Introduction: Why LC-MS/MS Is Central to Proving Biosimilarity Using LC-MS
To Prove Biosimilarity Using LC-MS, companies must meet strict scientific and regulatory requirements. This is not just a routine lab task—it is a critical step required by global health authorities. Organizations like the FDA Office of Pharmaceutical Quality and the EMA Committee for Medicinal Products for Human Use expect developers to demonstrate analytical similarity at a deep molecular level. The most reliable way to Prove Biosimilarity Using LC-MS is through liquid chromatography-tandem mass spectrometry (LC-MS/MS). Without strong LC-MS/MS data, a biosimilarity submission is often considered incomplete.
Biosimilars are far more complex than small-molecule drugs. They include monoclonal antibodies, fusion proteins, and cytokines. These molecules depend heavily on their structure to work properly. Elements such as amino acid sequence, three-dimensional structure, post-translational modifications (PTMs), and glycosylation patterns all affect safety and performance. Because of this complexity, no single method can fully describe them. However, LC-MS/MS comes closest by offering detailed, high-resolution analysis across multiple attributes.
This article explains how to Prove Biosimilarity Using LC-MS, covering regulatory expectations, key analytical steps, and a clear LC-MS/MS workflow. It also reflects the proven strategies used by ResolveMass Laboratories Inc. to produce accurate, submission-ready data. Each section is written to help you understand how analytical evidence supports both regulatory approval and scientific confidence.
Explore Our Services: Comprehensive Biosimilar Characterization Using Mass Spectrometry
Share via:
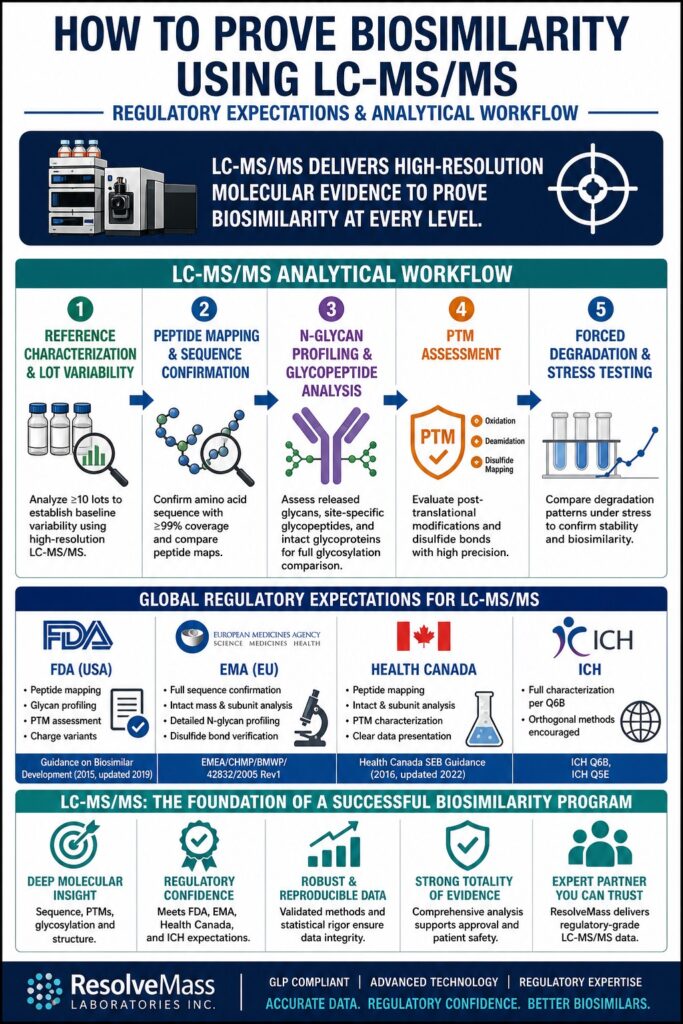
Regulatory Expectations for Proving Biosimilarity Using LC-MS/MS
Global regulatory agencies require strong LC-MS/MS data to support biosimilarity claims. The FDA, EMA, and Health Canada all emphasize that analytical comparisons must be thorough, well-structured, and supported by statistics. To Prove Biosimilarity Using LC-MS, developers must not only generate data but also explain it clearly within a scientific framework.
