
Introduction:
Peptide Sequencing of GLP-1 Peptide plays a critical role in modern pharmaceutical research, especially in the development of incretin-based therapies for diabetes and obesity. GLP-1 (Glucagon-Like Peptide-1) and its analogs are structurally complex peptides that require precise analytical characterization of GLP-1 peptide drugs.
LC-MS/MS (Liquid Chromatography–Tandem Mass Spectrometry) has emerged as the most reliable and sensitive technique for peptide sequencing due to its ability to provide high-resolution mass data and detailed fragmentation patterns. Advanced LC-MS characterization of GLP-1 peptides enables accurate structural elucidation. In this guide, we will explore the complete workflow, challenges, and best practices for sequencing GLP-1 peptides using LC-MS/MS.
Share via:
Summary:
- Peptide Sequencing of GLP-1 Peptide is essential for confirming structure, purity, and modifications in therapeutic peptides.
- LC-MS/MS is the gold-standard analytical technique for sequencing GLP-1 and its analogs.
- Accurate sequencing helps in drug development, biosimilar comparison, and regulatory requirements for GLP-1 peptide characterization.
- Key challenges include post-translational modifications (PTMs), isomer differentiation, and fragmentation complexity.
- Advanced workflows using high-resolution mass spectrometry and optimized fragmentation techniques significantly improve accuracy.
1: What is Peptide Sequencing of GLP-1 Peptide?
Peptide Sequencing of GLP-1 Peptide is the process of determining the exact amino acid sequence and identifying structural modifications using advanced techniques such as LC-MS/MS. For deeper insights, refer to peptide sequencing of GLP-1 drugs.
GLP-1 (Glucagon-Like Peptide-1) is a biologically active peptide widely used in therapeutic applications, particularly in diabetes and metabolic disorder treatments. Due to its structural complexity and modifications in analogs, precise sequencing is essential.
Key Characteristics of GLP-1 Peptides
GLP-1 peptides typically consist of:
- 30–40 amino acids forming the active peptide backbone
- Modified residues, such as fatty acid conjugation in long-acting analogs
- Terminal modifications, including amidation or truncation
Why is Peptide Sequencing of GLP-1 Peptide Important?
Peptide sequencing is critical for ensuring the quality, safety, and efficacy of GLP-1-based therapeutics:
- Confirms primary structure integrity
Ensures the amino acid sequence matches the intended design - Identifies impurities and degradation products
Detects unwanted variants formed during synthesis or storage - Detects post-translational modifications (PTMs)
Helps characterize oxidation, deamidation, and other modifications - Ensures biosimilarity and batch consistency
Essential for generic development and regulatory approval
2: Why Use LC-MS/MS for GLP-1 Peptide Sequencing?
LC-MS/MS is the preferred technique for Peptide Sequencing of GLP-1 Peptide because it enables both efficient separation and precise structural identification within a single, integrated workflow.
This is particularly important for GLP-1 peptide impurity characterization, where trace-level impurities must be detected with high sensitivity.
This dual capability makes it especially powerful for analyzing complex GLP-1 peptides and their modified analogs.
