: Impurity Profiling and Control
Impurity profiling identifies and quantifies process-related and degradation impurities to ensure safety, quality, and regulatory compliance. It is a mandatory component of Peptide Characterization for ANDA submission, as regulatory agencies require a clear understanding of all impurities present in the generic peptide compared to the reference listed drug (RLD).
Common pitfalls are explained in peptide sameness study deficiencies, which highlights regulatory risks.
Types of Impurities:
A comprehensive impurity profile includes multiple categories:
- Process-Related Impurities
Residual reagents, intermediates, or by-products formed during peptide synthesis and purification. - Degradation Products
Impurities formed over time due to environmental factors such as heat, light, pH, or oxidation. - Truncated or Modified Peptides
Variants resulting from incomplete synthesis, amino acid deletions, or chemical modifications.
Analytical Techniques:
Robust impurity profiling relies on advanced, high-sensitivity analytical methods:
- HRMS / LC-MS
Enables accurate mass detection and structural identification of low-level impurities. - HPLC / UPLC
Provides high-resolution separation and quantification of impurities. - Forced Degradation Studies
Intentionally stress the peptide under extreme conditions to identify potential degradation pathways and products.
Regulatory Thresholds:
Regulatory expectations are aligned with ICH guidelines and require:
- Identification and Qualification of Impurities Above ICH Limits
Any impurity exceeding specified thresholds must be structurally identified and toxicologically evaluated. - Comparative Impurity Profile vs. RLD
The impurity profile of the generic peptide must be comparable to that of the reference product, with no new or higher-level impurities of concern.
Why It’s Critical:
Impurities are closely scrutinized because they directly influence product quality and patient safety:
- Drug Safety
Unknown or elevated impurities may pose toxicological risks. - Efficacy
Impurities can interfere with the intended biological activity of the peptide. - Stability
Degradation products can reduce shelf-life and compromise product performance.
5: Higher-Order Structure (HOS) Analysis
Higher-order structure (HOS) analysis ensures that the peptide’s three-dimensional conformation matches the reference listed drug (RLD), confirming structural and functional equivalence. In Peptide Characterization for ANDA submission, this step is essential because identical amino acid sequences do not always guarantee identical biological behavior.
This is a key requirement under sameness evaluation of synthetic peptides for ANDA.
Techniques Used:
A combination of orthogonal spectroscopic techniques is used to fully evaluate peptide conformation:
- 1D & 2D NMR Spectroscopy
Provides detailed insights into atomic-level structure, molecular dynamics, and spatial arrangement of the peptide. - Circular Dichroism (CD)
Assesses secondary structure content by measuring differences in absorption of circularly polarized light. - FTIR Spectroscopy
Identifies characteristic vibrational modes associated with secondary structural elements such as α-helices and β-sheets.
What is Evaluated?
HOS analysis focuses on confirming structural integrity beyond the primary sequence:
- Secondary Structure (α-Helix, β-Sheet)
Determines the proportion and arrangement of structural motifs critical for peptide function. - Folding Patterns
Evaluates how the peptide folds in solution, which directly influences receptor binding and activity. - Conformational Stability
Assesses how stable the structure remains under different environmental conditions (pH, temperature, solvent).
Even if the sequence is identical, structural differences can affect biological activity, making HOS analysis essential.
