
Introduction:
Peptide Sameness Testing Methods are advanced analytical strategies used to demonstrate that a generic or follow-on peptide drug is structurally and chemically identical to the reference listed drug (RLD). These methods are critical for regulatory approval, particularly in ANDA submissions, where bioequivalence alone is not sufficient for complex peptides.
At ResolveMass Laboratories Inc., we specialize in high-resolution mass spectrometry, impurity profiling, and orthogonal analytical characterization to establish peptide sameness with scientific rigor and regulatory alignment. Our expertise spans peptide characterization in drug development, characterization of peptides for FDA submissions, and comprehensive peptide sameness study for ANDA programs.
Summary:
- What Peptide Sameness Testing Methods are and why they are critical for regulatory approval
- Key analytical techniques used to establish peptide sameness
- How primary structure, higher-order structure, impurity profile, and physicochemical properties are evaluated
- Regulatory expectations for ANDA and generic peptide submissions
- Why advanced mass spectrometry and orthogonal methods are essential
- How ResolveMass Laboratories Inc. ensures scientifically robust and regulator-ready peptide sameness data
Video Overview of Peptide Sameness Testing Methods
1: Why Are Peptide Sameness Testing Methods Critical?
Peptide Sameness Testing Methods are critical because even small structural differences in peptide drugs can directly impact safety, efficacy, and regulatory approval.
Peptide drugs are inherently complex and highly sensitive to manufacturing conditions. Minor variations in:
- Amino acid sequence
- Disulfide bond arrangement
- Post-translational modifications
- Impurity profiles
can alter biological activity, stability, or immunogenic potential.
For this reason, regulatory authorities such as the U.S. Food and Drug Administration and the European Medicines Agency require comprehensive analytical characterization aligned with current FDA requirements for peptide characterization.
They expect robust scientific evidence demonstrating structural and physicochemical sameness — not just pharmacokinetic bioequivalence — before approving generic or follow-on peptide products.
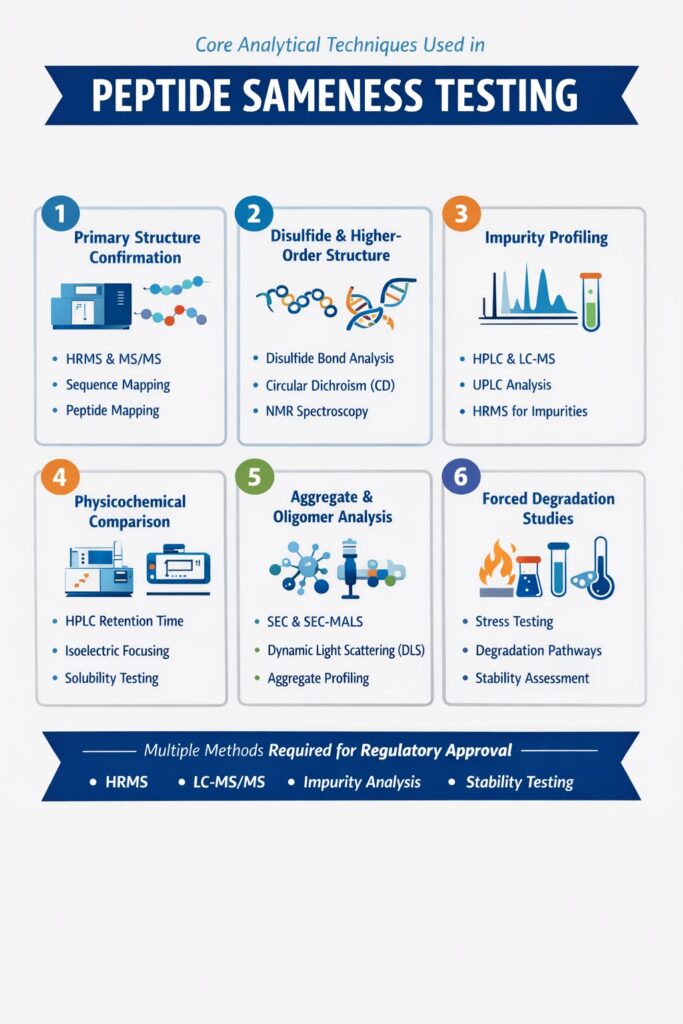
2: Core Analytical Techniques Used in Peptide Sameness Testing
Below are the most important Peptide Sameness Testing Methods used in regulatory submissions to demonstrate structural, physicochemical, and impurity equivalence with the reference listed drug (RLD). Each section answers the key question upfront to ensure clarity for both regulatory reviewers and AI-driven search engines.
