Introduction
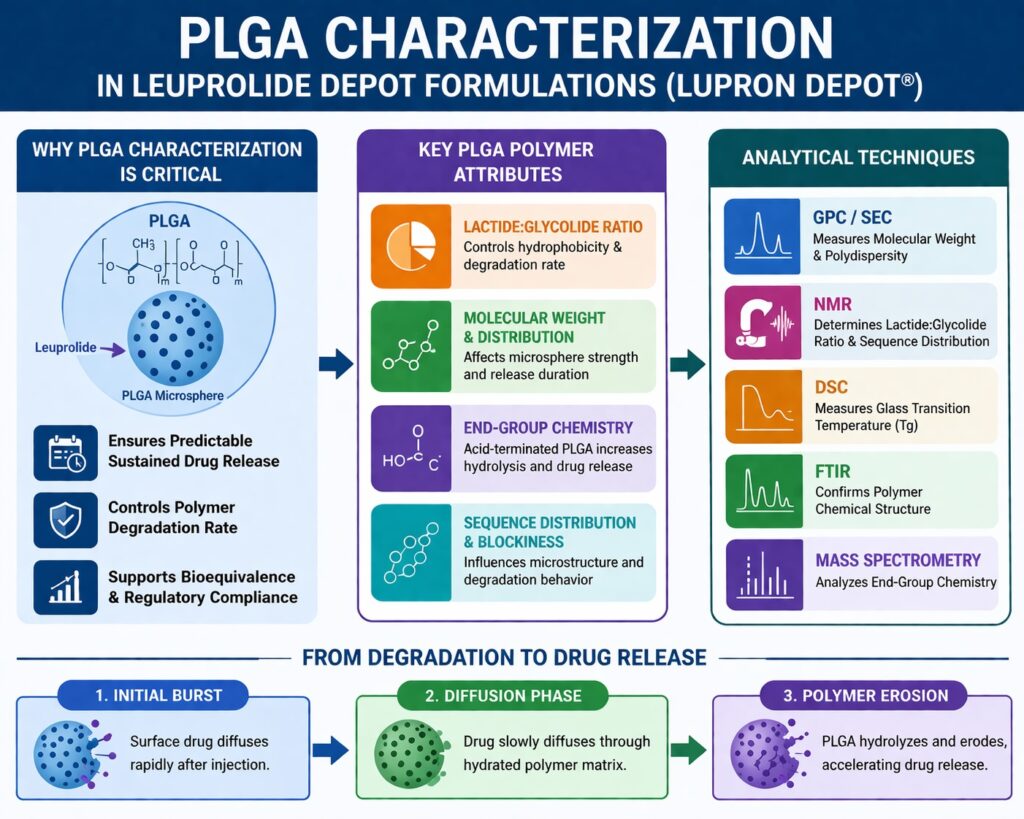
PLGA Characterization Lupron Depot is essential because the polymer’s structural and physicochemical properties directly influence how leuprolide is released from long-acting microsphere formulations. In products like Lupron Depot®, poly(lactic-co-glycolic acid) (PLGA) forms biodegradable microspheres that slowly degrade after injection. As the polymer gradually breaks down, the drug is released in a controlled and sustained manner over weeks or months. Another important feature of PLGA is its biodegradability. The polymer breaks down into lactic acid and glycolic acid, which are naturally processed by the body. Because of this safe degradation pathway, PLGA is widely used in long-acting injectable drug delivery systems. It is also highly compatible with peptide drugs such as leuprolide.
Expert Guidance on Formulation: Learn How to Develop Generic Leuprolide Depot
Unlike simple polymer analysis, PLGA characterization in leuprolide depot formulations must connect polymer properties with drug release behavior and microsphere degradation. Small differences in molecular weight, copolymer ratio, or end-group chemistry can change how the polymer degrades and how the drug diffuses. Since depot products release drugs over long periods, these differences may affect clinical performance. Microsphere structure, polymer crystallinity, and water absorption also influence degradation pathways. These factors determine how the polymer interacts with body fluids and how the drug travels through the matrix. Understanding these relationships allows formulation scientists to design stable and predictable drug delivery systems.
Deep Dive into Polymer Science: Explore PLGA Characterization for Reference Listed Drugs (RLD)
For this reason, advanced analytical strategies are needed to ensure consistent polymer quality and reproducible drug release. Proper characterization helps identify variability early in development and ensures reliable manufacturing. Ultimately, this analytical foundation supports safe and effective long-acting therapies.
