
Summary:
- Scientific Objective: To prove that a synthetic generic version of Retatrutide is molecularly identical to the Reference Listed Drug (RLD) across its 39-amino acid primary sequence and its higher-order solution structure.
- Primary Structure Fidelity: 100% sequence coverage is achieved through high-resolution mass spectrometry (HRMS) and LC-MS/MS sequencing, specifically verifying the C20 fatty diacid moiety at Lys17.
- Impurity Profiling Standards: Regulatory compliance requires the identification of all impurities 0.10% and a hard cap of 0.5% for any new impurity not found in the RLD.
- Higher-Order Structure (HOS): Multidimensional 1D and 2D NMR (HSQC, TOCSY, NOESY) provide the “structural fingerprint” required to statistically prove conformational equivalence to the RLD.
- Regulatory Pathway: Synthetic peptides referencing rDNA-origin drugs are evaluated via the 505(j) Abbreviated New Drug Application (ANDA) pathway, provided “sameness” is analytically demonstrated.
- ResolveMass Expertise: ResolveMass Laboratories Inc. provides FDA-registered, ISO 9001-certified analytical packages that eliminate regulatory blind spots and support rapid ANDA approval.
1. Introduction
A Retatrutide Sameness Study is the mandatory analytical validation required to prove that a generic synthetic peptide has the exact same active ingredient as its Reference Listed Drug (RLD). This falls under broader regulatory frameworks such as Peptide Sameness Study for ANDA and Active Ingredient Sameness in ANDA.
As a 39-amino acid triple hormone receptor agonist (GLP-1, GIP, and Glucagon receptors), Retatrutide represents a highly complex molecule. Even minor variations can impact safety and efficacy. At ResolveMass Laboratories Inc., we follow advanced Analytical Strategies for Sameness Study supported by Orthogonal Analytical Techniques for ANDA Sameness Evaluation.
Retatrutide’s structural complexity is comparable to other GLP-1 analogs like Liraglutide Sameness Study, Semaglutide Sameness Study, and newer peptides such as Tirzepatide Sameness Study and Exenatide Sameness Study.
2. Analytical Strategy Overview (Multi-Technique Approach)
An effective analytical strategy for a peptide sameness study must utilize multiple independent scientific principles to eliminate analytical blind spots. ResolveMass employs an orthogonal approach, meaning we use different techniques (e.g., MS vs. NMR) to verify the same quality attributes, providing the “weight of evidence” required by regulatory agencies like the FDA and EMA.
A robust Retatrutide sameness study relies on a multi-dimensional analytical approach aligned with FDA Peptide Sameness Study Requirements.
| Quality Attribute | Primary Technique | Orthogonal Confirmation |
| Primary Sequence | LC-MS/MS Sequencing | Peptide Mapping / AAA |
| Impurity Profile | RP-UPLC-HRMS | Capillary Electrophoresis (CE) |
| Secondary Structure | 2D NMR (NOESY/HSQC) | Circular Dichroism (CD) |
| Aggregation State | Dynamic Light Scattering (DLS) | Size-Exclusion (SEC-MALS) |
| Molecular Weight | High-Resolution Orbitrap MS | Intact Mass Deconvolution |
This aligns with industry best practices for Sameness Evaluation of Synthetic Peptides for ANDA.
