Introduction
Peptide Mapping is a crucial process in proteomics and drug development, providing detailed insights into protein structures, functions, and therapeutic potential. Through accurate peptide sequence identification, researchers can decode the primary structure of peptides, identify biomarkers, and develop peptide-based therapeutics. At Resolvemass Laboratories, we specialize in leveraging advanced techniques to deliver precise Peptide Mapping and sequence identification services, supporting a wide range of scientific and industrial applications.
Explore our specialized solutions for complex molecular structures: Peptide-Oligonucleotide Conjugates (POCs) Synthesis Services
Share via:
The Importance of Peptide Sequence Identification
Peptides are short chains of amino acids and are integral to many biological processes and therapeutic strategies. Accurate identification of their sequences is vital for:
Protein Characterization
- Structural Analysis: Knowing the exact amino acid sequence helps in understanding protein structure and function. For example, identifying active sites or binding domains in proteins can reveal how they interact with other molecules.
- Functional Insights: The sequence provides clues about the biological role of the peptide and its impact on cellular processes.
Biomarker Discovery
- Disease Diagnosis: Identifying peptide biomarkers associated with specific diseases can aid in early diagnosis and prognosis. For example, certain peptides may be indicative of cancer or cardiovascular diseases.
- Therapeutic Targets: Peptides can serve as potential targets for new drugs or therapies. Identifying specific peptide sequences can lead to the development of targeted treatments.
Therapeutic Development
- Peptide Drugs: Designing drugs based on peptide sequences requires accurate sequencing to ensure efficacy and safety. This includes developing peptide-based vaccines, hormone replacements, and enzyme inhibitors.
- Formulation Optimization: Understanding peptide sequences helps in optimizing formulations to enhance stability, bioavailability, and activity.
Synthetic Biology
- Peptide Engineering: Engineering peptides with desired properties for applications in medicine, agriculture, or industry involves precise sequence knowledge to ensure that modifications yield the desired effects.
Emerging Trends in Peptide Sequencing Technologies
Recent advancements in analytical technologies are rapidly transforming peptide sequencing workflows, enabling faster and more accurate identification than ever before. Innovations such as ultra-high-resolution mass spectrometry and hybrid fragmentation techniques are improving sequence coverage and sensitivity, especially for complex biological samples. These advancements allow researchers to detect subtle sequence variations and rare peptides that were previously difficult to identify.
In addition, automation and miniaturization of analytical platforms are making peptide sequencing more accessible and efficient. High-throughput systems can now process large sample volumes with minimal human intervention, reducing variability and increasing reproducibility. As these technologies continue to evolve, they are expected to further streamline workflows and enhance the scalability of peptide-based research and development.
Ensure the integrity of your therapeutic candidates: Comprehensive Peptide Characterization Service
Advanced Techniques for Peptide Sequence Identification
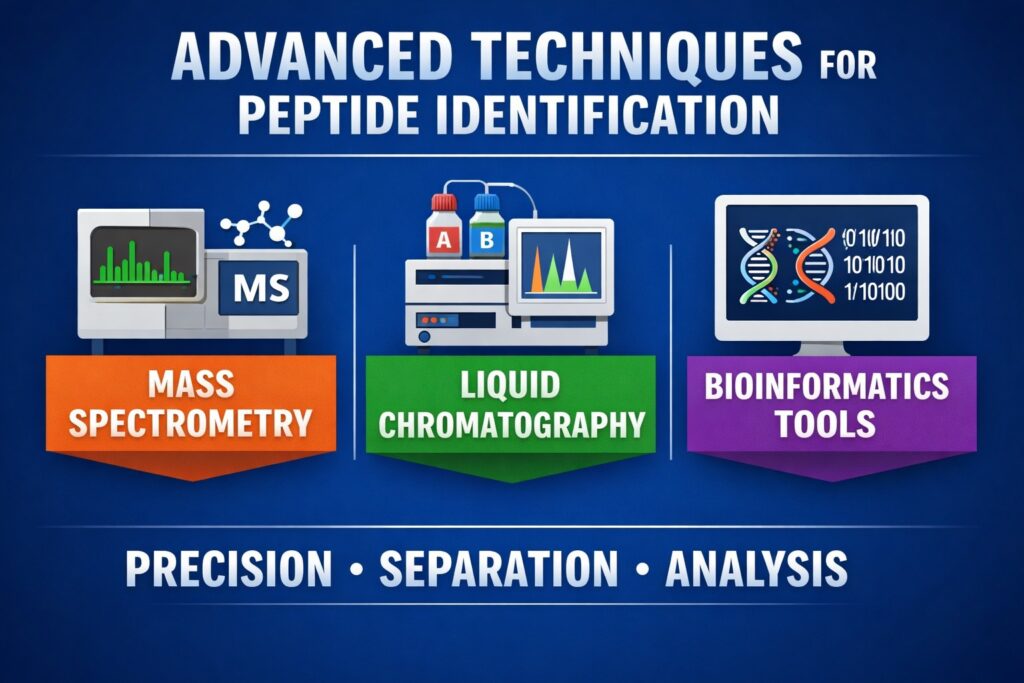
Resolvemass Laboratories employs a combination of cutting-edge technologies to achieve accurate and reliable peptide sequence identification:
Mass spectrometry is a pivotal technique in peptide sequencing due to its sensitivity and ability to provide detailed structural information.
