
Introduction:
Peptide Mapping GLP-1 Peptides is a fundamental analytical strategy used in the structural characterization of GLP-1 analogs, enabling precise identification of amino acid sequences and post-translational modifications (PTMs). As GLP-1-based therapeutics continue to dominate the metabolic disease landscape, accurate analytical techniques such as LC-MS characterization of GLP-1 peptides are essential to ensure safety, efficacy, and regulatory compliance.
At ResolveMass Laboratories Inc., advanced peptide mapping workflows aligned with analytical characterization of GLP-1 peptide drugs are implemented to deliver high-resolution, reproducible, and regulatory-grade data for pharmaceutical and biotech applications.
Share via:
Summary:
- Peptide Mapping GLP-1 Peptides is a critical analytical technique used to confirm sequence integrity, detect modifications, and ensure product quality.
- It combines enzymatic digestion, LC-MS/MS analysis, and data interpretation to achieve high-confidence structural characterization.
- It integrates advanced approaches such as peptide sequencing of GLP-1 peptide workflows to achieve high-confidence structural characterization.
- Essential for drug development, biosimilar comparison, stability studies, and regulatory submissions, it supports compliance with regulatory requirements for GLP-1 peptide characterization.
- High-resolution mass spectrometry (HRMS) significantly improves accuracy, sensitivity, and reproducibility, especially when combined with de novo GLP-1 peptide sequencing accuracy strategies.
- A robust workflow ensures compliance with FDA, EMA, and Health Canada guidelines.
1: What is Peptide Mapping of GLP-1 Peptides?
Peptide mapping of GLP-1 peptides is an analytical technique that breaks the peptide into smaller fragments and analyzes them using LC-MS/MS to confirm its structure and integrity. This approach is closely related to GLP-1 peptide sequencing analytical techniques.
This method combines enzymatic digestion, liquid chromatography (LC), and tandem mass spectrometry (MS/MS), similar to workflows used in GLP-1 analog peptide sequencing workflow.
How It Works
- Enzymatic Digestion
The GLP-1 peptide is cleaved into predictable smaller fragments using specific enzymes (e.g., trypsin). - Chromatographic Separation (LC)
The resulting peptide fragments are separated based on their chemical properties to reduce complexity. - Mass Spectrometric Detection (MS/MS)
Each fragment is analyzed to determine its mass and sequence, enabling precise structural characterization.
What It Helps Identify
- Primary Amino Acid Sequence (validated via peptide sequencing of GLP-1 drugs)
Confirms that the peptide structure matches the intended design. - Post-Translational Modifications (PTMs)
Detects modifications such as oxidation, acylation, or glycation. - Impurities or Degradation Products (analyzed through GLP-1 peptide impurity characterization)
Identifies unwanted byproducts formed during synthesis or storage. - Batch-to-Batch Consistency
Ensures reproducibility and quality across manufacturing lots.
This technique is essential for Peptide Mapping GLP-1 Peptides, as it provides the high-resolution data required for pharmaceutical development, quality control, and regulatory compliance.
2: Why is Peptide Mapping GLP-1 Peptides Important?
Peptide Mapping GLP-1 Peptides is critical because GLP-1 analogs often contain structural modifications that directly influence their biological activity and stability. These aspects are deeply connected with GLP-1 peptide stability analytical methods.
These peptides are highly sensitive to even minor changes, making precise structural characterization essential for ensuring safety, efficacy, and regulatory acceptance.