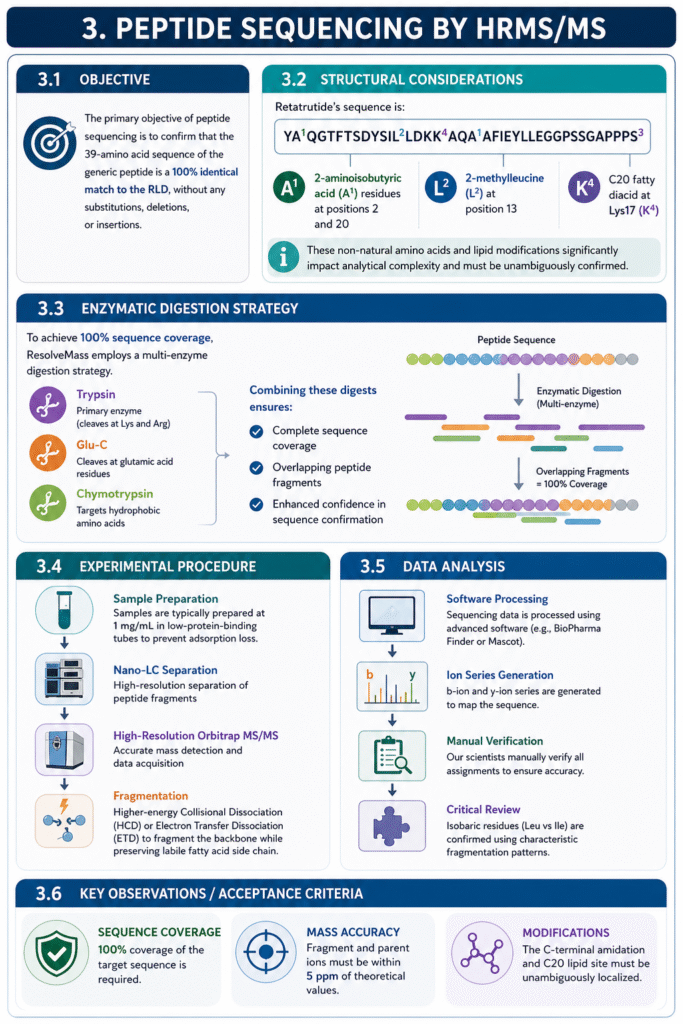
3. Peptide Sequencing by HRMS/MS
3.1 Objective
The primary objective of peptide sequencing is to confirm that the 39-amino acid sequence of the generic peptide is a 100% identical match to the RLD, without any substitutions, deletions, or insertions.
3.2 Structural Considerations
Retatrutide’s sequence is: YA^{1}QGTFTSDYSIL^{2}LDKK^{4}AQA^{1}AFIEYLLEGGPSSGAPPPS^{3} Critical features that must be verified include the 2-aminoisobutyric acid (A^1) residues at positions 2 and 20, the 2-methylleucine (L^2) at position 13, and the C20 fatty diacid at Lys17 (K^4).
Advanced LC-MS/MS sequencing ensures full coverage, similar to studies conducted in Liraglutide Sameness Study for ANDA Submission and Octreotide Sameness Study for ANDA Submission.
3.3 Enzymatic Digestion Strategy
To achieve 100% coverage, ResolveMass employs a multi-enzyme approach. While Trypsin is the standard, Retatrutide’s unique sequence often requires Glu-C or Chymotrypsin to penetrate regions with low basic residue density. Combining these digests ensures that every peptide fragment is overlapping and verified.
3.4 Experimental Procedure
Samples are typically prepared at 1 mg/mL in low-protein-binding tubes to prevent adsorption loss. We utilize nano-LC coupled to High-Resolution Orbitrap MS/MS, using Higher-energy Collisional Dissociation (HCD) or Electron Transfer Dissociation (ETD) to fragment the peptide backbone while preserving the labile fatty acid side chain.
3.5 Data Analysis
Sequencing data is processed using advanced software (e.g., BioPharma Finder or Mascot), which generates a b- and y-ion series to map the sequence. Our scientists manually verify all assignments to ensure that isobaric residues (like Leu and Ile) are correctly identified using characteristic fragmentation patterns.
3.6 Key Observations / Acceptance Criteria
- Sequence Coverage: 100% coverage of the target sequence is required.
- Mass Accuracy: Fragment and parent ions must be within 5 ppm of theoretical values.
- Modifications: The C-terminal amidation and C20 lipid site must be unambiguously localized.
4. Peptide Mapping (Comparative Fingerprinting)
Peptide mapping acts as a structural fingerprint comparison between generic and RLD.
This approach is a core component of Sameness Evaluation in ANDA.
4.1 Objective
Peptide mapping serves as a “molecular fingerprint” to demonstrate that the generic and RLD have identical structural compositions when digested into fragments.
4.2 Strategy
This is a Multi-Attribute Method (MAM) where the intact peptide is enzymatically cleaved into a set of characteristic peptides, which are then compared side-by-side between the generic and RLD batches.
4.3 Experimental Workflow
- Denaturation/Reduction: Unfolding the peptide to expose all cleavage sites.
- Enzymatic Digestion: Overnight incubation with Trypsin or Glu-C.
- LC-MS Analysis: Separation via RP-UPLC followed by HRMS detection.
4.4 LC Conditions
We use high-efficiency C18 columns (e.g., 130 A) with a shallow acetonitrile gradient (5% to 45%) over 45-60 minutes to ensure the separation of closely related variants.
4.5 Data Analysis
The software performs peak-to-peak matching between the generic and RLD chromatograms. Mirror plots and overlay chromatograms are generated to visually and quantitatively assess comparability.
