
Introduction:
Protein Characterization is the comprehensive analysis of a protein’s structure, function, and physicochemical properties. It ensures that proteins used in pharmaceuticals are safe, effective, and consistent.
In modern biopharmaceutical development, Protein Characterization is indispensable for:
- Understanding molecular identity
- Ensuring batch-to-batch consistency
- Supporting regulatory submissions
- Confirming biosimilarity
At ResolveMass Laboratories Inc., advanced analytical platforms and scientific expertise enable precise and regulatory-aligned protein characterization strategies.
Summary:
- Protein Characterization is essential for understanding structure, function, purity, and stability of proteins.
- It plays a critical role in drug development, biosimilars, and biologics approval.
- Advanced analytical tools like LC-MS, HRMS, NMR, and electrophoresis are central to accurate characterization.
- Regulatory agencies require detailed protein profiling to ensure safety and efficacy.
- A multi-technique approach ensures robust, reproducible, and regulatory-compliant data.
1: Why is Protein Characterization Important?
Protein Characterization is important because it confirms the identity, purity, structure, and functionality of proteins used in therapeutics. It ensures that biologics are safe, effective, and consistent across development and manufacturing.
Key Reasons
- Regulatory Compliance: Required by global authorities such as FDA, EMA, and ICH to support drug approval
- Quality Assurance: Ensures batch-to-batch consistency, safety, and product reliability
- Biosimilar Development: Demonstrates analytical similarity to reference biologics
- Process Optimization: Helps refine upstream and downstream manufacturing processes
Even small structural variations—such as minor changes in folding or post-translational modifications—can significantly impact drug efficacy, stability, and immunogenicity. Hence, robust Protein Characterization is essential for minimizing risks and ensuring therapeutic success.
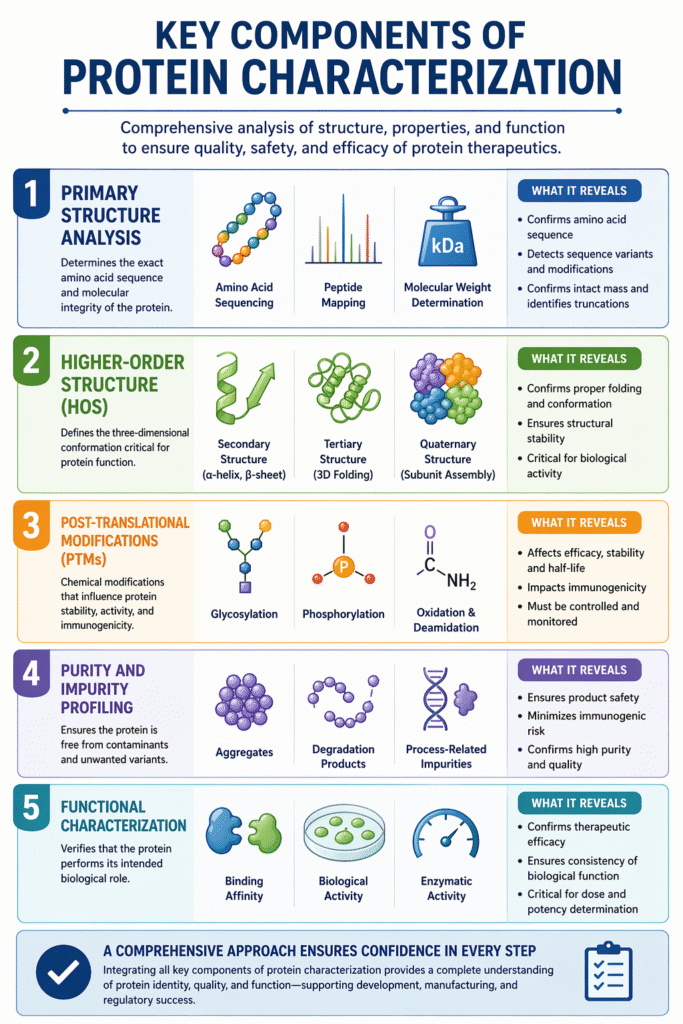
2: Key Components of Protein Characterization
Protein Characterization involves evaluating structural, physicochemical, and functional attributes to fully understand a protein’s identity and behavior. A comprehensive approach ensures accurate assessment for development, quality control, and regulatory compliance.