Key Importance:
Regulatory Compliance
- Required by global regulatory agencies such as the U.S. Food and Drug Administration and European Medicines Agency
- Supports submissions aligned with GLP-1 peptide characterization regulatory requirements
Quality Control
- Ensures batch-to-batch consistency in manufacturing
- Detects impurities using GLP-1 peptide impurity sequencing analysis
Structural Confirmation
- Validates the primary amino acid sequence
- Confirms presence and position of post-translational modifications (PTMs)
Stability Studies
- Identifies degradation pathways under stress conditions (e.g., heat, oxidation)
- Helps establish shelf-life and storage conditions
Biosimilar Development
- Enables comparative analysis with reference GLP-1 products
- Demonstrates similarity in structure and function for regulatory approval
- Relies on accurate structural comparison enabled by GLP-1 peptide sequencing challenges solutions
Overall, Peptide Mapping GLP-1 Peptides plays a critical role in ensuring that GLP-1 therapeutics meet the highest standards of quality, safety, and regulatory compliance, making it indispensable in modern pharmaceutical development.
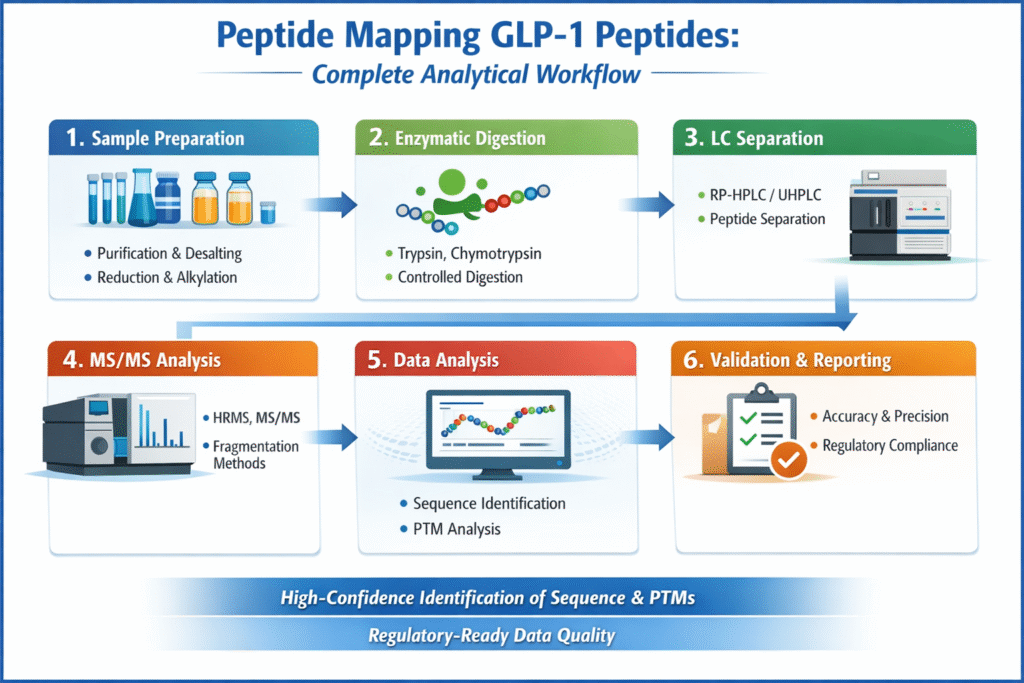
3: Complete Analytical Workflow for Peptide Mapping GLP-1 Peptides
The complete workflow integrates multiple advanced analytical techniques often implemented by a specialized CRO for GLP-1 peptide characterization.
From digestion to validation, this workflow aligns with industry-grade GLP-1 peptide sequencing CRO services to ensure reproducibility and compliance.
A well-optimized workflow ensures high-confidence sequence confirmation, PTM identification, and regulatory-ready data quality.
1. Sample Preparation
Sample preparation ensures the purity and integrity of GLP-1 peptides prior to analysis.
Key Steps
- Protein/peptide purification
- Desalting to remove buffer salts
- Reduction and alkylation (if required)
Why It Matters
- Eliminates contaminants that interfere with MS signals
- Improves enzymatic digestion efficiency
- Enhances overall data quality
2. Enzymatic Digestion
Enzymatic digestion cleaves GLP-1 peptides into predictable fragments for detailed analysis.
