PRODUCT NAME
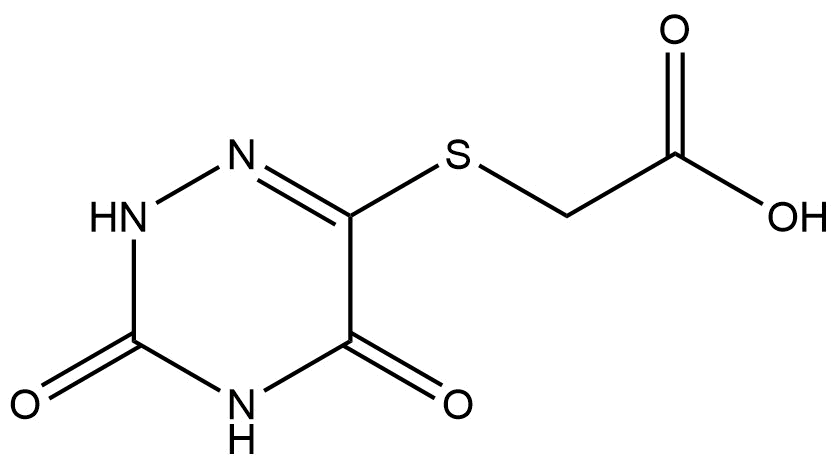
2-((3,5-Dioxo-2,3,4,5-tetrahydro-1,2,4-triazin-6-yl)thio)acetic Acid
CAS Number: 88891-73-0
OVERVIEW
2-((3,5-dioxo-2,3,4,5-tetrahydro-1,2,4-triazin-6-yl)thio)acetic acid | CAS 88891-73-0 is a specialized heterocyclic sulfur-containing organic compound of high interest in chemical research and development. Its unique combination of a triazinone core and a thioacetic acid moiety makes it a valuable intermediate in the synthesis of pharmaceuticals, agrochemicals, and advanced molecular scaffolds.
The compound belongs to the class of triazinones, specifically 1,2,4-triazin-6-ones, which are widely used in medicinal chemistry due to their ability to interact with a variety of biological targets. The appended thioacetic acid functionality introduces both nucleophilicity and chelating potential, enhancing the compound’s applicability in organic transformations.
ResolveMass Laboratories offers this product with assured purity, traceability, and consistent batch-to-batch quality, suitable for both industrial and academic research needs.
CHEMICAL STRUCTURE AND PROPERTIES
Molecular Formula: C6H6N4O4S
Molecular Weight: 203.17 g/mol
The structure integrates:
-
A 1,2,4-triazin-6-one core, featuring two carbonyl (C=O) groups at positions 3 and 5.
-
A thioether linkage at the 6-position, connecting the triazinone ring to the acetic acid group.
-
A terminal acetic acid moiety, imparting acidic functionality and potential for salt formation.
Key physicochemical aspects include:
-
Moderate polarity due to carbonyl and carboxyl groups.
-
Potential for hydrogen bonding as donor and acceptor.
-
The presence of sulfur imparts distinctive reactivity in substitution and coordination chemistry.
The compound may exist in multiple tautomeric or protonation states depending on pH, which can influence its solubility, reactivity, and interaction with biological systems.
SYNTHETIC UTILITY AND APPLICATIONS
Pharmaceutical Intermediates
Owing to its heterocyclic structure and functional handles, this compound is widely used as a building block in medicinal chemistry. It can be elaborated into:
-
Antiviral and anticancer lead structures — triazine derivatives have demonstrated activity against a range of targets.
-
Enzyme inhibitors — the carbonyl and thioacetic acid groups allow for interaction with enzyme active sites.
-
Prodrug precursors — modification of the acetic acid group facilitates the tuning of pharmacokinetic properties.
The triazinone scaffold is present in several drug classes, and the introduction of a thioether side chain enables further structural diversity through subsequent derivatization.
Agrochemical Research
In agrochemical development, 1,2,4-triazinone derivatives are explored for herbicidal, fungicidal, and insecticidal activities. The presence of a thioacetic acid side chain may improve binding to agricultural biological targets and enable formulation into complex agrochemical agents.
Organic Synthesis Intermediate
The product serves as a versatile intermediate in synthetic organic chemistry:
-
The carboxylic acid group allows for esterification, amide formation, and coupling reactions.
-
The thioether linkage can undergo oxidation to sulfoxides/sulfones or participate in substitution reactions.
-
The triazinone ring is amenable to functionalization at multiple sites.
These features make the compound a strategic choice for constructing complex molecular architectures.
Coordination Chemistry and Materials Science
The combination of nitrogen donors, carbonyl oxygens, and the thiol backbone offers coordinating sites for metal ions. This enables:
-
Ligand design for metal complexes.
-
Potential applications in catalysis and supramolecular chemistry.
-
Exploration in novel functional materials where chelation or molecular recognition is desired.
MECHANISM OF ACTION IN BIOLOGICAL TARGETING (RESEARCH CONTEXT)
While 2-((3,5-dioxo-2,3,4,5-tetrahydro-1,2,4-triazin-6-yl)thio)acetic acid itself is primarily a research intermediate, its derivatives engage biological systems in distinct ways:
-
The triazinone ring mimics nucleotide bases and has been investigated for interactions with nucleic acid-binding proteins.
-
The acetic acid group enhances water solubility and can participate in hydrogen bonding with enzymes and receptors.
-
The sulfur atom offers soft Lewis base character, enabling interactions with metal centers in bioenzymes or coordination complexes.
These modes of interaction are foundational hypotheses in early-stage drug discovery and mechanistic enzymology.
QUALITY SPECIFICATIONS AND ANALYTICAL CHARACTERIZATION
ResolveMass Laboratories provides this compound with a full suite of quality controls:
-
Purity: Typically supplied with >98% purity (or as specified by customer requirement), verified by validated analytical methods.
-
Analytical Testing: Includes but is not limited to:
-
NMR Spectroscopy
-
Mass Spectrometry (MS)
-
High-Performance Liquid Chromatography (HPLC)
-
Infrared (IR) Spectroscopy
-
-
Identity Confirmation: Through combined spectral analysis.
Each lot is accompanied by a Certificate of Analysis (CoA) detailing test results and compliance with quality standards.
SAFETY AND HANDLING (RESEARCH LAB CONTEXT)
Researchers should consult the Safety Data Sheet (SDS) before use. General laboratory practices include:
-
Use of personal protective equipment (PPE).
-
Handling within fume hoods when appropriate.
-
Proper waste disposal in accordance with institutional and regulatory guidelines.
Detailed hazard classifications and emergency measures are outlined in the SDS associated with each shipment.
CUSTOM SERVICES AND FORMULATION SUPPORT
In addition to off-the-shelf supply, ResolveMass Laboratories provides:
-
Custom synthesis services tailored to specific derivatives or analogs.
-
Route optimization for improved yield and scalability.
-
Analytical method development to support customer workflows.
Our experienced technical team can collaborate on feasibility studies and project-specific solutions.
CONCLUSION
2-((3,5-dioxo-2,3,4,5-tetrahydro-1,2,4-triazin-6-yl)thio)acetic acid | CAS 88891-73-0 is a high-value research intermediate with broad applicability across medicinal chemistry, agrochemical discovery, organic synthesis, and materials research. Its distinct functional groups and heterocyclic framework make it an indispensable tool in the synthesis of complex molecules and innovative chemical entities.
For dependable quality, reliable supply, and comprehensive technical support, ResolveMass Laboratories is your preferred partner for this and related specialty chemicals.

Reviews
There are no reviews yet.