2.1 Primary Structure Confirmation in Peptide Sameness Testing Methods
Primary structure confirmation verifies the exact amino acid sequence and molecular weight of the peptide, ensuring it matches the reference product precisely.
Primary structure is the foundation of all Peptide Sameness Testing Methods. Even a single amino acid substitution, deletion, or truncation can impact biological function and regulatory acceptance.
ResolveMass applies advanced peptide characterization techniques and applications to ensure complete sequence integrity.
Key Techniques
a. High-Resolution Mass Spectrometry (HRMS)
HRMS provides accurate molecular weight confirmation with exceptional precision.
We are recognized as peptide mass spectrometry experts delivering:
- Exact molecular mass
- Detection of truncations
- Identification of sequence variants
- Sub-ppm mass accuracy
For unknown peaks or structural ambiguity, we apply strategies similar to those discussed in how to identify unknown peptides by LCMS testing.
HRMS establishes that the peptide’s molecular formula aligns with the reference standard, forming the analytical backbone of sameness assessment.
b. Tandem Mass Spectrometry (MS/MS)
MS/MS enables sequence mapping through controlled fragmentation of the peptide backbone.
What it confirms:
- Full sequence coverage
- Backbone integrity
- Detection of single amino acid substitutions
- Localization of modifications
This technique strengthens confidence in structural identity within Peptide Sameness Testing Methods.
c. Peptide Mapping
Peptide mapping provides detailed structural confirmation through enzymatic digestion and fragment analysis.
Typical workflow:
- Enzymatic digestion (e.g., trypsin)
- LC-MS/MS analysis of fragments
- Comparative overlay with RLD chromatogram
Regulatory Expectation:
Complete sequence coverage with no unexplained fragments or unmatched peaks.
2.2 Disulfide Bond and Higher-Order Structure in Peptide Sameness Testing Methods
Disulfide bond analysis ensures correct cysteine pairing, while higher-order structure analysis confirms conformational similarity with the reference product.
This is particularly critical for complex generics such as:
- Peptide Characterization of Ganirelix Generic Project
- Peptide Characterization of Lanreotide Generic Project
Orthogonal structural confirmation strengthens regulatory defensibility.
Incorrect disulfide connectivity can significantly alter biological activity and stability.
a. Disulfide Bond Mapping
Disulfide mapping verifies correct cysteine pairing without structural distortion.
Approaches include:
- Non-reduced peptide mapping
- MS-based confirmation of cysteine connectivity
- Comparative analysis with reference product
Accurate disulfide confirmation is essential in complex Peptide Sameness Testing Methods.
b. Circular Dichroism (CD)
CD spectroscopy evaluates secondary structure characteristics.
What it confirms:
- Alpha-helix content
- Beta-sheet composition
- Global conformational similarity
CD provides a structural fingerprint that supports conformational equivalence.
c. NMR Spectroscopy
NMR offers high-resolution conformational insights.
Applications:
- Structural fingerprinting
- Verification of higher-order structural equivalence
- Confirmation of folding integrity
For complex peptides, regulators expect orthogonal confirmation to eliminate ambiguity in higher-order structure comparisons.
2.3 Impurity Profiling as Part of Peptide Sameness Testing Methods
Impurity profiling ensures that the type, level, and nature of impurities in the generic peptide are comparable to the reference product.
Our approach aligns with insights from:
- Impurity Profiling in Peptides – Why It Matters in Drug Development
- What Is Peptide Purity by HPLC and Why It Matters
- Peptide Purity Testing in United States
We also perform detailed peptide degradation product characterization during forced degradation studies.
Impurity comparability is a major regulatory focus in Peptide Sameness Testing Methods.
Key Techniques
| Technique | Purpose | Regulatory Relevance |
|---|---|---|
| RP-HPLC | Purity and related substances | Release specification |
| UPLC | High-resolution impurity separation | Batch consistency |
| LC-MS | Impurity identification | Structural confirmation |
| HRMS | Trace-level impurity detection | Toxicological risk evaluation |
Types of Peptide Impurities
- Deamidation products
- Oxidation products
- Truncated sequences
- Aggregates
- Process-related impurities
Regulators expect impurity qualification, structural identification, and scientific justification for any observed differences versus the RLD.