4.6 Key Observations / Acceptance Criteria
- Fingerprint Match: No “new” peaks should be present in the generic sample that aren’t in the RLD.
- Retention Time Drift: Peaks for identical fragments must elute within a narrow window (typically <0.2 min).
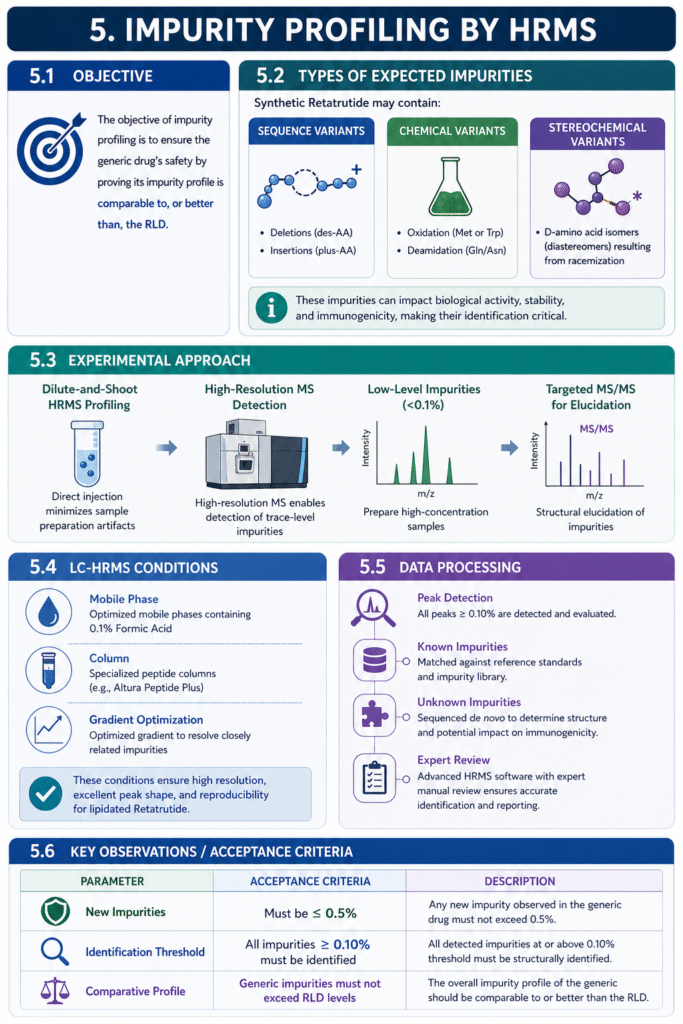
5. Impurity Profiling by HRMS
5.1 Objective
The objective of impurity profiling is to ensure the generic drug’s safety by proving its impurity profile is comparable to, or better than, the RLD.
5.2 Types of Expected Impurities
Synthetic Retatrutide may contain:
- Sequence variants: Deletions (des-AA) or insertions (plus-AA).
- Chemical variants: Oxidation (Met or Trp) and deamidation (Gln/Asn).
- Stereochemical variants: D-amino acid isomers (diastereomers) resulting from racemization.
Failure to meet these criteria is a common issue highlighted in Peptide Sameness Study Deficiencies.
5.3 Experimental Approach
We utilize a “dilute-and-shoot” HRMS profiling method for high-sensitivity detection. For low-level impurities (<0.1%), we prepare high-concentration samples and utilize targeted MS/MS for structural elucidation.
5.4 LC-HRMS Conditions
Optimized mobile phases containing 0.1% Formic Acid and specialized columns (e.g., Altura Peptide Plus) are used to manage the hydrophobicity of lipidated Retatrutide.
5.5 Data Processing
All peaks 0.10% are identified and characterized. Unknown impurities are sequenced de novo to determine their structure and potential impact on immunogenicity.
5.6 Key Observations / Acceptance Criteria
- New Impurities: Any new impurity must be 0.5%.
- Identification Threshold: All impurities 0.10% must be identified.
- Comparison: Impurities in the generic should not exceed the levels found in the RLD.
6. Intact Mass Analysis
6.1 Objective
Intact mass analysis confirms the overall molecular weight of the peptide to ensure the primary structure is intact and the correct salt form is present.
6.2 Method
HRMS (Orbitrap) is used to analyze the undigested peptide. The raw spectra (showing multiple charge states) are deconvoluted to determine the monoisotopic and average molecular weights.