1. Primary Structure Analysis
Primary structure analysis confirms the exact amino acid sequence and molecular integrity of the protein.
- Amino Acid Sequencing: Determines the precise order of amino acids
- Peptide Mapping: Verifies sequence and detects modifications or inconsistencies
- Molecular Weight Determination: Confirms intact mass and detects truncations or variants
2. Higher-Order Structure (HOS)
Higher-order structure defines the three-dimensional conformation critical for protein function.
- Secondary Structure: α-helix and β-sheet arrangements
- Tertiary Structure: Overall 3D folding of a single polypeptide chain
- Quaternary Structure: Assembly of multiple subunits into a functional complex
3. Post-Translational Modifications (PTMs)
PTMs are chemical modifications that influence protein stability, activity, and immunogenicity.
- Glycosylation: Impacts stability, solubility, and biological activity
- Phosphorylation: Regulates protein function and signaling pathways
- Oxidation & Deamidation: Common degradation pathways affecting stability
4. Purity and Impurity Profiling
This component ensures the protein is free from contaminants and unwanted variants.
- Aggregates: Can impact safety and increase immunogenicity
- Degradation Products: Formed due to instability or stress conditions
- Process-Related Impurities: Residual host cell proteins, DNA, or reagents
5. Functional Characterization
Functional characterization verifies that the protein performs its intended biological role.
- Binding Affinity: Measures interaction with target molecules
- Biological Activity: Confirms therapeutic efficacy
- Enzymatic Activity: Evaluates catalytic function where applicable
A robust Protein Characterization strategy integrates all these components to deliver comprehensive, reliable, and regulatory-ready insights for biologics and biosimilars.
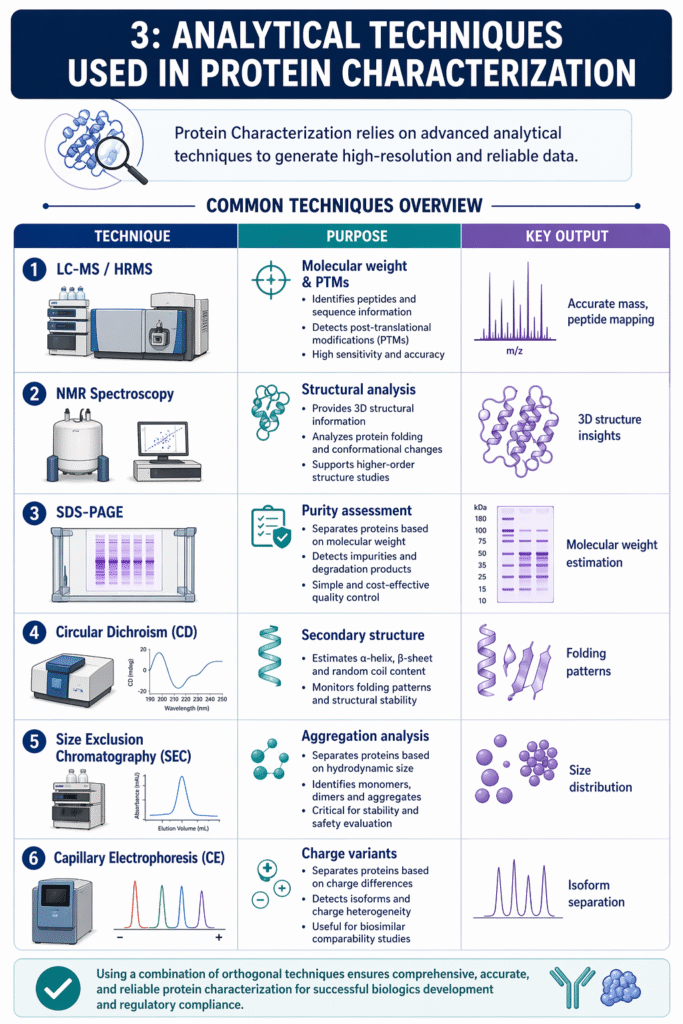
3: Analytical Techniques Used in Protein Characterization
Protein Characterization relies on advanced analytical techniques to generate high-resolution, accurate, and reproducible data. These techniques collectively provide insights into molecular structure, purity, and functional integrity.