Share via:
Summary:
- PLGA polymer properties directly control the drug release behavior in leuprolide depot formulations such as Lupron Depot®.
- Critical characterization parameters include lactide:glycolide ratio, molecular weight distribution, end-group chemistry, polymer microstructure, and degradation kinetics.
- Advanced analytical techniques such as GPC/SEC, NMR spectroscopy, DSC, FTIR, and mass spectrometry are essential to determine polymer attributes impacting peptide release.
- Variability in PLGA source, intrinsic viscosity, and sequence distribution can significantly affect microsphere stability and pharmacokinetic profiles.
- Accurate PLGA characterization in Lupron Depot formulations ensures regulatory compliance, bioequivalence evaluation, and reproducible long-acting drug delivery.
- Regulatory agencies including the FDA require detailed polymer characterization data for reference and generic depot formulations.
Why PLGA Characterization is Critical for Lupron Depot Formulations
The main goal of PLGA Characterization Lupron Depot is to ensure predictable polymer degradation and controlled leuprolide release during the dosing period. Since these injections are given less frequently than standard therapies, maintaining a consistent release rate is extremely important. Polymer characterization confirms that the formulation will perform as expected inside the body.
Leuprolide depot products use microsphere-based controlled release technology. As the PLGA polymer gradually hydrolyzes, small pores develop in the microspheres. These pores allow the drug to slowly diffuse out of the polymer matrix. Therefore, the degradation rate of PLGA directly controls the drug release profile.
Characterization also helps scientists understand how polymer properties influence release behavior. By measuring parameters such as molecular weight and copolymer composition, researchers can predict polymer performance during storage and after injection. These insights are valuable during formulation development and manufacturing scale-up.
Key reasons PLGA characterization is essential:
- Ensuring consistent degradation rate of the polymer matrix
- Maintaining controlled release of leuprolide over 1–6 months
- Supporting generic drug development and bioequivalence studies
- Identifying batch-to-batch variability in polymer raw materials
- Meeting regulatory expectations for long-acting injectable products
Research shows that differences in intrinsic viscosity, molecular weight, and copolymer composition can change the release kinetics of leuprolide from PLGA microspheres. These changes can occur even when other formulation variables remain the same. Therefore, precise polymer characterization is critical for reliable therapeutic performance.
Key Polymer Attributes Evaluated in PLGA Characterization Lupron Depot
Several polymer properties must be measured to fully understand how leuprolide is released from PLGA microspheres. These attributes influence water absorption, polymer degradation, and drug diffusion through the matrix. Accurate measurement of these factors helps scientists design depot formulations with predictable performance.
Polymer properties also affect manufacturing processes such as emulsification, solvent evaporation, and microsphere drying. Changes in polymer structure may influence particle size, encapsulation efficiency, and mechanical stability. Because of this, detailed polymer characterization is required for both product development and quality control.
1. Lactide:Glycolide Ratio in PLGA Characterization Lupron Depot
The lactide:glycolide (L:G) ratio determines the hydrophobicity and degradation rate of PLGA polymers. This ratio represents the proportion of lactic acid and glycolic acid units in the copolymer chain. Because these two monomers have different chemical properties, their ratio strongly affects polymer behavior.
| L:G Ratio | Polymer Behavior | Impact on Leuprolide Release |
|---|---|---|
| 50:50 | Fast degradation | Faster drug release |
| 65:35 | Moderate degradation | Sustained release |
| 75:25 or higher | Slower degradation | Extended release |
Higher lactide content increases polymer hydrophobicity and slows hydrolysis. This slows down water penetration and delays polymer breakdown. As a result, formulations with higher lactide content usually support longer dosing intervals.
The L:G ratio can also influence polymer crystallinity and mechanical strength. Balanced compositions often degrade more uniformly, while highly lactide-rich polymers degrade more slowly but steadily. Choosing the correct ratio is therefore essential for designing effective depot formulations.
2. Molecular Weight and Distribution
Molecular weight is one of the most important factors affecting PLGA degradation and microsphere stability. Polymers with higher molecular weight contain longer chains that require more hydrolysis steps before they break down. Because of this, high molecular weight PLGA generally degrades more slowly.