1. FDA Expectations (United States)
The FDA uses a “fingerprint-like” approach when evaluating biosimilars. Multiple batches of the reference product are compared with the biosimilar across key quality attributes. LC-MS/MS is essential for confirming structural consistency.
Tier 1 Attributes: Require strict statistical testing. LC-MS/MS is used for sequence confirmation, peptide mapping, and glycan profiling. These directly impact clinical outcomes and must closely match.
Tier 2 Attributes: Evaluated using acceptable ranges. LC-MS/MS assesses PTMs such as oxidation and deamidation, which affect stability.
Tier 3 Attributes: Focus on descriptive data. LC-MS/MS supports primary structure analysis, while other tools examine higher-order structure.
This structured approach helps regulators focus on the most critical quality attributes while still reviewing the full dataset.
2. EMA Expectations (European Union)
The EMA strongly supports the use of advanced LC-MS/MS technologies. High-resolution systems such as Orbitrap and Q-TOF are preferred because they provide precise molecular data.
Key requirements include:
- Full sequence confirmation using bottom-up or middle-down techniques
- Intact mass and subunit analysis
- Detailed N-glycan profiling at both released and site-specific levels
- Disulfide bond verification
The EMA also expects developers to use modern, high-performance tools to support biosimilarity claims.
3. Health Canada Expectations (Canada)
Health Canada follows ICH guidelines, especially Q6B. Developers must show that the biosimilar matches the reference product in all structural aspects.
LC-MS/MS is required for:
- Peptide mapping
- Intact and subunit mass analysis
- PTM characterization
Clear data presentation and strong scientific reasoning are essential for approval in Canada.
Need Compliance Guidance? Regulatory Support for Generic and Biosimilar Drug Development
| Regulatory Body | Required LC-MS/MS Analysis | Key Guidance Document |
| FDA (USA) | Peptide mapping, glycan profiling, PTM assessment, charge variants | FDA Guidance on Biosimilar Development (2015, updated 2019) |
| EMA (EU) | Bottom-up/middle-down sequencing, intact mass, N-glycan profiling, disulfide mapping | EMEA/CHMP/BMWP/42832/2005 Rev1 |
| Health Canada | Primary/secondary structure confirmation, PTM characterization | Health Canada SEB Guidance (2016, updated 2022) |
| ICH | Full characterization per Q6B; orthogonal methods encouraged | ICH Q6B, ICH Q5E |
Step-by-Step LC-MS/MS Analytical Workflow to Prove Biosimilarity Using LC-MS
A structured workflow is necessary to Prove Biosimilarity Using LC-MS effectively. Each step adds a layer of evidence that supports the final conclusion.
Phase 1: Reference Product Characterization and Lot Variability Analysis
This step defines the comparison baseline. At least ten batches of the reference product are tested to understand natural variability.
High-resolution LC-MS/MS generates detailed data such as sequence coverage, glycan profiles, and PTM patterns. This baseline helps define what “similar” means in real regulatory terms.
Phase 2: Peptide Mapping and Sequence Confirmation
Peptide mapping confirms the amino acid sequence and detects any differences.
LC-MS/MS provides accurate peptide identification with over 99% sequence coverage. Side-by-side comparison of peptide maps ensures alignment between the biosimilar and reference product.
Accelerate Your Pipeline: Analytical Services for Generic Drug Development
Phase 3: N-Glycan Profiling and Glycopeptide Analysis
Glycosylation plays a major role in how biologics function.