Matrix-Assisted Laser Desorption/Ionization (MALDI-MS)
- Principle: MALDI-MS involves embedding peptides in a matrix and using a laser to ionize them. The ions are then analyzed based on their mass-to-charge ratio.
- Applications: Suitable for analyzing peptide mixtures, protein mass fingerprinting, and large biomolecules. It provides rapid, high-throughput analysis but may have lower resolution for complex mixtures.
- Electrospray Ionization (ESI-MS)
- Principle: ESI-MS involves ionizing peptides in a liquid phase and introducing them into the mass spectrometer. The ions are then analyzed to determine their mass and structure.
- Applications: Ideal for coupling with liquid chromatography (LC) for detailed analysis of peptide mixtures. It offers high resolution and sensitivity, making it suitable for complex samples.
- Tandem Mass Spectrometry (MS/MS)
- Principle: MS/MS involves multiple rounds of mass analysis. Peptides are first ionized and separated, then fragmented into smaller ions for further analysis.
- Applications: Provides detailed fragmentation patterns that are used for de novo sequencing and identifying post-translational modifications. It offers high specificity and sensitivity for complex peptide mixtures.
Liquid chromatography separates peptide mixtures before mass spectrometric analysis, enhancing resolution and sensitivity.
- High-Performance Liquid Chromatography (HPLC)
- Principle: HPLC uses a high-pressure liquid solvent to separate peptides based on their interactions with a stationary phase in the column.
- Applications: Provides high resolution and is widely used for separating complex peptide mixtures prior to MS analysis. It is essential for achieving clean, well-resolved peptide peaks.
- Reverse-Phase Liquid Chromatography (RP-LC)
- Principle: RP-LC separates peptides based on their hydrophobicity, using a non-polar stationary phase and a polar mobile phase.
- Applications: Commonly used for peptide separation due to its effectiveness in resolving peptides based on their hydrophobic properties. It enhances the sensitivity and specificity of peptide identification.
3. Bioinformatics Tools
Bioinformatics tools are crucial for analyzing MS data and identifying peptide sequences.
- Database Searching
- Principle: Matches experimental MS/MS spectra against theoretical spectra from protein sequence databases (e.g., NCBI, UniProt).
- Applications: Allows for the identification of known peptides and proteins by comparing experimental data with existing protein databases.
- De Novo Sequencing
- Principle: Uses algorithms to interpret MS/MS fragmentation data to determine peptide sequences without prior knowledge of the protein sequence.
- Applications: Essential for identifying novel peptides or proteins not present in existing databases. It provides detailed sequence information from experimental data.
- Post-Translational Modification Analysis
- Principle: Identifies and characterizes modifications such as phosphorylation, glycosylation, and acetylation.
- Applications: Crucial for understanding the functional implications of modifications on peptide activity and stability. It provides insights into regulatory mechanisms and disease mechanisms.
Get precise data on your peptide primary structures: Expert Peptide Sequencing Service
Role of Artificial Intelligence in Peptide Identification
Artificial intelligence (AI) and machine learning are increasingly being integrated into peptide sequence identification to improve data interpretation and predictive accuracy. AI-driven algorithms can analyze vast datasets generated by mass spectrometry, identifying patterns and correlations that may not be apparent through traditional methods. This significantly enhances the speed and reliability of peptide identification, especially in large-scale proteomics studies.
Moreover, machine learning models can predict peptide fragmentation patterns, retention times, and post-translational modifications with high precision. These predictive capabilities reduce dependency on existing databases and improve de novo sequencing outcomes. As AI continues to mature, it is expected to play a central role in automating complex analytical processes and enabling more intelligent decision-making in peptide research.