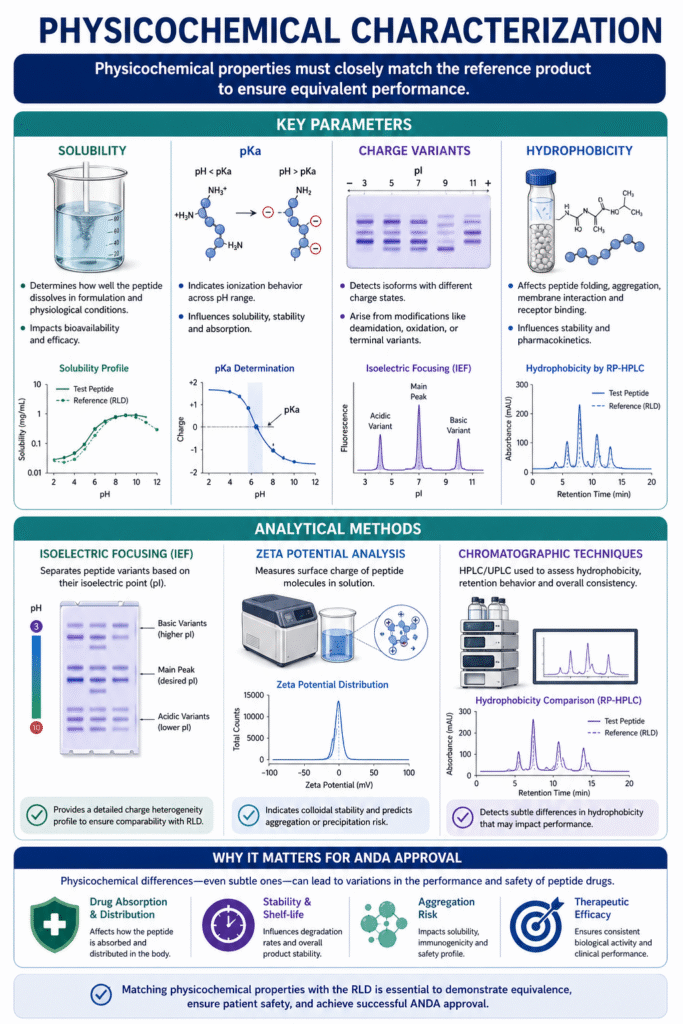
6: Physicochemical Characterization
Physicochemical characterization ensures that critical properties such as solubility, charge, and hydrophobicity of the generic peptide closely match the reference listed drug (RLD), enabling equivalent in vivo and in vitro performance. It is a key component of Peptide Characterization for ANDA submission, bridging structural confirmation with functional behavior.
These evaluations are critical for meeting expectations outlined in peptide sameness study services in United States and peptide sameness study services in Canada.
Key Parameters:
A robust physicochemical profile includes evaluation of the following attributes:
- Hydrophobicity
Impacts peptide folding, aggregation potential, and interaction with biological membranes or receptors. - Solubility
Determines how well the peptide dissolves under physiological and formulation conditions, directly affecting bioavailability. - pKa
Defines ionization behavior across pH ranges, influencing stability, solubility, and absorption. - Charge Variants
Identifies isoforms with different charge states, often arising from modifications like deamidation or terminal variations.
Methods:
Multiple orthogonal techniques are applied to accurately assess physicochemical properties:
- Isoelectric Focusing (IEF)
Separates peptide variants based on their isoelectric point (pI), enabling detection of charge heterogeneity. - Zeta Potential Analysis
Measures surface charge and predicts colloidal stability in solution. - Chromatographic Techniques (HPLC/UPLC)
Used to evaluate hydrophobicity, retention behavior, and overall physicochemical consistency.
Why it is Critical for ANDA Approval
Physicochemical differences—even subtle ones—can lead to variations in:
- Drug absorption and distribution
- Stability and shelf-life
- Aggregation or degradation behavior
Regulatory agencies expect close alignment of these properties with the RLD, supported by validated analytical data.
In Peptide Characterization for ANDA submission, physicochemical characterization ensures that the generic peptide not only matches structurally but also performs equivalently under real-world conditions, reinforcing therapeutic consistency and regulatory acceptance.
7: Orthogonal Analytical Techniques
Orthogonal analytical techniques use multiple, independent methods to evaluate the same attribute, providing complementary evidence to confirm peptide sameness. In Peptide Characterization for ANDA submission, these approaches are essential to build a robust, defensible analytical package that satisfies regulatory scrutiny.
Learn more from orthogonal analytical techniques for ANDA sameness evaluation.
Why Use Orthogonal Methods?
Using orthogonal techniques strengthens the reliability and credibility of analytical results:
- Reduce Analytical Bias
Different techniques rely on distinct scientific principles, minimizing the risk of method-specific errors or blind spots. - Improve Confidence in Results
Consistent findings across independent methods provide strong confirmation of structural and physicochemical sameness. - Meet Regulatory Expectations
Regulatory agencies expect multi-technique validation to ensure comprehensive characterization, especially for complex molecules like peptides.