Common Techniques Overview
| Technique | Purpose | Key Output |
|---|---|---|
| LC-MS / HRMS | Molecular weight & PTMs | Accurate mass, peptide mapping |
| NMR Spectroscopy | Structural analysis | 3D structure insights |
| SDS-PAGE | Purity assessment | Molecular weight estimation |
| Circular Dichroism (CD) | Secondary structure | Folding patterns |
| Size Exclusion Chromatography (SEC) | Aggregation analysis | Size distribution |
| Capillary Electrophoresis (CE) | Charge variants | Isoform separation |
Key Techniques Explained
1. LC-MS / HRMS
LC-MS and HRMS are essential for precise mass determination and detection of post-translational modifications.
- Enables peptide mapping and sequence confirmation
- Detects trace-level impurities and variants
- Provides high sensitivity and specificity
2. NMR Spectroscopy
NMR spectroscopy provides detailed insights into the three-dimensional structure of proteins.
- Analyzes protein folding and conformational changes
- Useful for higher-order structure (HOS) studies
- Supports comparability assessments
3. SDS-PAGE
SDS-PAGE is a widely used method for evaluating protein purity and approximate molecular weight.
- Separates proteins based on size
- Identifies degradation products and impurities
- Simple and cost-effective quality control tool
4. Circular Dichroism (CD)
CD spectroscopy evaluates the secondary structure and folding characteristics of proteins.
- Detects α-helix and β-sheet content
- Monitors structural stability under stress conditions
5. Size Exclusion Chromatography (SEC)
SEC is used to assess protein aggregation and size distribution.
- Identifies monomers, dimers, and higher aggregates
- Critical for stability and safety evaluation
6. Capillary Electrophoresis (CE)
CE separates proteins based on charge differences to analyze isoforms and variants.
- Detects charge heterogeneity
- Useful for biosimilar comparability studies
4: Role of Mass Spectrometry in Protein Characterization
Mass spectrometry is a cornerstone of Protein Characterization because it provides highly sensitive and accurate molecular-level insights into protein structure and composition. It enables detailed analysis that is critical for biologics, biosimilars, and complex therapeutic proteins.
Key Applications
- Peptide Mapping: Confirms the amino acid sequence by analyzing enzymatically digested protein fragments, ensuring structural integrity.
- PTM Identification: Detects subtle post-translational modifications such as glycosylation, oxidation, and deamidation that can impact protein function and stability.
- Impurity Profiling: Identifies trace-level impurities, degradation products, and process-related contaminants with high sensitivity.
- Intact Mass Analysis: Verifies the complete molecular weight of the protein, helping confirm overall structure and detect variants or truncations.
Why HRMS is Critical
High-resolution mass spectrometry (HRMS) enables detection of even the smallest structural variations. This is especially important for:
- Regulatory submissions, where detailed molecular characterization is mandatory
- Biosimilar development, where precise comparison with reference biologics is required
- Complex biologics, including monoclonal antibodies and peptides
5: Challenges in Protein Characterization
Protein Characterization presents challenges due to structural complexity and sensitivity of proteins.