2.4 Physicochemical Property Comparison in Peptide Sameness Testing Methods
Physicochemical testing confirms that the peptide behaves identically to the reference product under validated analytical conditions.
Our regulatory-aligned testing supports both:
- Peptide Characterization for IND and NDA
- Peptide Sameness Study Services in United States
- Peptide Sameness Study Services in Canada
This comparison strengthens totality-of-evidence arguments in regulatory submissions.
Analytical Approaches
- Reverse Phase HPLC retention time comparison
- Isoelectric focusing (pI determination)
- Solubility profiling
- Hydrophobicity assessment
Retention time alignment within validated limits supports chromatographic equivalence, while pI and solubility comparisons reinforce physicochemical similarity.
2.5 Aggregate and Oligomer Analysis in Peptide Sameness Testing Methods
Aggregation studies confirm that the generic peptide does not form higher molecular weight species at levels different from the reference product.
This testing is often integrated within our broader peptide testing services for pharmaceutical R&D.
Aggregation can influence:
- Immunogenicity
- Stability
- Biological activity
Techniques Used
- Size Exclusion Chromatography (SEC)
- SEC-MALS (Multi-Angle Light Scattering)
- Dynamic Light Scattering (DLS)
Comparative aggregate profiling is a critical safety evaluation component within Peptide Sameness Testing Methods.
2.6 Forced Degradation and Stability Studies in Peptide Sameness Testing Methods
Forced degradation studies evaluate how the peptide degrades under stress and compare degradation pathways with the reference product.
We support synthesis-phase investigations as outlined in:
- Analytical Support in Peptide Synthesis – Why It’s Essential
- Solid vs Liquid Phase Peptide Synthesis – Which Method Is Better?
Matching degradation behavior strengthens structural equivalence claims.
Stress Conditions Evaluated
- Acid hydrolysis
- Base hydrolysis
- Oxidative stress
- Thermal stress
- Photolytic exposure
These studies help:
- Identify degradation pathways
- Confirm stability-indicating method suitability
- Compare degradant profiles with RLD
Equivalent degradation pathways provide strong regulatory evidence of molecular sameness.
Why Multiple Peptide Sameness Testing Methods Are Required
Regulatory agencies expect a totality-of-evidence approach, meaning no single analytical technique is sufficient. Instead, multiple orthogonal Peptide Sameness Testing Methods are integrated to eliminate scientific uncertainty.
By combining:
- HRMS
- LC-MS/MS
- Peptide mapping
- Chromatographic impurity profiling
- Structural spectroscopy
- Aggregation studies
- Stability testing
manufacturers can build a comprehensive and regulator-ready demonstration of peptide sameness.
3: Orthogonal Strategy: Why Multiple Peptide Sameness Testing Methods Are Required
Regulatory agencies require orthogonal analytical approaches. No single technique is sufficient.
Example Orthogonal Matrix:
| Attribute | Primary Technique | Orthogonal Confirmation |
|---|---|---|
| Molecular Weight | HRMS | MALDI-TOF |
| Sequence | LC-MS/MS | Peptide mapping |
| Disulfide Bonds | MS mapping | NMR |
| Impurities | RP-HPLC | LC-HRMS |
Selecting the right analytical CRO is critical. See Top 5 Things to Look for in a Peptide Testing Laboratory and Peptide Synthesis Service – How to Choose the Right CRO Partner.
This layered approach strengthens regulatory defensibility.
4: Regulatory Expectations for Peptide Sameness Testing Methods
For ANDA submissions, regulators require comprehensive analytical characterization of peptides — bioequivalence alone is not sufficient.
Key Regulatory Expectations
- Bioequivalence alone is insufficient for peptide drugs due to structural complexity.
- Complete structural characterization is required, including sequence confirmation and molecular weight accuracy.
- Impurity comparability is mandatory, covering both related substances and degradation products.
- Scientific justification is needed for any minor analytical differences.
Regulatory submissions must align with global expectations, including formal characterization of peptides for FDA requirements.
Agencies such as the U.S. Food and Drug Administration evaluate submissions using a totality-of-evidence approach, where all data from Peptide Sameness Testing Methods collectively confirm structural and physicochemical equivalence to the reference product.