Key parameters include:
- Number-average molecular weight (Mn)
- Weight-average molecular weight (Mw)
- Polydispersity index (PDI)
Higher molecular weight polymers often provide longer drug release and stronger microsphere structure. These properties help maintain stability during storage and after injection. However, very high molecular weight may slow drug diffusion excessively.
Technical Insight: Understanding PLGA Polymer Molecular Weight and PDI
Broad molecular weight distribution can cause uneven degradation. Some chains may degrade quickly while others remain intact, leading to inconsistent drug release. For this reason, controlling molecular weight distribution is important for reliable product performance.
Advanced Analysis: The Role of PLGA PDI in Pharmaceutical Formulations
3. Polymer End-Group Chemistry in PLGA Characterization Lupron Depot
PLGA polymers may be ester-terminated or acid-terminated, depending on the polymerization process used during manufacturing. These end groups affect how easily the polymer undergoes hydrolysis. Even small changes in end-group chemistry can influence degradation behavior.
Acid-terminated polymers usually degrade faster because they promote autocatalytic hydrolysis. The acidic groups accelerate further breakdown of polymer chains inside microspheres. This effect can increase polymer erosion and accelerate drug release.
End-group chemistry becomes particularly important when developing generic versions of Lupron Depot. Small differences in polymer chemistry may lead to measurable changes in drug release behavior. Because of this, regulatory agencies require careful analysis of polymer end groups.
4. Sequence Distribution and Blockiness
Although PLGA is considered a random copolymer, the arrangement of lactide and glycolide units can vary depending on polymerization conditions. These structural differences influence polymer microstructure and degradation behavior. Studying sequence distribution helps scientists better predict polymer performance.
Important parameters include sequence length, randomness, and blockiness index. These features describe how monomers are arranged along the polymer chain. Random distributions usually lead to more uniform degradation patterns.
These structural variations can influence water penetration, chain mobility, and hydrolysis rate inside microspheres. Uneven sequence distribution may lead to irregular degradation and variable drug release. Analytical methods such as NMR spectroscopy are commonly used to study copolymer microstructure.
Analytical Techniques Used in PLGA Characterization Lupron Depot
Reliable PLGA Characterization Lupron Depot requires several complementary analytical methods. No single technique can fully describe the complex structure of PLGA polymers used in depot formulations. Scientists therefore combine multiple techniques to obtain a complete understanding of polymer properties.
These analytical tools provide information about molecular weight, chemical structure, thermal properties, and polymer composition. Together, they build a comprehensive profile of the polymer material used in microsphere formulations.
Major Analytical Methods
| Analytical Technique | Purpose |
|---|---|
| Gel Permeation Chromatography (GPC/SEC) | Molecular weight and polydispersity |
| Nuclear Magnetic Resonance (NMR) | Copolymer composition and sequence distribution |
| Differential Scanning Calorimetry (DSC) | Glass transition temperature |
| Fourier Transform Infrared Spectroscopy (FTIR) | Polymer chemical structure |
| Mass Spectrometry | End-group analysis |
| Intrinsic Viscosity Measurement | Polymer chain length estimation |
Using multiple analytical techniques improves the reliability of polymer characterization and helps detect small differences between polymer batches.
Development Partnership: Find the right CRO for Leuprolide Depot Development
GPC/SEC Analysis
Size exclusion chromatography (SEC), also called gel permeation chromatography (GPC), is widely used to measure PLGA molecular weight distribution. The technique separates polymer chains according to their size in solution. This allows accurate determination of Mn, Mw, and PDI.
In Lupron Depot formulations, GPC is often used to monitor polymer degradation during release studies. By measuring molecular weight changes over time, researchers can track how the polymer breaks down in physiological conditions. This information helps explain the drug release pattern.
GPC analysis is also useful for comparing polymer batches and verifying manufacturing consistency. Similar GPC profiles suggest stable polymer quality and predictable performance. Because of this, GPC is commonly used during both product development and quality control.
NMR Spectroscopy
NMR spectroscopy is a key tool for determining lactide:glycolide ratio, copolymer sequence distribution, and polymer end groups. This technique provides detailed information about the molecular structure of PLGA.
Proton NMR (¹H NMR) allows precise measurement of polymer composition. By analyzing characteristic peaks in the spectrum, researchers can calculate the percentage of lactide and glycolide units in the polymer chain. This information is important for regulatory documentation.