Common Enzymes Used
- Trypsin
- Chymotrypsin
- Glu-C
Best Practices
- Optimize enzyme-to-substrate ratio
- Control temperature and incubation time
- Prevent over-digestion to avoid fragment loss
3. Liquid Chromatography (LC) Separation
LC separates peptide fragments to reduce complexity before mass spectrometry analysis.
Advantages
- Improves detection sensitivity
- Minimizes ion suppression
- Enhances resolution of closely related fragments
Common Techniques
- Reverse-phase HPLC (RP-HPLC)
- Ultra-high-performance LC (UHPLC)
4. Mass Spectrometry (MS/MS) Analysis
Mass spectrometry identifies peptide fragments and determines their sequence with high precision.
Key Technologies
- High-Resolution Mass Spectrometry (HRMS)
- Tandem MS (MS/MS)
- Orbitrap and Q-TOF systems
Fragmentation Methods
- CID (Collision-Induced Dissociation)
- HCD (Higher-energy Collisional Dissociation)
- ETD (Electron Transfer Dissociation)
5. Data Analysis and Interpretation
Advanced computational tools combined with expert review reconstruct peptide sequences and identify structural features.
Key Outputs
- Peptide sequence confirmation
- Post-translational modification (PTM) identification
- Sequence coverage percentage
Tools Used
- De novo sequencing software
- Database search algorithms
6. Validation and Reporting
For pharmaceutical applications, validation is non-negotiable.
Key Validation Parameters
- Accuracy (mass error, identification confidence)
- Precision (intra/inter-day reproducibility)
- Specificity (ability to distinguish variants)
- Sensitivity (LOD/LOQ for impurities)
- Robustness (method stability under small variations)
Documentation Requirements
- System suitability tests
- Audit trails
- Raw data + processed results
- SOP compliance
Regulatory Alignment
- ICH Q2 (R2) for analytical validation
- GLP/GMP data integrity principles
Final Insight
A high-quality peptide mapping workflow for GLP-1 peptides is not just about instrumentation—it’s about control at every step:
- Controlled digestion → reproducible fragments
- High-resolution LC-MS → confident identification
- Rigorous validation → regulatory acceptance
When optimized end-to-end, this workflow delivers:
- Near-complete sequence coverage
- Reliable PTM profiling
- Batch-to-batch comparability
This structured workflow is the backbone of Peptide Mapping GLP-1 Peptides, enabling laboratories to achieve high-resolution, reproducible, and regulatory-grade structural characterization essential for pharmaceutical development and quality control.
4: Key Challenges in Peptide Mapping GLP-1 Peptides
Peptide mapping of GLP-1 peptides is challenging due to their structural complexity, presence of modifications, and analytical limitations. These factors can affect accuracy, sensitivity, and reproducibility of results.
The challenges in peptide mapping are closely associated with limitations addressed in GLP-1 peptide sequencing challenges.
- PTMs complicate analysis
- Complex fragmentation reduces confidence
- Low-level impurities require GLP-1 peptide impurity characterization
- Instability requires robust GLP-1 peptide stability analytical methods
1. Post-Translational Modifications (PTMs)
GLP-1 peptides often contain multiple chemical modifications such as acylation, oxidation, and glycation.
Challenges
- PTMs alter molecular mass and fragmentation behavior
- Multiple modified forms (isoforms) complicate identification
- Require high-resolution mass spectrometry for accurate detection
2. Complex Fragmentation Patterns
GLP-1 peptides do not always fragment in predictable ways during MS/MS analysis.
Challenges
- Incomplete or weak fragment ion signals
- Overlapping spectra
- PTM-dependent variability in fragmentation
Impact
- Difficult sequence reconstruction
- Lower confidence in de novo sequencing
3. Low Abundance Impurities
Impurities and degradation products are often present at trace levels.
Challenges
- Low signal intensity compared to main peptide
- Requires highly sensitive instrumentation
- Difficult to differentiate from background noise
Impact
- Risk of missing critical impurities
- Compromises quality assessment
4. Sample Instability
GLP-1 peptides can degrade during sample preparation and handling.