Key Advantages of LC-MS/MS
- High sensitivity for low-abundance peptides
Detects trace-level impurities and minor variants that are critical in pharmaceutical analysis - Accurate mass measurement for sequence confirmation
High-resolution mass spectrometry ensures precise molecular weight determination - Fragmentation analysis (MS/MS) for amino acid mapping
Generates sequence-specific fragment ions (b/y ions) to confirm peptide structure - Ability to detect minor modifications and impurities
Identifies PTMs such as oxidation, deamidation, and fatty acid conjugation
Comparison with Other Techniques
| Technique | Capability for GLP-1 Sequencing | Limitation |
|---|---|---|
| Edman Degradation | Partial sequencing | Not suitable for modified peptides |
| NMR | Structural insights | Low sensitivity |
| LC-MS/MS | Full sequencing + PTMs detection | Requires technical expertise |
Why LC-MS/MS is the Gold Standard
- Combines chromatographic separation (LC) with mass-based detection (MS/MS)
- Handles complex peptide mixtures and modified analogs effectively
- Meets regulatory expectations for peptide characterization
- Provides high reproducibility and robustness for routine analysis
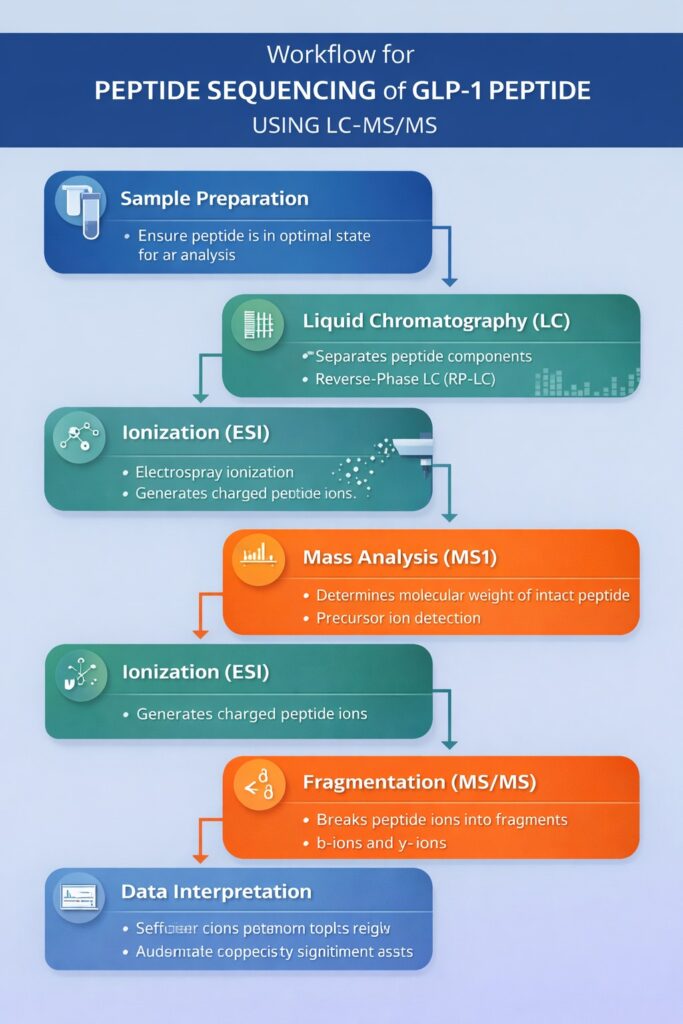
3: Workflow for Peptide Sequencing of GLP-1 Peptide Using LC-MS/MS
The workflow for Peptide Sequencing of GLP-1 Peptide using LC-MS/MS involves a systematic process of sample preparation, separation, ionization, mass analysis, fragmentation, and data interpretation to accurately determine peptide structure and modifications.
This workflow is often implemented by specialized CRO for GLP-1 peptide characterization providers to ensure regulatory-grade data quality.
Step-by-Step Process:
1. Sample Preparation
Sample preparation ensures the peptide is in an optimal state for accurate LC-MS/MS analysis.
- Dissolution in suitable solvent (e.g., water + acetonitrile)
- Reduction and alkylation (if disulfide bonds are present)
- Enzymatic digestion (optional for bottom-up sequencing approach)
✔ Key Insight: Clean and well-prepared samples significantly reduce noise and improve detection sensitivity.
2. Liquid Chromatography (LC)
LC separates peptide components before they enter the mass spectrometer.
- Reverse-phase LC (RP-LC) is most commonly used
- Separates GLP-1 peptides, impurities, and variants
- Minimizes matrix interference and ion suppression
✔ Key Insight: Proper chromatographic separation improves downstream identification and quantification accuracy.
3. Ionization (ESI)
Electrospray Ionization (ESI) converts peptides into charged ions suitable for MS detection.
- Generates multiply charged peptide ions
- Enhances detection of large biomolecules like GLP-1
- Compatible with LC flow rates
✔ Key Insight: Multiple charge states improve fragmentation efficiency and sequence coverage.
4. Mass Analysis (MS1)
MS1 analysis determines the molecular weight of the intact peptide.
- Measures precursor ion mass with high accuracy
- Confirms molecular weight of GLP-1 peptide
- Detects mass shifts due to modifications
✔ Key Insight: High-resolution MS enables differentiation of closely related peptide species.
5. Fragmentation (MS/MS)
MS/MS fragmentation breaks peptide ions into smaller fragments for sequencing.
- Produces b-ions and y-ions
- Provides amino acid sequence information
- Enables localization of modifications
✔ Key Insight: Optimized fragmentation conditions are critical for complete sequence coverage.