Common Challenges:
- Structural heterogeneity
- Labile post-translational modifications
- Low-level impurity detection
- Protein aggregation and instability
- Complex peptide mapping for large proteins
Mitigation Strategies:
- Use of orthogonal analytical techniques
- Advanced sample preparation methods
- High-resolution instrumentation
- Robust method development and validation
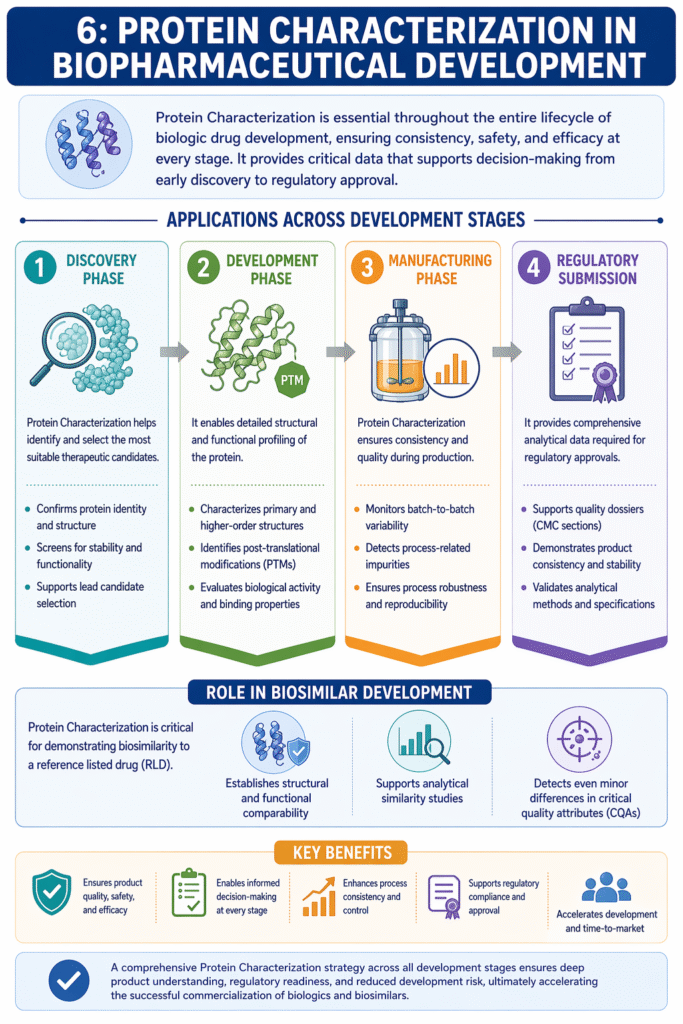
6: Protein Characterization in Biopharmaceutical Development
Protein Characterization is essential throughout the entire lifecycle of biologic drug development, ensuring consistency, safety, and efficacy at every stage. It provides critical data that supports decision-making from early discovery to regulatory approval.
Applications Across Development Stages
1. Discovery Phase
Protein Characterization helps identify and select the most suitable therapeutic candidates.
- Confirms protein identity and structure
- Screens for stability and functionality
- Supports lead candidate selection
2. Development Phase
It enables detailed structural and functional profiling of the protein.
- Characterizes primary and higher-order structures
- Identifies post-translational modifications (PTMs)
- Evaluates biological activity and binding properties
3. Manufacturing Phase
Protein Characterization ensures consistency and quality during production.
- Monitors batch-to-batch variability
- Detects process-related impurities
- Ensures process robustness and reproducibility
4. Regulatory Submission
It provides comprehensive analytical data required for regulatory approvals.
- Supports quality dossiers (CMC sections)
- Demonstrates product consistency and stability
- Validates analytical methods and specifications
Role in Biosimilar Development
Protein Characterization is critical for demonstrating biosimilarity to a reference listed drug (RLD).
- Establishes structural and functional comparability
- Supports analytical similarity studies
- Detects even minor differences in critical quality attributes (CQAs)
7: Regulatory Expectations for Protein Characterization
Regulatory agencies require comprehensive and scientifically justified Protein Characterization data to ensure the safety, efficacy, and quality of biologic products. A robust analytical package is essential for successful regulatory approval.
Key Guidelines
- ICH Q6B: Defines specifications and acceptance criteria for biotechnological and biological products
- ICH Q5E: Provides guidance on demonstrating comparability after manufacturing changes
- FDA Biosimilar Guidelines: Outline requirements for analytical similarity and biosimilar approval pathways
These guidelines emphasize a risk-based and science-driven approach to Protein Characterization.
Regulatory Focus Areas
- Structural Integrity:
Confirmation of primary structure and higher-order structure (HOS) to ensure correct protein folding and identity - Impurity Profiling:
Identification and quantification of process-related impurities, aggregates, and degradation products - Functional Activity:
Demonstration of biological activity, potency, and mechanism of action - Stability Data:
Evaluation of protein stability under various conditions to establish shelf life and storage requirements
Why It Matters
A well-documented Protein Characterization strategy builds regulatory confidence and significantly improves approval success rates. It ensures:
- Reduced risk of regulatory queries or delays
- Complete understanding of critical quality attributes (CQAs)
- Strong support for CMC documentation
8: Best Practices for Effective Protein Characterization
Effective Protein Characterization requires a strategic, multi-technique, and validated approach.