Challenges
- Susceptible to hydrolysis and oxidation
- Environmental exposure (temperature, light) affects stability
- Adsorption losses during handling
Impact
- Changes in peptide structure before analysis
- Reduced accuracy and reproducibility
Conclusion
These challenges make peptide mapping of GLP-1 peptides a technically demanding process, requiring optimized workflows, high-resolution instrumentation, and expert data interpretation to ensure accurate and reliable results.
5: Best Practices to Improve Accuracy
Accuracy in peptide mapping can be significantly improved by combining optimized workflows, advanced instrumentation, and expert data interpretation.
Improving accuracy often involves combining advanced instrumentation with expert-driven workflows, including outsourcing to outsource GLP-1 peptide sequencing services.
Recommended Strategies
1. Use High-Resolution MS Systems
- Enables precise mass measurement
- Improves identification of closely related peptide fragments
- Enhances detection of low-abundance species
2. Apply Multiple Fragmentation Techniques
- Use complementary methods such as CID, HCD, and ETD
- Improves sequence coverage and confidence
- Helps in accurate identification of PTMs
3. Optimize Digestion Protocols
- Control enzyme-to-substrate ratio
- Maintain optimal temperature and incubation time
- Prevent over- or under-digestion
4. Perform Replicate Analyses
- Ensures reproducibility of results
- Reduces experimental variability
- Improves confidence in data interpretation
5. Include Manual Expert Validation
- Confirms automated sequence assignments
- Identifies potential errors or ambiguities
- Enhances overall data reliability
These practices are essential for achieving high confidence in de novo GLP-1 peptide sequencing accuracy.
6: Applications of Peptide Mapping GLP-1 Peptides
Applications span across pharmaceutical development and advanced analytics, often supported by analytical characterization of GLP-1 peptide drugs.
Major Applications
- Drug development and characterization
- Biosimilar comparison studies
- Quality control and batch release testing
- Stability and degradation studies
- Regulatory submissions
Analytical Workflow Summary Table
| Step | Purpose | Key Techniques |
|---|---|---|
| Sample Preparation | Remove impurities | Desalting, purification |
| Enzymatic Digestion | Generate fragments | Trypsin, Glu-C |
| LC Separation | Reduce complexity | RP-HPLC, UHPLC |
| MS/MS Analysis | Identify fragments | HRMS, Orbitrap |
| Data Analysis | Sequence reconstruction | Software + expert review |
| Validation | Ensure reliability | Regulatory guidelines |
7: Why Choose ResolveMass Laboratories Inc.?
ResolveMass Laboratories Inc. combines cutting-edge instrumentation, validated workflows, and deep scientific expertise to deliver high-quality peptide mapping services.
ResolveMass Laboratories Inc. offers specialized expertise in GLP-1 peptide sequencing CRO services and end-to-end analytical solutions.
Key Strengths
- Advanced HRMS platforms
- Experienced analytical scientists
- Regulatory-compliant workflows
- Expertise in LC-MS characterization of GLP-1 peptides
- Fast turnaround times
- Customized solutions for GLP-1 analysis
Conclusion:
Peptide Mapping GLP-1 Peptides is a powerful and essential analytical approach for ensuring accurate structural characterization, quality control, and regulatory compliance of GLP-1 therapeutics.
By integrating advanced techniques such as peptide sequencing of GLP-1 peptide and leveraging expert-driven workflows, laboratories can achieve high-confidence, reproducible results.
ResolveMass Laboratories Inc. remains a trusted partner for organizations seeking reliable, regulatory-grade solutions, including outsource GLP-1 peptide sequencing services tailored to evolving pharmaceutical needs.
