
Introduction:
Peptide Sameness vs Biosimilar Comparability is one of the most misunderstood concepts in modern pharmaceutical development. While both involve comparison to a reference listed drug, the scientific expectations, regulatory frameworks, and analytical depth differ significantly.
For generic and biologic developers, understanding this distinction directly impacts regulatory strategy, cost, risk, and approval timelines. At ResolveMass Laboratories Inc., we specialize in advanced mass spectrometry and structural characterization that supports both pathways with regulatory-grade analytical data, including Peptide Sameness Study for ANDA and global regulatory submissions.
Summary:
- Peptide Sameness Study vs Biosimilar Comparability refers to two different regulatory and analytical pathways for generic peptide drugs and biosimilar biologics.
- Peptide sameness focuses on proving identical structure, purity, and physicochemical properties to the reference peptide.
- Biosimilar comparability evaluates high similarity, not exact sameness, to a reference biologic.
- Peptide generics typically follow ANDA or hybrid pathways, while biosimilars follow dedicated biologics frameworks.
- Analytical depth differs: peptides require precise molecular identity confirmation, whereas biosimilars require multi-layered structural, functional, and clinical comparison.
- Regulatory agencies such as Health Canada, U.S. Food and Drug Administration, and European Medicines Agency clearly differentiate these pathways.
Share via:
Quick Video Explanation: Peptide Sameness Study vs Biosimilar Comparability
1: What Is a Peptide Sameness Study?
A peptide sameness study is a comprehensive analytical program designed to prove that a generic peptide drug is structurally, chemically, and functionally identical to its reference listed drug (RLD). In simple terms, regulators expect confirmation that the proposed generic peptide is the same molecule, not merely similar.
Our dedicated Peptide Sameness Study Services in Canada and Peptide Sameness Study Services in United States are aligned with global submission requirements.
Unlike complex biologics produced in living systems, synthetic peptides are well-defined chemical entities. Because they are manufactured through controlled chemical synthesis, regulatory agencies such as the U.S. Food and Drug Administration, Health Canada, and European Medicines Agency require precise demonstration of molecular identity.
To meet regulatory expectations, a peptide sameness study must demonstrate:
- Identical primary amino acid sequence
- Exact molecular weight match
- Comparable impurity profile
- Consistent higher-order structure (if applicable)
- Absence of unexpected modifications or degradants
For regulatory clarity, sponsors often review FDA Requirements for Peptide Characterization and detailed Characterization of Peptides for FDA guidance.
Because peptide therapeutics can be sensitive to oxidation, deamidation, truncation, or racemization, even minor differences must be ruled out using advanced analytical techniques.
Analytical Components of a Peptide Sameness Study
A scientifically rigorous study relies on orthogonal, high-resolution techniques as described in our guide on Peptide Sameness Testing Methods and broader Peptide Characterization Techniques and Applications.
1. High-Resolution Mass Spectrometry (HRMS)
- Confirms exact molecular mass
- Detects minor mass differences
- Identifies trace-level impurities
- Supports isotopic pattern verification
HRMS ensures the proposed generic matches the reference product at the molecular level with high mass accuracy.
HRMS confirms exact molecular mass and detects trace impurities. Learn more from our Peptide Mass Spectrometry Experts team.
2. LC-MS/MS Peptide Mapping
Peptide mapping confirms complete amino acid sequence and PTMs. For deeper insight, see:
- Confirms complete amino acid sequence
- Verifies fragmentation patterns
- Detects sequence variants or truncations
- Confirms site-specific modifications
Peptide mapping provides sequence-level confirmation and is considered one of the most critical components of sameness evaluation.
- Peptide Mapping vs Peptide Sequencing: Key Differences
- Peptide Mapping for PTM Analysis
- How to Identify Unknown Peptides by LCMS Testing
3. Amino Acid Sequence Confirmation
- Validates primary structure
- Confirms absence of substitutions
- Detects terminal modifications
Even a single amino acid difference can alter biological activity, making sequence confirmation essential.
4. Disulfide Bond Mapping (If Applicable)
For peptides containing cysteine residues:
- Confirms correct disulfide pairing
- Detects mispaired or scrambled bonds
- Ensures proper folding consistency
Incorrect disulfide bonding can significantly impact peptide stability and activity.
