PRODUCT OVERVIEW
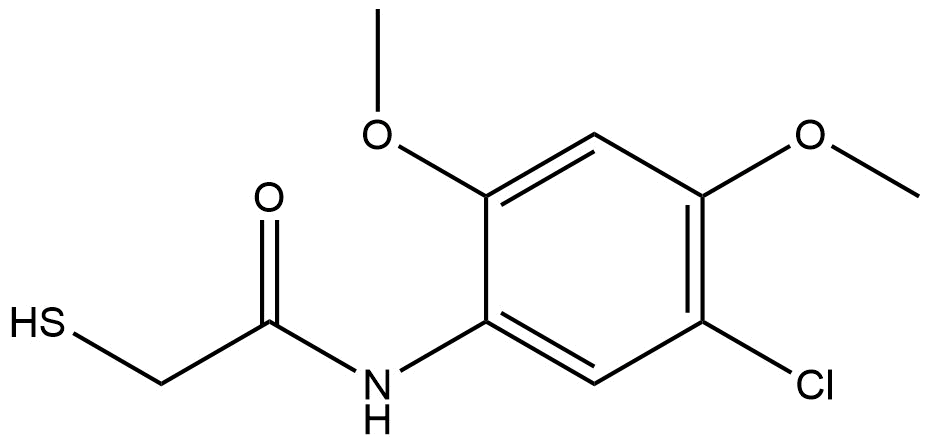
N-(5-chloro-2,4-dimethoxyphenyl)-2-mercaptoacetamide | CAS 946676-63-7 is a specialized chemical intermediate widely used in pharmaceutical, agrochemical, and materials science research. Structurally, this compound combines a substituted phenyl ring with a thiol-containing acetamide moiety, making it valuable in the synthesis of heterocyclic frameworks, ligand development, and as a key intermediate in advanced organic transformations.
This high-purity compound is supplied by ResolveMass Laboratories Inc., ensuring stringent quality control, traceability, and reliability for research and development applications. With its distinctive functional groups, N-(5-chloro-2,4-dimethoxyphenyl)-2-mercaptoacetamide is essential for chemists seeking selective reactivity and modular synthesis options.
CHEMICAL INFORMATION
-
Chemical Name: N-(5-chloro-2,4-dimethoxyphenyl)-2-mercaptoacetamide
-
CAS Number: 946676-63-7
-
Molecular Formula: C10H12ClNO3S
-
Molecular Weight: 261.72 g/mol
-
Structure: Substituted phenyl ring with chloro and dimethoxy groups, linked to a mercaptoacetamide functionality
The molecule features an aromatic ring substituted at multiple positions: chlorine at the 5-position and methoxy groups at the 2- and 4-positions. The terminal mercaptoacetamide provides a sulfur-containing functional group that is particularly reactive in nucleophilic substitution, metal coordination, and heterocycle formation reactions.
APPLICATIONS of N-(5-chloro-2,4-dimethoxyphenyl)-2-mercaptoacetamide | CAS 946676-63-7
Pharmaceutical Synthesis
N-(5-chloro-2,4-dimethoxyphenyl)-2-mercaptoacetamide is primarily used as an intermediate in the synthesis of bioactive compounds. Its thiol group can undergo cyclization, condensation, and substitution reactions, enabling the construction of:
-
Thiazole and thiadiazole rings
-
S-linked heterocycles for kinase inhibitors
-
Custom ligands for targeted drug scaffolds
Medicinal chemists leverage this intermediate to explore structure-activity relationships (SAR) and develop candidate molecules with improved pharmacokinetics and bioavailability.
Agrochemical Research
In agrochemical development, this compound aids in the design of new herbicidal and fungicidal agents through selective functionalization and ring modification strategies. Its scaffold supports the introduction of diverse substituents that influence biological efficacy.
Materials Chemistry
The sulfur-containing mercaptoacetamide group serves as a valuable ligand for metal coordination and surface functionalization. Applications include:
-
Surface modifiers for nanoparticles
-
Chelating agents in catalyst systems
-
Building blocks for polymeric materials with enhanced properties
SYNTHETIC UTILITY
This intermediate’s versatility arises from its dual reactive centers:
-
Thiol Reactivity: The mercapto group can participate in S-alkylation, Michael addition, and disulfide formation.
-
Amide Reactivity: The acetamide linkage serves as a stable connection point for further acylations or cross-coupling reactions.
-
Aromatic Substitution: The 5-chloro and methoxy substituents modulate the electron density of the aromatic ring, enabling selective functionalization via electrophilic or nucleophilic aromatic substitution techniques.
These characteristics make it a highly adaptable building block in multistep synthetic sequences.
ANALYTICAL & QUALITY ASSURANCE
At ResolveMass Laboratories Inc., product quality is validated through comprehensive analytical characterization, including:
-
High-Performance Liquid Chromatography (HPLC) purity assessment
-
Nuclear Magnetic Resonance (NMR) spectroscopy for structural confirmation
-
Mass Spectrometry (MS) verification
-
Elemental analysis to confirm stoichiometry
Each batch is accompanied by a Certificate of Analysis (CoA), providing end users with transparent documentation of analytical results and compliance specifications.
SAFETY INFORMATION
This compound should be treated as a potentially hazardous research chemical. Key safety considerations include:
-
Irritant hazards: May cause irritation to skin, eyes, and respiratory tract.
-
Toxicity Profile: Not intended for food, drug, or cosmetic use without appropriate regulatory approvals.
-
Environmental Caution: Avoid release into waterways; dispose of waste per local regulations.
WHY CHOOSE THE RESOLVEMASS PRODUCT?
ResolveMass Laboratories Inc. offers:
-
High Purity & Consistency: Rigorous QC ensures reproducible performance.
-
Reliable Supply Chain: On-time delivery with comprehensive documentation.
-
Expert Technical Support: Assistance with product selection and application support.
Whether for pharmaceutical R&D, agrochemical development, or materials science, our N-(5-chloro-2,4-dimethoxyphenyl)-2-mercaptoacetamide meets the quality standards demanded by modern research enterprises.
Reviews
There are no reviews yet.