Examples:
| Attribute | Technique 1 | Technique 2 |
|---|---|---|
| Identity | LC-MS | NMR |
| Purity | HPLC | CE |
| Structure | CD | FTIR |
8: Stability and Degradation Studies
Stability and degradation studies demonstrate that the generic peptide behaves similarly to the reference listed drug (RLD) under various stress conditions, confirming comparable stability profiles. This is a vital requirement in Peptide Characterization for ANDA submission, as it ensures the product maintains quality, safety, and efficacy throughout its shelf life.
These are essential components of peptide sameness study for ANDA.
Key Studies:
A comprehensive stability assessment includes multiple stress conditions:
- Thermal Degradation
Evaluates the impact of elevated temperatures on peptide integrity and degradation rate. - Oxidative Stress
Assesses susceptibility to oxidation (e.g., methionine or tryptophan oxidation), which can alter peptide activity. - pH Stability
Determines how the peptide behaves across a range of pH conditions, simulating physiological and formulation environments.
Outcomes:
These studies generate critical data required for regulatory evaluation:
- Degradation Pathways
Identifies how the peptide breaks down under stress, including specific chemical modifications. - Impurity Formation Trends
Tracks the formation and progression of degradation-related impurities over time. - Shelf-Life Prediction
Supports determination of product expiry based on stability data and degradation kinetics.
9: Comparative Analytical Assessment
Comparative analytical assessment directly evaluates whether the generic peptide is highly similar to the reference listed drug (RLD) through side-by-side testing and data-driven comparison. In Peptide Characterization for ANDA submission, this step integrates all analytical evidence into a clear demonstration of sameness.
Case-specific examples include:
Key Components:
A robust comparative assessment includes:
- Side-by-Side Testing
The generic peptide and RLD are analyzed under identical experimental conditions to ensure direct comparability. - Statistical Comparison
Quantitative tools are used to evaluate similarity, including variance analysis, similarity factors, and confidence intervals. - Acceptance Criteria Alignment
Predefined regulatory criteria are applied to confirm that observed differences (if any) fall within acceptable limits.
Deliverables:
The outcome of comparative assessment is a comprehensive dataset that supports regulatory submission:
- Overlay Chromatograms
Visual comparison of retention times and peak profiles to confirm similarity in impurity and main peak distribution. - Spectral Comparisons
Direct comparison of mass spectra, NMR spectra, or CD profiles to validate structural equivalence. - Quantitative Similarity Metrics
Statistical outputs that objectively demonstrate equivalence between the test product and the RLD.
10: Common Deficiencies in ANDA Submissions
Most ANDA rejections occur due to incomplete or insufficient peptide characterization data, particularly when critical analytical evidence is missing or inadequately justified. In the context of Peptide Characterization for ANDA submission, even small gaps can raise major regulatory concerns.
Avoid common issues by reviewing peptide sameness study deficiencies.
Frequent Issues:
Regulatory agencies commonly identify the following deficiencies:
- Inadequate Impurity Identification
Failure to fully characterize and qualify impurities above ICH thresholds, or lack of structural elucidation for unknown peaks. - Lack of Orthogonal Methods
Over-reliance on a single analytical technique without complementary methods to confirm results. - Poor Peptide Mapping Resolution
Insufficient chromatographic separation leading to co-eluting peaks and unclear sequence confirmation. - Missing Higher-Order Structure (HOS) Data
Absence of conformational analysis (e.g., NMR, CD), which is critical for demonstrating functional equivalence.
Regulatory Impact
These deficiencies can result in:
- Information Requests (IRs) or Complete Response Letters (CRLs)
- Extended review timelines
- Increased development costs
- Potential ANDA rejection
11: Why expert Analytical Partner Matter
Working with an experienced analytical partner ensures that your peptide characterization strategy meets regulatory expectations, delivers high-quality data, and accelerates ANDA approval timelines. In Peptide Characterization for ANDA submission, the complexity of analytical requirements makes expert support not just beneficial—but essential.