5. Impurity Profiling
- Compares impurity spectrum between test and reference
- Identifies synthesis-related impurities
- Evaluates degradation products
- Assesses batch-to-batch consistency
Regulators expect a highly comparable impurity profile, especially for process-related and oxidative degradants.
Impurity comparison is central to sameness. Our technical insights include:
- Impurity Profiling in Peptides: Why It Matters in Drug Development
- What Is Peptide Purity by HPLC and Why It Matters
- Peptide Purity Testing in United States
6. Forced Degradation Studies
- Evaluates stability under stress conditions
- Compares degradation pathways
- Identifies potential vulnerability points
- Supports stability-indicating method validation
Forced degradation ensures both products behave similarly under thermal, oxidative, acidic, and photolytic stress.
Forced degradation ensures comparable stability behavior:
7. Orthogonal Chromatographic Evaluation
- Reverse-phase HPLC
- Ion-exchange chromatography (if required)
- Size exclusion chromatography (for aggregation assessment)
Using multiple separation principles ensures comprehensive structural and purity assessment.

2: What Is Biosimilar Comparability?
Biosimilar comparability is a structured scientific and regulatory process used to demonstrate that a biologic product is highly similar — but not identical — to an approved reference biologic, with no clinically meaningful differences in safety, purity, or potency.
Unlike synthetic small molecules or peptides, biologics such as monoclonal antibodies are produced in living cells. Because biological systems introduce natural variability (e.g., glycosylation patterns, post-translational modifications), exact molecular replication is not possible. Therefore, regulators require a comparability framework rather than proof of identity.
Regulatory authorities such as the U.S. Food and Drug Administration, European Medicines Agency, and Health Canada define biosimilarity as a product that is highly similar to the reference product notwithstanding minor differences in clinically inactive components, with no clinically meaningful differences in safety or effectiveness.
Why Exact Identity Is Impossible for Biologics
Biologics are:
- Produced in mammalian or microbial cell lines
- Sensitive to culture conditions
- Affected by purification and formulation processes
- Structurally complex (often >150 kDa proteins)
Even small manufacturing changes can alter:
- Glycosylation profiles
- Charge variants
- Aggregation levels
- Folding patterns
Because “the process defines the product,” regulators focus on demonstrating functional and clinical similarity, not molecular identity.
3: Peptide Sameness Study vs Biosimilar Comparability: Core Scientific Difference
The core scientific difference in Peptide Sameness Study vs Biosimilar Comparability is simple and definitive: peptide sameness requires proof of molecular identity, whereas biosimilar comparability requires proof of high similarity with no clinically meaningful differences.
This distinction arises from how these products are manufactured and how much structural variability is scientifically possible.
Core Comparison Table
| Parameter | Peptide Sameness Study | Biosimilar Comparability |
|---|---|---|
| Molecular Nature | Small, synthetic peptide | Large, cell-derived biologic |
| Expectation | Exact sameness | High similarity |
| Structural Variability | Minimal and controllable | Inherent biological variability |