6. Data Interpretation
Data analysis reconstructs the peptide sequence and confirms structural integrity.
- Software tools generate peptide maps and spectra
- Automated sequencing algorithms assist identification
- Manual validation ensures high confidence results
✔ Key Insight: Combining software analysis with expert review ensures regulatory-grade accuracy.
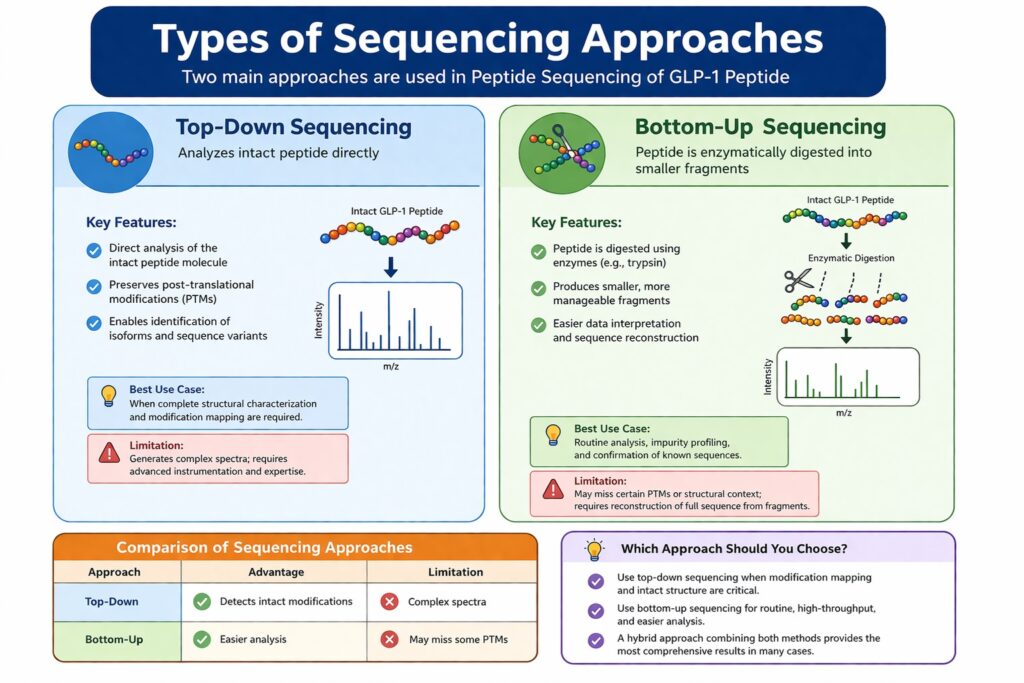
4: Types of Sequencing Approaches
Two main approaches are used in Peptide Sequencing of GLP-1 Peptide: top-down and bottom-up sequencing, each offering unique advantages depending on the analytical objective.
Top-Down Sequencing
Top-down sequencing analyzes the intact GLP-1 peptide directly, making it ideal for detecting full structural integrity and modifications.
Key Features:
- Direct analysis of the intact peptide molecule
- Preserves post-translational modifications (PTMs)
- Enables identification of isoforms and sequence variants
✔ Best Use Case:
When complete structural characterization and modification mapping are required.
✔ Limitation:
- Generates complex spectra
- Requires advanced instrumentation and expertise
Bottom-Up Sequencing
Bottom-up sequencing involves enzymatic digestion of the peptide into smaller fragments, simplifying sequence analysis.
Key Features:
- Peptide is digested using enzymes (e.g., trypsin)
- Produces smaller, more manageable fragments
- Easier data interpretation and sequence reconstruction
✔ Best Use Case:
Routine analysis, impurity profiling, and confirmation of known sequences.
✔ Limitation:
- May miss certain PTMs or structural context
- Requires reconstruction of full sequence from fragments
Comparison of Sequencing Approaches
| Approach | Advantage | Limitation |
|---|---|---|
| Top-Down | Detects intact modifications | Complex spectra |
| Bottom-Up | Easier analysis | May miss some PTMs |
Which Approach Should You Choose?