The Resolvemass Laboratories Advantage
Resolvemass Laboratories offers a comprehensive approach to peptide sequence identification, leveraging advanced technologies and customized solutions:
1. Customized Sample Preparation
Proper sample preparation is essential for accurate peptide analysis.
- Protein Extraction and Digestion
- Methods: Efficiently extract proteins from biological samples using techniques such as homogenization or sonication. Digest proteins into peptides using enzymes like trypsin to prepare them for LC-MS analysis.
- Importance: Ensures that peptides are in a suitable form for analysis and that the sample is free from contaminants that could interfere with results.
- Peptide Enrichment
- Methods: Use affinity chromatography or other techniques to isolate specific peptide classes (e.g., phosphopeptides) to enhance detection sensitivity.
- Importance: Focuses the analysis on peptides of interest, improving the accuracy of detection and quantification.
2. Advanced Analytical Platforms
We utilize the latest technologies to achieve high-resolution peptide sequencing.
- High-Resolution Mass Spectrometers
- Instruments: Orbitrap and time-of-flight (TOF) mass spectrometers provide exceptional mass accuracy and resolution.
- Advantages: Allows for precise measurement of peptide masses and detailed structural information, essential for accurate identification.
- Multi-Dimensional LC-MS
- Techniques: Combine various separation techniques (e.g., ion exchange, reverse-phase) to achieve comprehensive peptide coverage.
- Advantages: Enhances the resolution and sensitivity of peptide analysis, providing a more complete profile of the peptide mixture.
3. Expert Data Analysis
Our team employs advanced bioinformatics tools for accurate peptide sequence identification.
- Database Searches and De Novo Sequencing
- Tools: Use software such as Mascot, SEQUEST, and PEAKS for database searches and de novo sequencing.
- Advantages: Provides accurate peptide identification and sequence information, facilitating comprehensive analysis of complex samples.
- Post-Translational Modification Analysis
- Tools: Utilize specialized software for identifying and characterizing peptide modifications.
- Advantages: Offers insights into the functional implications of modifications and helps in understanding biological processes and disease mechanisms.
Quality Control and Validation in Peptide Mapping
Ensuring the accuracy and reliability of peptide sequence identification requires robust quality control and validation strategies. Analytical workflows must include steps such as calibration of instruments, use of internal standards, and verification of reproducibility across multiple runs. These measures help minimize experimental errors and ensure that results are consistent and dependable.
Validation also involves cross-checking identified peptide sequences using complementary techniques or independent datasets. This is particularly important in clinical and pharmaceutical applications, where incorrect identification can lead to significant consequences. Implementing stringent validation protocols enhances confidence in the results and ensures compliance with regulatory and scientific standards.
Verify the consistency of your biosimilar products: Professional Peptide Sameness Study
Case Study: Peptide Biomarker Discovery
A biotechnology company sought our expertise to identify peptide biomarkers for a novel cancer therapy.
Applications of Peptide Mapping in Biopharmaceutical Industry
Peptide mapping plays a critical role in the development and quality control of biopharmaceutical products. It is widely used to confirm the identity and structural integrity of therapeutic proteins, ensuring that they meet regulatory requirements. By analyzing peptide fragments, researchers can detect sequence variations, degradation products, and impurities that may affect drug safety and efficacy.
Additionally, peptide mapping is essential for biosimilar development, where demonstrating similarity to a reference product is crucial. Detailed peptide analysis helps establish comparability in terms of structure and function. This ensures that biosimilars perform equivalently to original biologics, supporting their approval and commercialization in the healthcare market.
Challenges
- Complex Biological Samples: Peptides were extracted from human serum, a complex matrix with a wide dynamic range.
- Low-Abundance Biomarkers: The goal was to detect low-abundance peptide biomarkers relevant to cancer therapy.
Solution
- Sample Preparation and Enrichment: Optimized protein extraction, digestion, and peptide enrichment to enhance detection sensitivity.
- Advanced LC-MS/MS Analysis: Employed multi-dimensional LC coupled with high-resolution MS/MS to achieve comprehensive peptide coverage.
- Bioinformatics Analysis: Used database searches and de novo sequencing to identify peptide biomarkers and characterize post-translational modifications.
Outcome
The successful identification of peptide biomarkers provided valuable insights into therapeutic mechanisms and efficacy. Our comprehensive approach enabled the biotechnology company to advance their drug development program.