6.3 Observations
The observed mass of Retatrutide (approx. 4731 Da) must match the theoretical value within sub-5 ppm. This is the first step in proving the Retatrutide Sameness Study meets regulatory requirements.
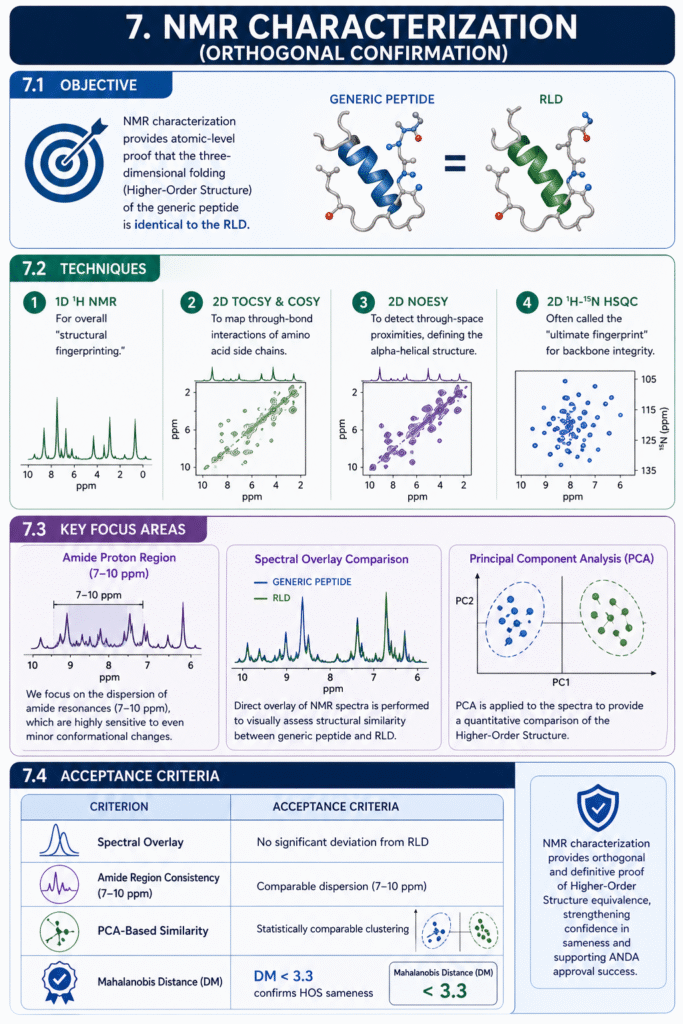
7. NMR Characterization (Orthogonal Confirmation)
7.1 Objective
NMR characterization provides atomic-level proof that the three-dimensional folding (Higher-Order Structure) of the generic peptide is identical to the RLD.
This step is critical to avoid regulatory delays under FDA Peptide Sameness Study Requirements.
7.2 Techniques
- 1D ^1H NMR: For overall “structural fingerprinting.”
- 2D TOCSY & COSY: To map through-bond interactions of amino acid side chains.
- 2D NOESY: To detect through-space proximities, defining the alpha-helical structure.
- 2D ^1H-^{15}N HSQC: Often called the “ultimate fingerprint” for backbone integrity.
7.3 Key Focus Areas
We focus on the dispersion of amide resonances (7-10 ppm), which are highly sensitive to even minor conformational changes. Principal Component Analysis (PCA) is applied to the spectra to provide a quantitative comparison.
7.4 Acceptance Criteria
The Mahalanobis distance (DM) calculated from PCA of the NMR spectra must be less than 3.3 to statistically confirm HOS sameness.
8. Comparative Sameness Assessment (RLD vs Test)
A comparative sameness assessment involves side-by-side testing of at least three batches of the generic product against three batches of the RLD. This assessment integrates data from all previous sections (MS, NMR, mapping) into a single report that demonstrates equivalence in primary, secondary, and higher-order structures, as well as purity and biological activity. ResolveMass ensures that these studies are conducted at both release and end-of-shelf-life to guarantee performance consistency.
These studies are routinely performed under specialized services such as:
9. Regulatory Considerations for ANDA
The regulatory pathway for generic Retatrutide is governed by the 505(j) ANDA pathway for highly purified synthetic peptides. While bioequivalence is often self-evident for injectables (Q1/Q2 sameness), the “active ingredient sameness” is the most scrutinized portion of the CMC (Chemistry, Manufacturing, and Controls) section. Failure to identify impurities at the 0.10% level or lacking HOS data (like 2D NMR) are common reasons for Complete Response Letters (CRLs). ResolveMass’s FDA-registered facility (FEI: 3042696771) ensures all data meets the ALCOA+ principles of data integrity required for global submissions.