NMR also helps reveal polymer microstructure and monomer arrangement along the chain. These structural insights help scientists understand differences in degradation behavior and drug release performance.
Thermal Characterization
Thermal analysis methods such as Differential Scanning Calorimetry (DSC) are used to measure the glass transition temperature (Tg) of PLGA polymers. Tg represents the temperature at which the polymer changes from a rigid glassy state to a flexible rubbery state.
The Tg of PLGA influences polymer chain mobility, microsphere stability, and drug diffusion rate. When Tg is lower, the polymer becomes more flexible at body temperature. This flexibility may increase water penetration and accelerate drug diffusion.
Lower Tg values often lead to faster early drug release, while higher Tg values maintain a rigid structure for longer periods. Understanding Tg therefore helps researchers select polymers that provide the desired sustained-release profile.
Relationship Between PLGA Degradation and Leuprolide Release
Drug release from Lupron Depot microspheres occurs through a combination of diffusion and polymer degradation. At the beginning, the drug diffuses through small pores present in the polymer matrix. As degradation continues, the polymer structure weakens and forms additional channels for drug release.
The release process usually follows three phases:
1. Initial Burst Release
- Rapid diffusion of surface-associated drug
- Influenced by microsphere morphology and polymer porosity
This phase occurs soon after injection and is mainly caused by drug molecules located near the microsphere surface. Proper formulation design helps reduce excessive burst release.
2. Diffusion-Controlled Phase
- Slow diffusion of leuprolide through hydrated polymer matrix
During this stage, the drug moves gradually through water-filled channels within the polymer. Polymer density, porosity, and chain mobility strongly influence the diffusion rate.
3. Polymer Erosion Phase
- Accelerated release due to PLGA hydrolysis and matrix breakdown
As polymer degradation progresses, large pores and cracks form within the microspheres. These openings allow the remaining drug to escape more easily.
Key polymer parameters affecting this process include molecular weight decline, autocatalytic hydrolysis, and pore formation. Understanding these relationships helps scientists design depot formulations with reliable release behavior.
Case Study Results: Leuprolide Depot Case Study: Achieving Sustained Release
Regulatory Expectations for PLGA Characterization
Regulatory agencies require detailed PLGA Characterization Lupron Depot for both reference and generic formulations. Because depot products involve complex drug delivery systems, regulators need strong analytical evidence to confirm product quality.
The FDA recommends evaluating polymer composition, molecular weight distribution, intrinsic viscosity, end-group chemistry, and degradation behavior. These attributes help confirm that the polymer used in the formulation behaves similarly to the reference product.
Both reference listed drugs (RLD) and generic versions must demonstrate comparable polymer properties. Similar polymer characteristics help ensure comparable drug release kinetics and therapeutic performance.
As a result, polymer characterization has become a key part of regulatory submissions for long-acting injectable medicines.
Compliance Guidance: Analytical Requirements for ANDA of Generic Drugs
Challenges in PLGA Characterization for Leuprolide Depot Systems
Despite advanced analytical techniques, several challenges remain when characterizing PLGA polymers used in depot formulations. These challenges arise from polymer complexity and interactions between the polymer and the drug.
1. Polymer Source Variability
PLGA produced by different manufacturers may have small differences in monomer purity, polymerization process, or molecular weight distribution. Even when the nominal composition is the same, these variations can influence polymer microstructure.
Such differences may affect microsphere formation and drug release behavior. Therefore, selecting consistent polymer suppliers is important for stable product performance.
2. Autocatalytic Degradation
PLGA degradation produces lactic acid and glycolic acid, which accelerate further polymer hydrolysis. This process creates an acidic microenvironment inside microspheres.
The internal acidity may lead to uneven degradation, peptide instability, and unpredictable release patterns. Careful formulation design is required to control this effect.
3. Polymer–Peptide Interactions
Leuprolide may interact with PLGA through ionic interactions, surface adsorption, or local pH changes. These interactions can affect drug encapsulation efficiency and release behavior.
Understanding these interactions is important when interpreting polymer characterization data. Comprehensive analysis helps distinguish polymer effects from drug-related factors.
Safety Protocols: Nitrosamine Risk Assessment in Generic Drugs
Emerging Analytical Approaches in PLGA Characterization Lupron Depot
Recent research has introduced advanced analytical techniques that improve PLGA characterization accuracy. These technologies allow scientists to study polymer behavior in greater detail.