Challenges in Peptide Sequence Identification
Despite significant technological advancements, peptide sequence identification still presents several challenges. One major issue is the complexity of biological samples, which often contain a wide range of peptides with varying abundances. Detecting low-abundance peptides amidst highly abundant ones requires highly sensitive instruments and optimized workflows, which can be resource-intensive.
Another challenge lies in accurately identifying post-translational modifications and sequence variants. These modifications can alter peptide mass and fragmentation patterns, complicating data interpretation. Additionally, incomplete or inaccurate databases may limit the effectiveness of database-driven identification methods. Addressing these challenges requires continuous improvements in analytical techniques and computational tools.
Future Perspectives in Peptide Mapping
The future of peptide mapping is poised for remarkable growth, driven by continuous innovation in analytical technologies and computational methods. Emerging approaches such as single-molecule sequencing and real-time mass spectrometry analysis are expected to further enhance sensitivity and speed. These advancements will enable researchers to study peptides in greater detail and uncover new biological insights.
Furthermore, integration of multi-omics data, including genomics and transcriptomics, will provide a more comprehensive understanding of biological systems. This holistic approach will improve the identification and functional characterization of peptides, paving the way for personalized medicine and targeted therapies. As the field continues to evolve, peptide mapping will remain a cornerstone of modern biomedical research.
Conclusion
Peptide sequence identification continues to be a cornerstone of modern proteomics, driving innovation in biomarker discovery, drug development, and biopharmaceutical analysis. With the integration of advanced analytical techniques, artificial intelligence, and robust validation strategies, the accuracy and efficiency of peptide mapping have significantly improved. These advancements are enabling researchers to tackle increasingly complex biological challenges with greater confidence.
At Resolvemass Laboratories, our commitment extends beyond delivering precise results—we aim to stay at the forefront of technological and scientific progress. By continuously adopting emerging tools and refining our methodologies, we empower researchers and organizations to unlock deeper insights into peptide structures and functions. As the field evolves, our expertise ensures that clients remain equipped to harness the full potential of peptide-based innovations.
Contact Us
For more information about our peptide sequence identification services and how we can assist with your research and development needs, please Resolvemass laboratories & contact us
FAQs on Peptide Mapping
Peptide mapping typically involves enzymatic digestion of proteins followed by separation and analysis using techniques like liquid chromatography and mass spectrometry. The resulting peptide fragments are identified and compared to known sequences. This allows researchers to reconstruct the protein and detect any variations or modifications.
Common techniques include high-performance liquid chromatography (HPLC) and mass spectrometry (MS), often used together as LC-MS. Enzymes like trypsin are used to digest proteins into peptides. Advanced tools such as tandem MS provide detailed structural information, enabling precise identification of peptide sequences.
Peptide mapping is widely used in biopharmaceuticals for protein characterization, quality control, and biosimilar comparison. It also plays a role in biomarker discovery and disease research. Additionally, it helps in detecting impurities, degradation, and post-translational modifications in protein-based products.
One key challenge is analyzing complex samples that contain many peptides with different concentrations. Low-abundance peptides can be difficult to detect. Another issue is identifying post-translational modifications, which can alter peptide behavior and complicate data interpretation.
In quality control, peptide mapping verifies the consistency and integrity of protein-based drugs. It ensures that each batch matches the expected structure and sequence. This helps detect any changes during manufacturing or storage, maintaining product safety and effectiveness.
Peptide mapping focuses on analyzing peptide fragments generated from proteins, often to confirm identity and detect modifications. Protein sequencing, on the other hand, aims to determine the complete amino acid sequence of a protein. While related, peptide mapping is more commonly used for routine analysis and validation.
Reference:
- Degueldre, M., Degryse, B., Dejonckheere, E., & others. (2019). Native peptide mapping: A simple method to routinely monitor higher order structure of monoclonal antibodies. mAbs, 11(8), 1409–1420. https://doi.org/10.1080/19420862.2019.1634460
- Mendoza, L., Deutsch, E. W., Sun, Z., Campbell, D. S., Shteynberg, D., & Moritz, R. L. (2018). Flexible and fast mapping of peptides to a proteome with ProteoMapper. Journal of Proteome Research, 17(1), 433–439. https://doi.org/10.1021/acs.jproteome.7b00605
- Banks, J. F., & Gulcicek, E. E. (1997). Rapid peptide mapping by reversed-phase liquid chromatography on nonporous silica with on-line electrospray time-of-flight mass spectrometry. Analytical Chemistry, 69(19), 3973–3978. https://doi.org/10.1021/ac970226t