- Use top-down sequencing when modification mapping and intact structure are critical
- Use bottom-up sequencing for routine, high-throughput, and easier analysis
- In many cases, a hybrid approach combining both methods provides the most comprehensive results
5: Key Challenges in Peptide Sequencing of GLP-1 Peptide
The key challenges in Peptide Sequencing of GLP-1 Peptide primarily arise from complex fragmentation behavior and the difficulty of accurately detecting structural modifications and low-level impurities.
Many of these challenges are addressed through advanced GLP-1 peptide stability analytical methods, which help track degradation pathways and ensure reliable results.
GLP-1 peptides and their analogs often exhibit subtle structural variations, making precise sequencing both technically demanding and analytically intensive.
Common Challenges
- Isobaric amino acids (e.g., leucine vs isoleucine)
These amino acids have identical masses, making them difficult to differentiate using standard MS techniques - Post-translational modifications (PTMs)
Modifications such as oxidation, deamidation, and fatty acid conjugation complicate spectral interpretation - Low-abundance impurities
Trace-level impurities may be masked by dominant peptide signals - Co-eluting peaks
Incomplete chromatographic separation can lead to overlapping signals and misidentification - Signal suppression in complex matrices
Matrix components can reduce ionization efficiency, affecting sensitivity and accuracy
Practical Considerations to Overcome Challenges
- Optimize LC gradient for better separation
Enhances resolution between closely related peptide species - Use high-resolution mass spectrometry
Improves mass accuracy and helps distinguish similar compounds - Apply advanced fragmentation techniques
Enhances sequence coverage and modification detection - Validate results with orthogonal techniques
Confirms findings using complementary analytical methods
6: Role of Fragmentation Techniques
Fragmentation techniques are essential in Peptide Sequencing of GLP-1 Peptide because they generate sequence-specific fragment ions that enable accurate peptide mapping and structural characterization.
In LC-MS/MS, intact peptide ions are broken into smaller fragments, producing patterns (such as b-ions and y-ions) that reveal the amino acid sequence and location of modifications.
Common Fragmentation Methods
1. CID (Collision-Induced Dissociation)
CID fragments peptides by colliding them with inert gas molecules, making it widely used for routine sequencing.
- Produces mainly b-ions and y-ions
- Reliable and well-established technique
- Suitable for general peptide sequencing
2. HCD (Higher-energy Collisional Dissociation)
HCD is a higher-energy version of CID that provides improved fragmentation efficiency and higher-quality spectra.
- Generates rich and high-resolution fragment spectra
- Compatible with modern high-resolution MS systems
- Ideal for quantitative and qualitative analysis
3. ETD (Electron Transfer Dissociation)
ETD fragments peptides by transferring electrons, preserving labile modifications that may be lost in CID/HCD.
- Preserves post-translational modifications (PTMs)
- Produces c-ions and z-ions
- Ideal for analyzing modified peptides and large biomolecules
When to Use Which Technique
| Fragmentation Method | Best Use Case | Key Advantage |
|---|---|---|
| CID | Routine peptide sequencing | Reliable and widely applicable |
| HCD | High-resolution and quantitative work | Rich, high-quality spectra |
| ETD | PTM analysis and labile modifications | Preserves sensitive modifications |
Practical Insight
- Use CID or HCD for standard GLP-1 peptide sequencing workflows
- Use ETD when analyzing peptides with labile or complex modifications
- Combining multiple fragmentation techniques can significantly improve sequence coverage and confidence

7: Importance of Detecting Modifications in GLP-1 Peptides
Detecting modifications is crucial in Peptide Sequencing of GLP-1 Peptide because structural changes directly influence drug efficacy, stability, and regulatory compliance.
GLP-1 analogs are often intentionally modified to improve pharmacokinetics, but unintended changes can impact product quality. Accurate detection and characterization of these modifications are therefore essential.
Common Types of Modifications
- Fatty acid conjugation
Used to extend half-life by enhancing albumin binding (common in long-acting GLP-1 analogs) - Oxidation (e.g., Methionine oxidation)
Can occur during manufacturing or storage, affecting stability - Deamidation (Asparagine, Glutamine)
Leads to structural changes that may alter biological activity - Truncation or cleavage products
Results from degradation, impacting peptide integrity
Why Detecting Modifications Matters
- Affects bioavailability and efficacy
Structural changes can alter receptor binding and therapeutic performance - Critical for regulatory submissions
Regulatory agencies require detailed characterization of all variants and impurities - Ensures product stability
Helps monitor degradation pathways during stability studies - Supports biosimilarity assessments
Essential for comparing innovator and generic GLP-1 products
8: Data Analysis and Interpretation
Accurate data analysis and interpretation are essential in Peptide Sequencing of GLP-1 Peptide to ensure correct sequence assignment, reliable impurity identification, and confident characterization of structural modifications.