5: Common Challenges in Peptide Sameness Testing Methods
Peptide Sameness Testing Methods must detect extremely subtle structural and impurity differences to meet regulatory expectations. Below are the most common scientific and regulatory challenges — and how they are effectively addressed.
5.1 Detecting Low-Level Sequence Variants
Even a single amino acid substitution or truncation at trace levels can impact safety, efficacy, and regulatory acceptance.
Why It’s Challenging:
- Variants may be present at <0.1% levels
- Mass differences can be extremely small
- Co-existing impurities may mask signals
How We Address It at ResolveMass:
- High-resolution Orbitrap-based platforms with sub-ppm accuracy
- Targeted and untargeted LC-MS/MS workflows
- Deep sequence coverage through optimized fragmentation strategies
- Extracted ion chromatogram (XIC) analysis for trace detection
This ensures confident identification of even minor sequence deviations.
5.2 Resolving Co-Eluting Impurities
Co-eluting impurities can obscure accurate purity assessment and complicate impurity identification.
Why It’s Challenging:
- Similar hydrophobicity among peptide-related impurities
- Structural similarity to the main peptide
- Limitations of conventional HPLC resolution
How We Address It at ResolveMass:
- Ultra-high-performance liquid chromatography (UHPLC) method optimization
- Multi-column chemistry screening (C18, phenyl, polar embedded phases)
- Orthogonal LC-MS confirmation
- High-resolution MS for deconvolution of overlapping peaks
This layered analytical strategy strengthens impurity differentiation in Peptide Sameness Testing Methods.
5.3 Confirming Complex Disulfide Arrangements
Incorrect cysteine pairing can alter biological activity, making disulfide mapping a critical part of peptide sameness.
Why It’s Challenging:
- Multiple cysteine residues create pairing permutations
- Disulfide scrambling during sample prep
- Reduced vs non-reduced analytical complexity
How We Address It at ResolveMass:
- Non-reduced peptide mapping
- Controlled reduction/alkylation workflows
- MS-based disulfide bond mapping
- Orthogonal structural confirmation where necessary
This ensures precise confirmation of correct disulfide connectivity.
5.4 Differentiating Process-Related Impurities
Process-related impurities must be distinguished from degradation products and structurally related variants.
Why It’s Challenging:
- Synthetic reagents may leave trace adducts
- Side reactions during solid-phase peptide synthesis
- Similar mass profiles to genuine degradants
How We Address It at ResolveMass:
- HRMS-based structural elucidation
- Accurate mass confirmation of impurity molecular formula
- Comparative profiling against reference product
- Forced degradation studies to distinguish degradants
These strategies support regulatory defensibility within Peptide Sameness Testing Methods.
5.5 Aligning Analytical Methods with Reference Product Data
Regulatory agencies expect analytical comparisons to be scientifically aligned with the reference listed drug (RLD).
Why It’s Challenging:
- Limited publicly available RLD analytical details
- Batch-to-batch variability
- Differences in excipients or formulation matrix
How We Address It at ResolveMass:
- Reverse engineering of analytical conditions
- Comparative batch testing strategies
- Method bridging and optimization
- Regulatory-aligned method validation protocols
Our approach ensures that comparative data is robust, reproducible, and submission-ready.

6: Our Technical Capabilities That Strengthen Peptide Sameness Testing Methods
At ResolveMass Laboratories Inc., we combine scientific expertise with regulatory awareness to deliver defensible analytical outcomes:
- High-resolution Orbitrap-based mass spectrometry platforms
- Advanced data processing and spectral interpretation software
- Regulatory-aligned method validation
- Comparative batch testing strategies
- Orthogonal analytical confirmation
By proactively addressing these technical and regulatory challenges, we reduce submission risk and accelerate approval timelines for peptide-based generics.
7: Why Choose ResolveMass Laboratories Inc. for Peptide Sameness Testing Methods?
ResolveMass Laboratories Inc. combines deep expertise in:
- Mass spectrometry-based peptide characterization
- Regulatory submission support
- Impurity identification and qualification
- Method development and validation
Our approach ensures:
- Scientifically defensible data
- Regulatory-ready reports
- Fast turnaround without compromising analytical rigor
- Transparent documentation aligned with global guidelines
We support pharmaceutical innovators and generic manufacturers across North America with advanced peptide analytical services.