Explore real-world project insights:
Advantages:
Partnering with a specialized laboratory provides multiple strategic benefits:
- Regulatory Expertise
Experienced teams understand FDA expectations, ICH guidelines, and common deficiencies—ensuring your data package is submission-ready from the start. - Advanced Instrumentation (HRMS, NMR)
Access to state-of-the-art technologies enables precise structural characterization, impurity profiling, and conformational analysis. - Customized Analytical Strategies
Tailored workflows are designed based on the specific peptide, reducing risk and improving efficiency. - Audit-Ready Documentation
Well-structured, compliant reports support smooth regulatory review and minimize queries or delays.
ResolveMass Laboratories Inc. specializes in comprehensive peptide characterization for ANDA submission, offering end-to-end analytical support aligned with FDA expectations.
Conclusion:
Peptide Characterization for ANDA submission is not just a regulatory requirement—it is the foundation for demonstrating equivalence, safety, and efficacy of generic peptide drugs.
A robust characterization strategy must include:
- Primary structure confirmation
- Peptide mapping
- Impurity profiling
- Higher-order structure analysis
- Orthogonal techniques
- Stability studies
Following a structured approach aligned with sameness evaluation in ANDA ensures regulatory success.
Partnering with experts like ResolveMass Laboratories Inc. ensures that your submission is scientifically sound, regulatory-compliant, and approval-ready.
Frequently Asked Questions:
Peptide Characterization for ANDA submission is the process of analytically proving that a generic peptide drug is structurally and functionally equivalent to the reference listed drug (RLD). It involves techniques like LC-MS, NMR, peptide mapping, and impurity profiling. Regulatory agencies require this data to ensure safety, efficacy, and quality. A well-executed characterization strategy is essential for successful ANDA approval.
Peptides are larger and structurally more complex than small molecules, with sequence variability and susceptibility to degradation. They can undergo modifications such as oxidation or deamidation, which impact function. Additionally, higher-order structure plays a key role in their biological activity. This complexity requires multiple orthogonal analytical techniques for accurate characterization.
Peptide mapping is a technique that breaks the peptide into smaller fragments using enzymes and analyzes them using LC-MS. It helps confirm sequence integrity, detect modifications, and compare the generic product with the RLD. This method acts as a molecular fingerprint, making it one of the most critical tools in ANDA submissions.
Impurity profiling identifies and quantifies all impurities present in the peptide, including degradation products and synthesis-related impurities. Regulatory agencies require impurities above certain thresholds to be identified and qualified. Any unknown or excessive impurity can lead to delays or rejection. Therefore, a robust impurity control strategy is essential.
Orthogonal techniques use different scientific principles to analyze the same attribute, such as LC-MS and NMR for identity confirmation. They reduce analytical bias and increase confidence in results. Regulatory authorities expect multiple independent methods to validate findings, especially for complex molecules like peptides.
Stability studies evaluate how the peptide behaves under stress conditions like heat, oxidation, and varying pH levels. They help identify degradation pathways, impurity trends, and shelf-life. These studies ensure that the generic product remains stable and effective throughout its lifecycle, matching the RLD.
Reference
- Wu L. Regulatory considerations for peptide therapeutics.https://books.rsc.org/books/edited-volume/801/chapter/540098
- Jois SD. Regulatory Issues for Peptide Drugs. InPeptide Therapeutics: Fundamentals of Design, Development, and Delivery 2022 Sep 27 (pp. 287-305). Cham: Springer International Publishing.https://link.springer.com/chapter/10.1007/978-3-031-04544-8_9
- Kuril AK, Saravanan K, Subbappa PK. Analytical considerations for characterization of generic peptide product: A regulatory insight. Analytical Biochemistry. 2024 Nov 1;694:115633.https://www.sciencedirect.com/science/article/pii/S0003269724001775
- Giri T, Sakharwade S, Subbappa P, Chinnakadoori SR, Sharma N. Regulatory Considerations in Synthetic Peptide Characterization: Techniques and Compliance. Separation Science Plus. 2025 Jun;8(6):e70057.https://analyticalsciencejournals.onlinelibrary.wiley.com/doi/abs/10.1002/sscp.70057
- De Groot AS, Roberts BJ, Mattei A, Lelias S, Boyle C, Martin WD. Immunogenicity risk assessment of synthetic peptide drugs and their impurities. Drug Discovery Today. 2023 Oct 1;28(10):103714.https://www.sciencedirect.com/science/article/pii/S1359644623002301