Recommended Practices:
- Use orthogonal methods for confirmation
- Implement validated analytical protocols
- Maintain data integrity and traceability
- Perform forced degradation studies
- Ensure high sensitivity and specificity
9: How ResolveMass Laboratories Inc. Supports Protein Characterization
ResolveMass Laboratories Inc. provides advanced, reliable, and regulatory-compliant Protein services.
Core Capabilities:
- High-resolution LC-MS/HRMS analysis
- Comprehensive peptide mapping and sequencing
- Detailed impurity and PTM profiling
- Expertise in complex biologics and peptides
- Customized analytical strategies for ANDA and biosimilars
With a strong scientific foundation and cutting-edge instrumentation, ResolveMass ensures accurate, reproducible, and submission-ready data.
Conclusion:
Protein Characterization is a critical pillar of modern biopharmaceutical development, ensuring safety, efficacy, and regulatory compliance.
From primary structure to functional activity, a comprehensive characterization strategy enables:
- Better product understanding
- Reduced development risks
- Faster regulatory approvals
As biologics and biosimilars continue to grow, the importance of robust Protein Characterization will only increase.
Frequently Asked Questions:
Protein Characterization is the process of analyzing a protein’s structure, function, purity, and physicochemical properties. It ensures the protein meets quality, safety, and regulatory requirements.
Protein Characterization ensures that biologic drugs are safe, effective, and consistent. Even small structural changes can impact efficacy or cause immunogenic reactions. It helps maintain batch-to-batch consistency during manufacturing. Regulatory agencies also require detailed characterization data for approval. Without it, product quality and patient safety cannot be guaranteed.
Peptide mapping is a technique where proteins are enzymatically digested into smaller peptides and analyzed, usually by LC-MS. It confirms the amino acid sequence and identifies modifications. This method is highly sensitive and can detect even small changes in structure. It is critical for verifying protein identity. It also supports comparability and regulatory submissions.
Mass spectrometry provides highly accurate and sensitive molecular data. It is used for peptide mapping, intact mass analysis, and PTM identification. It can detect trace impurities and structural variants. High-resolution MS enables precise measurement of molecular weight. This makes it a key tool in modern protein analysis.
Higher-order structure refers to the three-dimensional arrangement of a protein. It includes secondary, tertiary, and quaternary structures. This structure determines how the protein functions biologically. Techniques like NMR and CD are used to study HOS. Any change in HOS can affect protein activity and stability.
Protein Characterization is critical for proving similarity between a biosimilar and its reference product. It compares structural, functional, and impurity profiles. This ensures that the biosimilar performs similarly in clinical use. Regulatory agencies rely heavily on analytical similarity data. It reduces the need for extensive clinical trials.
Reference
- Higgins MJ, Novak JT. Characterization of exocellular protein and its role in bioflocculation. Journal of environmental engineering. 1997 May;123(5):479-85.https://ascelibrary.org/doi/abs/10.1061/(ASCE)0733-9372(1997)123:5(479)
- Yan C, Wu F, Jernigan RL, Dobbs D, Honavar V. Characterization of protein–protein interfaces. The protein journal. 2008 Jan;27(1):59-70.https://link.springer.com/article/10.1007/s10930-007-9108-x
- Li F, Zhang D, Fujise K. Characterization of fortilin, a novel antiapoptotic protein. Journal of Biological Chemistry. 2001 Dec 14;276(50):47542-9.https://www.jbc.org/article/S0021-9258(19)37294-1/fulltext
- Reid GE, McLuckey SA. ‘Top down’protein characterization via tandem mass spectrometry. Journal of mass spectrometry. 2002 Jul;37(7):663-75.https://analyticalsciencejournals.onlinelibrary.wiley.com/doi/abs/10.1002/jms.346