Conclusion
Peptide Sameness Testing Methods are essential for demonstrating structural, physicochemical, and impurity equivalence between a generic peptide and its reference product. Regulatory agencies require comprehensive analytical evidence — including primary structure confirmation, impurity profiling, higher-order structure analysis, and orthogonal validation — to establish sameness.
At ResolveMass Laboratories Inc., we apply advanced mass spectrometry, chromatography, and structural analysis techniques to generate robust, regulator-ready peptide sameness data. If you are planning an ANDA submission or developing a generic peptide product, partnering with an experienced analytical laboratory can significantly reduce regulatory risk and accelerate approval timelines.
Frequently Asked Questions:
Analytical techniques for peptides are used to determine their sequence, purity, structure, and stability.
The most common techniques include:
-High-Resolution Mass Spectrometry (HRMS) – Molecular weight confirmation
-LC-MS/MS – Sequence verification and PTM identification
-Peptide Mapping – Enzymatic digestion with fragment analysis
-HPLC / UPLC – Purity and impurity profiling
-Size Exclusion Chromatography (SEC) – Aggregate detection
-Circular Dichroism (CD) – Secondary structure analysis
-NMR Spectroscopy – Higher-order structure confirmation
-Isoelectric Focusing (IEF) – pI determination
These methods are widely used in Peptide Sameness Testing Methods, regulatory submissions, and drug development.
The four core techniques commonly used in analytical chemistry are:
a. Chromatography – Separation of compounds (HPLC, GC, UPLC)
b. Spectroscopy – Interaction with electromagnetic radiation (UV, IR, NMR)
c. Mass Spectrometry – Molecular weight and structural analysis
d. Electrochemical Analysis – Measurement of electrical properties (potentiometry, voltammetry)
In peptide analysis, chromatography and mass spectrometry are especially critical.
Peptide synthesis techniques are methods used to assemble amino acids into peptides.
The two primary approaches are:
Solid-Phase Peptide Synthesis (SPPS)
-Most widely used method
-Suitable for automated synthesis
-High efficiency for short-to-medium peptides
Liquid-Phase Peptide Synthesis (LPPS)
-Used for large-scale production
-Suitable for industrial applications
Other supporting techniques include:
-Microwave-assisted synthesis
-Native chemical ligation
-Recombinant peptide expression (for larger proteins)
Peptides are commonly classified based on length:
a. Dipeptides – 2 amino acids
b. Tripeptides – 3 amino acids
c. Oligopeptides – 4–20 amino acids
d. Polypeptides – More than 20 amino acids
They may also be categorized functionally as:
-Hormonal peptides
-Antimicrobial peptides
-Signaling peptides
-Therapeutic peptides
Peptide characterization methods confirm identity, purity, structure, and stability.
Key characterization techniques include:
~HRMS – Exact mass confirmation
~LC-MS/MS – Sequence mapping
~Peptide Mapping – Enzymatic digest comparison
~HPLC/UPLC – Purity and impurity analysis
~SEC – Aggregation analysis
~CD Spectroscopy – Secondary structure assessment
~NMR Spectroscopy – Conformational analysis
~Forced Degradation Studies – Stability and degradant profiling
These methods collectively form the foundation of regulatory-compliant peptide characterization and are essential in ANDA submissions and peptide drug development.
Reference
- Lamiaa A. Hassan, Sara M. Shatat , Basma M. Eltanany , Medhat A. Al-Ghobashy and Samah S. Abbas. Stability and biosimilarity assessment of infliximab using an orthogonal testing protocol and statistically-guided interpretation of peptide mapping.https://pubs.rsc.org/en/content/articlelanding/2019/ay/c9ay00903e/unauth
- Survey of peptide quantification methods and comparison of their reproducibility: A case study using oxytocin.https://www.sciencedirect.com/science/article/pii/S0731708518323306
- Analytical considerations for characterization of generic peptide product: A regulatory insight.https://www.sciencedirect.com/science/article/abs/pii/S0003269724001775
- Sara H Abdelghaffar , Maha A Hegazy , Basma M Eltanany. Stability and Biosimilarity Assessment of Bevacizumab Monoclonal Antibody; Orthogonal Testing Protocol Coupled With Peptide Mapping–Principal Component Analysis.https://academic.oup.com/jaoac/article/107/1/177/7247536