Examples include UV imaging for monitoring drug diffusion, high-resolution mass spectrometry for end-group analysis, micro-NMR for copolymer sequencing, and advanced SEC detectors for degradation studies. These methods provide higher sensitivity and improved structural insights.
By combining these modern tools with traditional analytical methods, researchers can better understand PLGA-based drug delivery systems. This integrated approach supports the development of safer and more effective long-acting injectable therapies.
Conclusion
PLGA Characterization Lupron Depot formulations is a critical analytical step for ensuring controlled drug release, product consistency, and regulatory compliance in long-acting leuprolide therapies. Since the polymer matrix controls drug diffusion and degradation, understanding polymer properties is essential for successful formulation design.
Key polymer attributes such as lactide:glycolide ratio, molecular weight distribution, polymer end groups, and degradation kinetics directly affect microsphere stability and drug release patterns. Careful analysis of these properties allows scientists to design predictable controlled-release systems.
Analytical techniques including GPC, NMR, DSC, FTIR, and mass spectrometry provide valuable insights into polymer structure and behavior. Together, these tools help optimize depot formulations and ensure consistent manufacturing quality.
As regulatory requirements for complex generics continue to evolve, advanced PLGA characterization strategies will remain essential for the development and evaluation of Lupron Depot and other long-acting injectable medicines.
Strategic Development: Accelerate Generic Drug Development with a Specialized CDMO
For specialized analytical support related to polymer characterization and complex drug delivery systems, contact the experts at ResolveMass Laboratories.
Frequently Asked Questions (FAQs)
Key parameters include the lactide:glycolide ratio, molecular weight distribution, intrinsic viscosity, and polymer end groups. These properties influence polymer hydrophobicity, degradation rate, and microsphere stability. Together, they determine how the drug diffuses through the matrix and how the polymer erodes. Adjusting these properties helps achieve the desired sustained-release profile.
Key parameters include the lactide:glycolide ratio, molecular weight distribution, intrinsic viscosity, and polymer end groups. These properties influence polymer hydrophobicity, degradation rate, and microsphere stability. Together, they determine how the drug diffuses through the matrix and how the polymer erodes. Adjusting these properties helps achieve the desired sustained-release profile.
The lactide:glycolide ratio is commonly measured using proton nuclear magnetic resonance (¹H-NMR) spectroscopy. This technique allows scientists to identify and quantify monomer units in the polymer chain. By analyzing the spectral peaks, the proportion of lactide and glycolide can be calculated. NMR also provides insights into polymer structure and composition.
Higher molecular weight PLGA usually degrades more slowly because longer polymer chains require more hydrolysis steps to break apart. This slower degradation leads to extended drug release from microspheres. Lower molecular weight polymers degrade faster and may cause quicker drug diffusion. Therefore, controlling molecular weight is important for designing the intended release duration.
Polymer end groups influence how quickly PLGA undergoes hydrolysis. Acid-terminated polymers typically degrade faster than ester-terminated polymers due to autocatalytic hydrolysis. This difference can affect microsphere stability and drug release kinetics. Careful evaluation of end-group chemistry is therefore necessary during formulation development.
Regulatory agencies require detailed characterization to confirm product quality, safety, and bioequivalence. Because long-acting injectables depend heavily on polymer behavior, regulators must verify that polymer attributes match those of the reference product. Detailed data ensure consistent drug release and therapeutic performance. This information supports regulatory approval.
PLGA degradation causes the polymer matrix to gradually break down and form pores. These pores allow leuprolide to diffuse out of the microsphere over time. As degradation progresses, larger channels form and accelerate drug release. The combination of diffusion and erosion creates the sustained-release profile.
Reference:
- Swayzer, D. V., & Gerriets, V. (2023). Leuprolide. In StatPearls. StatPearls Publishing. https://www.ncbi.nlm.nih.gov/books/NBK551662/
- U.S. Food and Drug Administration. (2012). Lupron Depot (leuprolide acetate for depot suspension) prescribing information (NDA 019732). https://www.accessdata.fda.gov/drugsatfda_docs/label/2012/019732s038lbl.pdf
- Chwalisz, K. (2023). Clinical development of the GnRH agonist leuprolide acetate depot. F&S Reports, 4(2 Suppl), 33–39. https://doi.org/10.1016/j.xfre.2022.11.011