| Clinical Requirement | Often limited or conditional | Usually required |
| Regulatory Route | Generic drug pathway | Biosimilar biologics pathway |
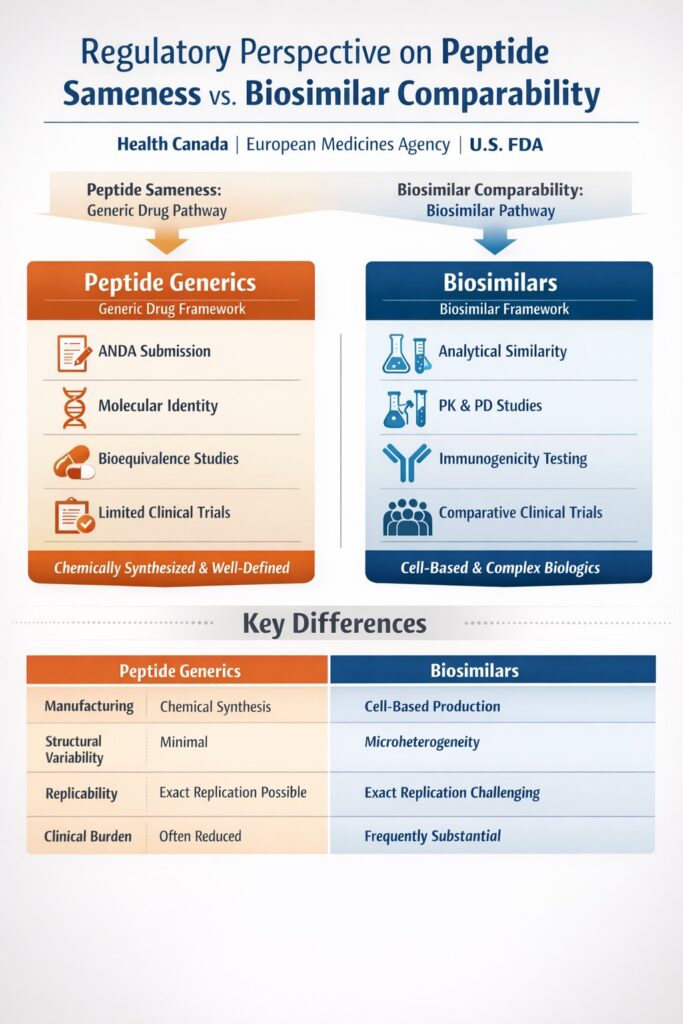
4: Regulatory Perspective on Peptide Sameness vs Biosimilar Comparability
From a regulatory standpoint, Peptide Sameness vs Biosimilar Comparability is treated differently because of molecular complexity, manufacturing variability, and clinical risk considerations. In short, synthetic peptides are evaluated under generic drug principles requiring proof of identity, while biologics follow biosimilar frameworks requiring demonstration of high similarity.
Global authorities such as the Health Canada, European Medicines Agency, and U.S. Food and Drug Administration clearly separate these regulatory pathways.
Peptide Generics: Regulatory Expectations
For synthetic peptides, regulators typically apply generic drug frameworks, provided the molecule qualifies as a chemically synthesized product rather than a complex biologic.
Key Regulatory Characteristics
- Commonly submitted under ANDA or abbreviated generic pathways
- Strong emphasis on analytical characterization
- Demonstration of molecular identity (sameness)
- Bioequivalence studies may be required depending on dosage form and route of administration
- Limited or no large-scale clinical efficacy trials in many cases
Why the Pathway Is More Streamlined
Peptides are:
- Chemically synthesized under controlled reactions
- Structurally well-defined
- Reproducible batch-to-batch
Because exact replication is scientifically achievable, regulators expect sponsors to prove identity through robust analytical data rather than extensive clinical programs.
Biosimilars: Regulatory Expectations
Under biosimilar pathways defined by major global agencies, sponsors must follow a stepwise comparability framework.
Regulatory Authorities Governing Biosimilars
- Health Canada
- European Medicines Agency
- U.S. Food and Drug Administration
Required Data Package Typically Includes:
- Extensive analytical similarity data
- Functional bioassays
- Pharmacokinetic (PK) studies
- Pharmacodynamic (PD) evaluation (if applicable)
- Immunogenicity assessment
- Comparative clinical trials (as needed)
The guiding principle is the “totality of evidence,” where analytical similarity forms the foundation and clinical data confirm absence of meaningful differences.
Why Regulators Draw a Clear Line
The distinction in Peptide Sameness vs Biosimilar Comparability exists because:
| Factor | Peptide Generics | Biosimilars |
|---|---|---|
| Manufacturing | Chemical synthesis | Cell-based production |
| Structural Variability | Minimal | Inherent microheterogeneity |
| Replicability | Exact replication possible | Exact replication impossible |
| Clinical Burden | Often reduced | Frequently substantial |
Biologics carry additional risks such as immunogenicity and structural heterogeneity, which justify expanded comparability requirements.
Peptide submissions often align with:
Real-world examples:
- Peptide Characterization of Ganirelix Generic Project
- Peptide Characterization of Lanreotide Generic Project
For sponsors outsourcing R&D:
- Peptide Testing Services for Pharmaceutical R&D
- Top 5 Things to Look for in a Peptide Testing Laboratory
5: Analytical Depth: How Testing Strategies Differ
Peptide Sameness Study vs Biosimilar Comparability in Analytical Testing
Peptide sameness relies heavily on structural confirmation, while biosimilar comparability requires multi-dimensional characterization.