LC-MS/MS analyzes glycans at multiple levels:
- Released glycans for overall composition
- Glycopeptides for site-specific details
- Intact proteins for full glycoform distribution
Matching these profiles is essential for proving biosimilarity.
| Analysis Level | Sample Prep | LC Mode | Key MS Information |
| Released N-glycan | PNGase F digestion; 2-AB or RapiFluor-MS labeling | HILIC or RP-HPLC | Glycan composition, antennarity, fucosylation, sialylation levels |
| Glycopeptide (site-specific) | Trypsin digestion (no glycan release) | RP-HPLC | Site-specific glycoform occupancy, macro- and micro-heterogeneity |
| Intact/Subunit glycoprotein | IdeS + DTT (Fc/2, Fab, LC) | RP or SEC-MS | Global glycoform distribution; charge state envelope analysis |
Phase 4: Post-Translational Modification (PTM) Assessment
PTMs must be measured and compared carefully.
LC-MS/MS detects changes such as oxidation and deamidation with high precision. Disulfide bond mapping confirms correct protein folding. These checks ensure stability and performance remain consistent.
Phase 5: Forced Degradation and Comparability Under Stress
Stress testing shows how products behave under harsh conditions.
LC-MS/MS tracks how proteins degrade over time. Similar degradation patterns between products provide strong evidence of biosimilarity and stability.
Specialized Peptide Support: Liraglutide Generic Development Services
or Lanreotide Generic Development Services
Method Qualification and Validation: Non-Negotiable Before Submission
All analytical methods must be qualified or validated before use. This ensures data is accurate, consistent, and reliable.
Qualification confirms the method works as intended. Validation proves it meets full regulatory standards. System suitability tests are run before each analysis to maintain quality. Reproducibility across labs is also important, especially for global submissions.
These steps ensure that the data used to Prove Biosimilarity Using LC-MS can stand up to regulatory review.
From Lab to Market: Manufacturing Scale-Up for Generic and Biosimilar Drugs
Choosing the Right LC-MS/MS Platform to Prove Biosimilarity Using LC-MS
Selecting the correct instrument is critical for success.
- Orbitrap systems are ideal for peptide mapping and PTM analysis
- Q-TOF instruments work well for intact mass and glycan studies
- Triple quadrupole systems are used for targeted quantitation
The right platform improves accuracy, efficiency, and data reliability.
| Instrument Type | Best Application | Mass Resolution | Regulatory Preference |
| Orbitrap (e.g., Exploris 480) | Bottom-up peptide mapping, PTM quantitation, DIA acquisition | ≥ 120,000 FWHM | FDA, EMA (preferred for HRAM) |
| Q-TOF (e.g., Waters G2-XS) | Intact mass, middle-up, glycan profiling | 20,000–40,000 FWHM | EMA, Health Canada |
| Triple Quadrupole (QqQ) | Targeted PTM quantitation, MRM-based comparability | Unit resolution | FDA Tier 2 equivalence testing |
| Orbitrap Eclipse (ETD-capable) | Disulfide mapping, glycopeptide characterization, HOS-informed fragmentation | ≥ 240,000 FWHM | FDA, EMA (advanced HOS) |
Statistical Data Interpretation and Totality-of-Evidence Assembly
Data alone is not enough. It must be interpreted correctly.
Quality attributes are ranked by importance. Statistical tools such as equivalence testing compare datasets. Techniques like principal component analysis (PCA) help visualize similarity.
All results are combined into a totality-of-evidence report. This includes LC-MS/MS data along with other analytical findings to create a complete and convincing case.
Advanced Molecule Support: Generic Peptide and Oligonucleotide Projects
ResolveMass Laboratories Inc.: Regulatory-Grade LC-MS/MS for Biosimilarity Programs
ResolveMass Laboratories Inc. is a trusted CRO specializing in mass spectrometry analysis of biologics and biosimilars. The company operates under GLP standards and uses compliant data systems.
Services include early-stage analysis, regulatory submission support, and post-approval studies. Their team combines technical expertise with advanced LC-MS/MS platforms to deliver high-quality results.
ResolveMass Laboratories helps clients successfully Prove Biosimilarity Using LC-MS by providing accurate, regulatory-ready data aligned with global expectations.