In LC-MS/MS workflows, even high-quality data can lead to incorrect conclusions without robust analysis strategies and expert validation.
Key Tools Used in Data Analysis
- Automated sequencing software
Rapidly processes MS/MS spectra to propose peptide sequences and identify fragments - Spectral libraries
Enable comparison with known peptide fragmentation patterns for faster identification - Manual verification
Critical step to confirm software-generated results and eliminate false positives
Best Practices for Reliable Interpretation
- Cross-check fragmentation patterns
Validate the presence of expected b-ions and y-ions for sequence confirmation - Validate with theoretical spectra
Compare experimental data with predicted fragmentation patterns - Confirm PTMs using high-resolution data
Detect subtle mass shifts and accurately localize modifications - Use multiple data analysis approaches
Combine automated tools with expert review for higher confidence
9: Applications in Pharmaceutical Development
Peptide Sequencing of GLP-1 Peptide is widely applied in pharmaceutical development to ensure drug quality, safety, efficacy, and regulatory compliance throughout the product lifecycle.
It plays a central role in meeting regulatory requirements for GLP-1 peptide characterization and ensuring compliance with global guidelines.
From early-stage research to commercial manufacturing, LC-MS/MS-based sequencing plays a critical role in characterizing GLP-1 peptides and their analogs.
Major Applications
- Biosimilar development
Confirms structural equivalence between innovator and generic GLP-1 products, including sequence and modifications - Impurity profiling
Identifies and characterizes process-related and degradation impurities at trace levels - Stability studies
Monitors degradation pathways such as oxidation, deamidation, and truncation over time - Formulation development
Ensures compatibility of GLP-1 peptides with excipients and delivery systems - Regulatory submissions
Provides critical analytical data required for IND, NDA, and ANDA filings
Regulatory Importance
- Required by global regulatory agencies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA)
- Supports compliance with ICH guidelines for peptide and biologics characterization
- Ensures data integrity, reproducibility, and traceability
- Demonstrates product consistency across batches
10: Best Practices for Accurate Sequencing
Following optimized workflows and validated analytical strategies is essential for achieving high accuracy, reproducibility, and regulatory compliance in Peptide Sequencing of GLP-1 Peptide.
Accurate sequencing is not only dependent on instrumentation but also on method optimization, validation, and expert interpretation.
Recommended Strategies
- Use high-resolution mass spectrometers
Enables precise mass measurement and better differentiation of closely related peptide species - Optimize fragmentation energy
Ensures efficient generation of informative fragment ions for complete sequence coverage - Perform replicate analyses
Improves confidence in results and helps identify variability or inconsistencies - Use internal standards
Supports accurate quantification and method reliability - Validate methods as per ICH guidelines
Ensures compliance with global regulatory expectations for analytical methods
Additional Expert Tips
- Optimize LC conditions (gradient, column, mobile phase) for improved separation
- Combine multiple fragmentation techniques (CID, HCD, ETD) for enhanced sequence coverage
- Regularly calibrate instruments to maintain mass accuracy
- Implement robust data review workflows combining software and manual validation
- Document all procedures to ensure traceability and audit readiness
11: Future Trends in GLP-1 Peptide Sequencing
Future advancements in Peptide Sequencing of GLP-1 Peptide are focused on improving sensitivity, speed, automation, and data intelligence through cutting-edge technologies.
As GLP-1 therapeutics continue to grow in demand, analytical techniques are evolving to deliver faster, more accurate, and highly reproducible results.