For Peptide Sameness:
- Accurate mass measurement
- Fragmentation pattern matching
- Impurity comparison
- Synthetic route evaluation
- Degradation pathway alignment
The goal: eliminate structural uncertainty.
For Biosimilar Comparability:
- Primary structure confirmation
- Glycosylation profiling
- Charge variant analysis
- Aggregation assessment
- Binding affinity studies
- Functional bioassays
The goal: demonstrate comparable biological activity and safety.
6: Manufacturing Considerations
Manufacturing variability is the reason Peptide Sameness vs Biosimilar Comparability differs so fundamentally.
Peptides
- Chemically synthesized
- Controlled reaction steps
- Predictable impurity profiles
- Highly reproducible batch-to-batch consistency
Biologics
- Produced in living cells
- Sensitive to culture conditions
- Glycosylation patterns vary
- Process defines product
This is why biologics can only be “biosimilar,” not identical.
7: Risk Assessment Differences
Peptide Sameness Study vs Biosimilar Comparability in Risk Evaluation
Risk in peptide generics is primarily chemical; risk in biosimilars is structural and immunological.
Peptide Risks
- Sequence truncation
- Oxidation
- Deamidation
- Racemization
- Synthetic impurities
These are measurable using advanced LC-MS workflows.
Biosimilar Risks
- Immunogenicity
- Glycan variability
- Protein aggregation
- Functional potency variation
Thus, biosimilar pathways inherently require broader datasets.
8: Cost and Timeline Implications
Peptide Sameness vs Biosimilar Comparability also differs significantly in development cost and time.
| Factor | Peptide Sameness | Biosimilar Comparability |
|---|---|---|
| Analytical Cost | Moderate | High |
| Clinical Trials | Limited/Conditional | Extensive |
| Development Time | Shorter | Longer |
| Regulatory Complexity | Defined | Multi-stage |
Peptide generic development is generally faster and more cost-efficient than biosimilar development.
9: When Does a Peptide Fall Into the Biosimilar Category?
Certain complex peptides may fall closer to biologics depending on size and manufacturing method.
Examples include:
- Long-chain polypeptides
- Peptides produced via recombinant expression
- Molecules with complex tertiary structures
Regulatory classification may depend on jurisdiction and molecular complexity.
10: Why Accurate Classification Is Critical
Misunderstanding Peptide Sameness vs Biosimilar Comparability can delay approval and increase regulatory risk.
Common mistakes include:
- Assuming peptide generics require full biosimilar pathways
- Underestimating analytical depth required for sameness
- Failing to perform orthogonal characterization
- Inadequate impurity comparison
Early regulatory strategy alignment prevents costly rework.
11: Role of Advanced Mass Spectrometry in Both Pathways
At ResolveMass Laboratories Inc., our expertise lies in high-resolution analytical platforms that support:
For Peptide Sameness:
- Exact mass confirmation
- MS/MS sequencing
- Impurity profiling at trace levels
- Forced degradation comparison
For Biosimilar Comparability:
- Peptide mapping
- Glycan characterization
- Post-translational modification analysis
- Stability comparison studies
Our laboratory workflows are aligned with global regulatory expectations, ensuring submission-ready data packages.
12: Peptide Sameness Study vs Biosimilar Comparability: Final Comparison Summary
Peptide Sameness Study vs Biosimilar Comparability differs in regulatory expectation, analytical depth, clinical requirements, and development cost.
To summarize:
- Peptide sameness proves molecular identity
- Biosimilar comparability proves clinical similarity
- Peptides are synthetic and reproducible
- Biologics are complex and variable
- Regulatory pathways are distinct
- Analytical requirements reflect molecular complexity
Understanding this difference ensures the right development strategy from day one.
Conclusion
The distinction between Peptide Sameness vs Biosimilar Comparability is not merely regulatory terminology—it defines the entire scientific and strategic approach to drug development.