Get Started Today: Formulation Development for Generic Drug Development
Conclusion
To Prove Biosimilarity Using LC-MS, developers must combine advanced technology with a strong analytical strategy. LC-MS/MS provides detailed insight into molecular structure, including sequence, glycosylation, and degradation patterns.
However, success depends on more than just instruments. Proper method validation, structured workflows, and clear data interpretation are all essential. Companies that invest early in LC-MS/MS analysis improve their chances of regulatory approval.
ResolveMass Laboratories Inc. offers the expertise and infrastructure needed to support every stage of biosimilar development, delivering reliable data that meets global regulatory standards.
Targeted Development: Expertise in Generic Peptide Semaglutide Projects
Frequently Asked Questions (FAQs)
Regulatory agencies generally expect at least 99% sequence coverage in peptide mapping studies. If any part of the sequence is not covered, it must be clearly explained and evaluated for its impact on product safety and function. In many cases, scientists use multiple digestion enzymes to improve coverage and ensure even difficult regions are properly analyzed.
A minimum of 10 reference product lots is commonly recommended, especially by the FDA for strong statistical comparison. Other agencies expect enough samples to reflect natural variability in the original product. Using too few lots may lead to unreliable conclusions and can weaken the overall biosimilarity claim.
LC-MS/MS is very effective for studying primary structure and modifications, but it cannot fully confirm higher-order structure on its own. Additional techniques such as circular dichroism, DSC, and HDX-MS are required for a complete understanding. However, advanced MS methods like native MS can still provide helpful supporting data.
These approaches differ in how much the protein is broken down before analysis. Bottom-up breaks proteins into small peptides for detailed mapping, while middle-up analyzes larger fragments for better structural context. Top-down studies the intact protein, giving a full overview but requiring more advanced instruments and data processing.
Deamidation can occur naturally or during sample preparation, so careful method control is essential. Scientists reduce artificial changes by optimizing digestion conditions such as pH, time, and temperature. Additional controls and alternative digestion methods are often used to confirm whether the modification is real or introduced during analysis.
The FDA recommends the two one-sided tests (TOST) approach for Tier 1 attributes. This method uses defined equivalence margins and confidence intervals to compare biosimilar and reference products. It is important to set these limits before starting the study and ensure enough samples are included for reliable statistical results.v
Released glycan analysis provides useful overall information, but it is usually not enough on its own. Site-specific glycopeptide analysis is also needed to understand how glycans are distributed across the molecule. This is especially important for complex biologics where glycosylation can affect function and stability.
Forced degradation studies show how a product behaves under stress conditions. If the biosimilar and reference product degrade in similar ways, it supports structural similarity. These studies also help confirm that analytical methods can detect important changes in the molecule.
Reference:
- Segu, Z., Stone, T., Berdugo, C., Roberts, A., Doud, E., & Li, Y. (2020). A rapid method for relative quantification of N-glycans from a therapeutic monoclonal antibody during trastuzumab biosimilar development. MAbs, 12(1), 1750794. https://pmc.ncbi.nlm.nih.gov/articles/PMC7188402/
- Nupur, N., Joshi, S., Guillarme, D., & Rathore, A. S. (2022). Analytical similarity assessment of biosimilars: Global regulatory landscape, recent studies and major advancements in orthogonal platforms. Frontiers in Bioengineering and Biotechnology, 10, 832059. https://pmc.ncbi.nlm.nih.gov/articles/PMC8865741/
- D’Atri, V., Guillarme, D., & Beck, A. (2025). Biopharmaceutical analysis—current analytical challenges, limitations, and perspectives. Analytical and Bioanalytical Chemistry. https://pmc.ncbi.nlm.nih.gov/articles/PMC12783247/
- Berkowitz, S. A., Engen, J. R., Mazzeo, J. R., & Jones, G. B. (2012). Analytical tools for characterizing biopharmaceuticals and the implications for biosimilars. Nature Reviews Drug Discovery, 11(7), 527–540. https://pmc.ncbi.nlm.nih.gov/articles/PMC3714370/