Key Innovations Shaping the Future
- AI-based spectral interpretation
Artificial intelligence and machine learning algorithms are transforming data analysis by enabling faster and more accurate peptide sequence identification, reducing reliance on manual validation - Ultra-high-resolution MS systems
Next-generation mass spectrometers offer exceptional mass accuracy and resolving power, allowing detection of even the smallest structural variations and trace-level impurities - Automated peptide mapping workflows
Fully automated systems streamline sample preparation, data acquisition, and analysis, significantly improving throughput and consistency - Integration with bioinformatics tools
Advanced software platforms enable seamless data integration, visualization, and interpretation, enhancing decision-making in complex peptide analysis
Impact on Pharmaceutical Development
- Faster turnaround times for sequencing and characterization
- Improved detection of low-level impurities and PTMs
- Enhanced reproducibility and reduced human error
- Better support for regulatory submissions with high-quality data
What This Means for GLP-1 Analysis
- Increased adoption of AI-driven analytics will make sequencing more efficient and scalable
- Automation will reduce operational variability and improve lab productivity
- Advanced MS technologies will push the boundaries of sensitivity and accuracy
- Integration of data science and analytical chemistry will redefine peptide characterization workflows
Conclusion:
Peptide Sequencing of GLP-1 Peptide using LC-MS/MS is the most reliable and advanced approach for structural characterization of therapeutic peptides. It enables precise identification of amino acid sequences, detection of modifications, and ensures regulatory compliance.
As GLP-1-based therapeutics continue to grow in importance, adopting robust LC-MS/MS sequencing strategies is essential for pharmaceutical companies aiming for quality, safety, and regulatory success.
Frequently Asked Questions:
Peptide analysis by LC-MS (Liquid Chromatography–Mass Spectrometry) is a technique used to separate, identify, and characterize peptides based on their mass and structure.
-LC separates complex peptide mixtures
-MS detects molecular weight and structure
-MS/MS provides fragmentation data for sequencing
-Widely used for impurity profiling, sequencing, and PTM analysis
Peptides are commonly classified based on their structure and function into four main types:
~Oligopeptides – Short chains (2–20 amino acids)
~Polypeptides – Longer chains (>20 amino acids)
~Cyclic peptides – Peptides with ring structures for enhanced stability
~Modified peptides – Peptides with chemical modifications (e.g., GLP-1 analogs)
The LC-MS method combines liquid chromatography for separation and mass spectrometry for detection and identification of compounds.
Basic Workflow:
-Sample injection into LC system
-Separation of components via chromatography
-Ionization (typically ESI)
-Mass detection (MS)
-Optional fragmentation (MS/MS) for structural analysis
✔ Used extensively in pharmaceutical, biotech, and proteomics research
Peptide sequencing is primarily performed using LC-MS/MS, which identifies amino acid sequences through fragmentation patterns.
Common Methods:
-LC-MS/MS sequencing (gold standard) – High sensitivity and accuracy
-Top-down sequencing – Intact peptide analysis
-Bottom-up sequencing – Enzymatic digestion followed by analysis
-Edman degradation – Traditional method (limited for modified peptides)
Reference
- Lee TS, Park EJ, Choi M, Oh HS, An Y, Kim T, Kim TH, Shin BS, Shin S. Novel LC-MS/MS analysis of the GLP-1 analog semaglutide with its application to pharmacokinetics and brain distribution studies in rats. Journal of Chromatography B. 2023 Apr 15;1221:123688.https://www.sciencedirect.com/science/article/pii/S1570023223000983
- Chappell DL, Lee AY, Castro-Perez J, Zhou H, Roddy TP, Lassman ME, Shankar SS, Yates NA, Wang W, Laterza OF. An ultrasensitive method for the quantitation of active and inactive GLP-1 in human plasma via immunoaffinity LC–MS/MS. Bioanalysis. 2014 Jan 1;6(1):33-42.https://www.tandfonline.com/doi/abs/10.4155/bio.13.280
- Zhang H, Xin B, Caporuscio C, Olah TV. Bioanalytical strategies for developing highly sensitive liquid chromatography/tandem mass spectrometry based methods for the peptide GLP‐1 agonists in support of discovery PK/PD studies. Rapid Communications in Mass Spectrometry. 2011 Nov 30;25(22):3427-35.https://analyticalsciencejournals.onlinelibrary.wiley.com/doi/abs/10.1002/rcm.5241
- Choi HI, Jeong HC, Jeong JW, Lee J, Kim DH, Ko KC, Chae YJ, Lee KR. Development and validation of an LC-MS/MS method for Tirzepatide, a dual GIP/GLP-1 receptor agonist, in rat plasma for application to a pharmacokinetic study. Journal of Chromatography B. 2025 Oct 26:124836.https://www.sciencedirect.com/science/article/pii/S1570023225003903