Peptide generics require rigorous structural confirmation to prove identity. Biosimilars require multi-layered analytical, functional, and clinical comparison to demonstrate high similarity without meaningful differences.
At ResolveMass Laboratories Inc., we provide regulatory-aligned peptide characterization and advanced mass spectrometry solutions tailored to both pathways. Our experience in structural elucidation, impurity profiling, and comparability assessment ensures sponsors receive defensible, submission-ready analytical data.
If you are planning a peptide generic or biosimilar development program, selecting the correct pathway early can significantly reduce time, cost, and regulatory risk.
Frequently Asked Questions:
A biosimilar is a biologic shown to be highly similar to a reference product with no clinically meaningful differences in safety, purity, or potency. An interchangeable product meets additional regulatory criteria demonstrating it can be substituted at the pharmacy level without prescriber intervention. In the United States, interchangeability is designated by the U.S. Food and Drug Administration and requires switching studies to confirm consistent safety and efficacy when alternating between products.
The comparability exercise is a stepwise scientific evaluation used to demonstrate high similarity between a proposed biosimilar and its reference biologic. It includes analytical characterization (structure, post-translational modifications), functional assays, non-clinical studies, and clinical trials. Regulatory agencies such as the European Medicines Agency require robust analytical evidence as the foundation, followed by confirmatory clinical data where necessary.
Not typically. Most synthetic peptides are regulated as generic drugs rather than biosimilars because they are chemically synthesized and structurally well-defined. If a peptide can be fully characterized and shown to be identical to the reference product, it follows a generic approval pathway. Biosimilar frameworks generally apply to larger, complex biologics produced in living systems.
A biosimilar study is a comprehensive development program designed to demonstrate high similarity to a reference biologic. It includes analytical similarity testing, in vitro functional studies, pharmacokinetic/pharmacodynamic comparisons, immunogenicity assessment, and at least one clinical efficacy study (if required). The goal is not to re-establish efficacy from scratch but to confirm no clinically meaningful differences from the original product.
Peptides are commonly classified into four main types based on structure and origin:
a. Oligopeptides – Short chains (typically 2–20 amino acids)
b. Polypeptides – Longer chains (20+ amino acids)
c. Cyclic peptides – Peptides forming ring structures
d. Peptide hormones – Biologically active regulatory peptides (e.g., insulin)
These classifications help determine analytical strategy and regulatory pathway.
Reference
- Recommendation for Clarifying FDA Policy in Evaluating “Sameness” of Higher Order Structure for Generic Peptide Therapeutics.https://link.springer.com/article/10.1208/s12248-024-00994-8
- Gregory C. Davis, John M. Beals, Craig Johnson, Mark H. Mayer, Bruce I. Meiklejohn, Bruce H. Mitlak.Recommendations regarding technical standards for follow-on biologics: comparability, similarity, interchangeability.https://www.tandfonline.com/doi/abs/10.1185/03007990903017313
- Robert J. Falconer, Dianne Jackson-Matthews, Stephen M. Mahler.Analytical strategies for assessing comparability of biosimilars.https://scijournals.onlinelibrary.wiley.com/doi/abs/10.1002/jctb.2629
- Neh Nupur , Sumit Kumar Singh , Gunjan Narula , Anurag S. Rathore. Assessing analytical comparability of biosimilars: GCSF as a case study.https://www.sciencedirect.com/science/article/abs/pii/S1570023216303312
- Boštjan Japelj, Gregor Ilc, Jaka Marušič, Jure Senčar, Drago Kuzman & Janez Plavec. Biosimilar structural comparability assessment by NMR: from small proteins to monoclonal antibodies.https://www.nature.com/articles/srep32201
- Assessment of Structural and Functional Comparability of Biosimilar Products: Trastuzumab as a Case Study.https://link.springer.com/article/10.1007/s40259-020-00404-3
- Arati Deshmukh, Rishank Goyal, Kalyana Sundaram, Kaustubh Dange, Tejshri Lakhote, Sanjay Niranjan, Jennifer Bharucha, Ashok Mishra, Bhavesh Vats. Analytical sameness methodology for the evaluation of structural, physicochemical, and biological characteristics of Armlupeg: A pegfilgrastim biosimilar case study.https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0289745