Frequently Asked Questions:
Peptide Mapping GLP-1 Peptides is an advanced analytical technique used to break down GLP-1 molecules into smaller fragments and analyze them using LC-MS/MS. It helps confirm the amino acid sequence and detect structural modifications. This method ensures that the peptide matches its intended design. It is widely used in pharmaceutical development and quality control. It also supports regulatory submissions by providing detailed structural data.
Peptide mapping is crucial because GLP-1 therapeutics often contain structural modifications that impact their performance. It ensures product quality, safety, and consistency across batches. The technique helps identify impurities and degradation products. It also confirms the presence of critical post-translational modifications. Regulatory agencies require such detailed characterization for drug approval.
Peptide mapping primarily uses enzymatic digestion, liquid chromatography (LC), and mass spectrometry (MS/MS). LC separates peptide fragments, while MS/MS identifies and sequences them. High-resolution instruments like Orbitrap and Q-TOF are commonly used. Multiple fragmentation techniques such as CID, HCD, and ETD improve accuracy. Together, these techniques provide high-confidence structural characterization.
Accuracy can be improved by using high-resolution mass spectrometry and optimized digestion protocols. Applying multiple fragmentation techniques enhances sequence coverage. Performing replicate analyses ensures reproducibility. Careful sample handling reduces degradation risks. Manual expert validation further improves confidence in the results.
Peptide mapping can detect various post-translational modifications such as oxidation, acylation, and glycation. These modifications can affect the peptide’s stability and biological activity. The technique identifies both the presence and location of these changes. This information is critical for understanding drug behavior. It also helps in maintaining product quality and consistency.
Impurities are identified using highly sensitive LC-MS/MS techniques. These methods can detect even trace-level degradation products. Advanced data analysis tools help differentiate impurities from background noise. Identification of impurities is essential for ensuring product safety. It also plays a key role in regulatory compliance.
Yes, peptide mapping is an essential requirement for regulatory approval of GLP-1 drugs. Agencies like the FDA and EMA require detailed structural characterization. It helps demonstrate identity, purity, and consistency of the product. The data supports quality control and stability studies. Without peptide mapping, regulatory approval is difficult to achieve.
Reference
- Maheshwari D, Badgujar D, Kumar G, Sharma N. Peptide Mapping Using Multienzyme Digestion Strategies Integrated with LC‐HRMS Workflow: A Case Study. Journal of Peptide Science. 2026 Mar;32(3):e70089.https://onlinelibrary.wiley.com/doi/abs/10.1002/psc.70089
- Sobhia ME. Structural basis for the identification of short novel peptides as potential GLP1R agonists using in silico approaches.https://scholar.archive.org/work/koov5qgmuzgkxdkkows2bptyge/access/wayback/https://assets.researchsquare.com/files/rs-1721735/v1/9755f71b-9a74-411d-925e-e5ff59971b26.pdf?c=1656000488
- Kim SH, Kim SS, Kim HJ, Park EJ, Na DH. Peptide mapping analysis of synthetic semaglutide and liraglutide for generic development of drugs originating from recombinant DNA technology. Journal of Pharmaceutical and Biomedical Analysis. 2025 Apr 15;256:116682.https://www.sciencedirect.com/science/article/pii/S0731708525000238
- Nielsen JC, Hjorringgaard C, Nygaard MM, Wester A, Elster L, Porsgaard T, Mikkelsen RB, Rasmussen S, Madsen AN, Schlein M, Vrang N. Machine-learning-guided peptide drug discovery: Development of GLP-1 receptor agonists with improved drug properties. Journal of Medicinal Chemistry. 2024 Jul 8;67(14):11814-26.https://pubs.acs.org/doi/abs/10.1021/acs.jmedchem.4c00417
- Jazayeri A, Rappas M, Brown AJ, Kean J, Errey JC, Robertson NJ, Fiez-Vandal C, Andrews SP, Congreve M, Bortolato A, Mason JS. Crystal structure of the GLP-1 receptor bound to a peptide agonist. Nature. 2017 Jun 8;546(7657):254-8.https://www.nature.com/articles/nature22800