These are central to Active Ingredient Sameness in ANDA and Sameness Evaluation in ANDA.
10. Conclusion
The Retatrutide Sameness Study represents one of the most advanced analytical challenges in peptide generic development. By leveraging:
- HRMS sequencing
- Peptide mapping
- Impurity profiling
- 2D NMR characterization
pharmaceutical companies can meet stringent regulatory expectations.
ResolveMass Laboratories Inc. delivers comprehensive solutions aligned with global standards, minimizing risks associated with Peptide Sameness Study Deficiencies and accelerating ANDA approvals.
Through scientifically rigorous methodologies and regulatory expertise, we ensure your synthetic peptide is not just similar—but molecularly identical to the innovator drug.
Frequently Asked Questions:
The study is crucial because regulatory authorities require proof that the generic drug contains the same active ingredient as the innovator product. Without demonstrating sameness, ANDA approval cannot be granted. It ensures there are no structural or impurity-related risks. The study minimizes chances of immunogenicity or reduced efficacy. It also strengthens the Chemistry, Manufacturing, and Controls (CMC) section. This makes the submission more robust and regulatory-compliant.
A combination of orthogonal analytical techniques is used to ensure comprehensive characterization. LC-MS/MS is applied for peptide sequencing and structural verification. HRMS is used for intact mass confirmation and impurity profiling. Peptide mapping provides comparative fingerprinting with the RLD. NMR (1D and 2D) confirms higher-order structure and conformational integrity. Additional tools like CE, DLS, and CD may also be used. This multi-technique approach ensures accuracy and reliability.
Regulatory guidelines require strict control and identification of impurities in peptide drugs. All impurities present at or above 0.10% must be identified and characterized. Any new impurity not found in the RLD must not exceed 0.5%. The impurity profile of the generic should be comparable to or better than the RLD. Advanced HRMS techniques are used for sensitive detection. This ensures product safety and compliance with global regulatory standards.
Peptide sequence identity is confirmed using high-resolution LC-MS/MS sequencing. The peptide is enzymatically digested into fragments and analyzed for sequence verification. A full 100% sequence coverage is required to ensure no amino acid mismatch. Fragment ions (b and y ions) are matched against theoretical values. Mass accuracy must typically fall within ±5 ppm. Special attention is given to modifications like fatty acid conjugation. This confirms exact structural identity with the RLD.
Reference
- Coskun T, Wu Q, Schloot NC, Haupt A, Milicevic Z, Khouli C, Harris C. Effects of retatrutide on body composition in people with type 2 diabetes: a substudy of a phase 2, double-blind, parallel-group, placebo-controlled, randomised trial. The Lancet Diabetes & Endocrinology. 2025 Aug 1;13(8):674-84.https://www.thelancet.com/journals/landia/article/PIIS2213-8587(25)00092-0/abstract
- Sinha B, Ghosal S. Efficacy and Safety of GLP‐1 Receptor Agonists, Dual Agonists, and Retatrutide for Weight Loss in Adults With Overweight or Obesity: A Bayesian NMA. Obesity. 2025 Nov;33(11):2046-54.https://onlinelibrary.wiley.com/doi/abs/10.1002/oby.24360
- Sanyal AJ, Kaplan LM, Frias JP, Brouwers B, Wu Q, Thomas MK, Harris C, Schloot NC, Du Y, Mather KJ, Haupt A. Triple hormone receptor agonist retatrutide for metabolic dysfunction-associated steatotic liver disease: a randomized phase 2a trial. Nature Medicine. 2024 Jul;30(7):2037-48.https://www.nature.com/articles/s41591-024-03018-2
- Hitaka K, Sugawara T, Matsumoto M, Nio Y. Efficacy of GLP-1 analog peptides, semaglutide, tirzepatide, and retatrutide on MC4R deficient obesity and their comparison: Animal Models. International Journal of Obesity. 2026 Feb 21:1-0.https://www.nature.com/articles/s41366-026-02025-2
- Kaur M, Misra S. A review of an investigational drug retatrutide, a novel triple agonist agent for the treatment of obesity. European Journal of Clinical Pharmacology. 2024 May;80(5):669-76.https://link.springer.com/article/10.1007/s00228-024-03646-0


