Introduction:
Establishing a successful pharmaceutical mirror of the reference listed drug requires a comprehensive Reverse Engineering Lupron Depot strategy that combines materials science, polymer chemistry, and advanced manufacturing controls. ResolveMass Laboratories Inc. approaches this challenge by recognizing that leuprolide acetate for depot suspension is not a simple drug product but a complex delivery system. In this system, the manufacturing process directly affects therapeutic performance. The goal of Reverse Engineering Lupron Depot is not only to identify ingredients but also to understand the internal structure responsible for sustained release. This includes studying formulation composition, processing conditions, and structural changes during degradation.
Learn more about the technical framework: Explore the Generic Drug Development Process for ANDA
The complexity of the Lupron Depot system comes from using poly(lactic-co-glycolic acid) (PLGA) to encapsulate leuprolide acetate, a water-soluble GnRH agonist, within a biodegradable matrix. This matrix slowly breaks down through hydrolysis. The release profile is triphasic, including an initial burst, a lag phase, and a secondary erosion-controlled phase. Reverse engineering must therefore analyze polymer molecular weight, end-group chemistry, and internal pore structure. Even small changes in these parameters can alter water penetration, polymer degradation, and drug diffusion pathways. A well-designed Reverse Engineering Lupron Depot program evaluates formulation and processing together to achieve functional equivalence.
Deep dive into the specialized requirements for this product: Understand the specific ANDA Requirements for Leuprolide Depot
Share via;
Summary:
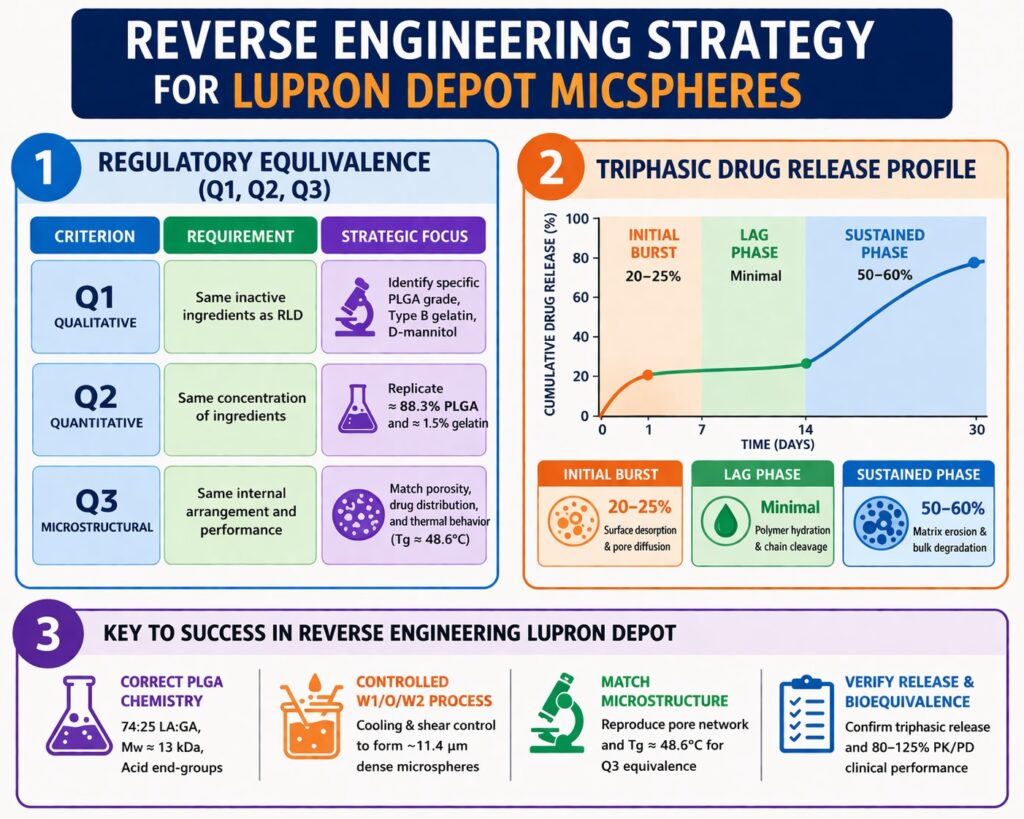
- Reverse Engineering Lupron Depot requires matching Q1, Q2, and Q3 equivalence, including composition, concentration, and internal microsphere microstructure to achieve regulatory approval.
- The formulation relies on acid-capped PLGA microspheres, where LA:GA ratio, molecular weight, and end-group chemistry control degradation and triphasic drug release.
- Manufacturing using the W1/O/W2 double emulsion process, cooling step, and controlled solvent removal is critical for replicating particle size and internal morphology.
- Microstructural equivalence must be confirmed using advanced techniques such as FIB-SEM, SR-µCT imaging, and thermal analysis to match pore structure and Tg.
- Drug release follows a triphasic profile (burst, lag, and erosion phases), and accurate replication requires controlling porosity, polymer hydration, and degradation kinetics.
- Bioequivalence depends on matching in vitro release, peptide stability, and clinical PK/PD endpoints, including sustained exposure and testosterone suppression.
Regulatory Rationale for Q1, Q2, and Q3 Equivalence in Reverse Engineering Lupron Depot
Regulatory approval for generic leuprolide acetate microspheres depends on demonstrating qualitative (Q1), quantitative (Q2), and microstructural (Q3) equivalence. FDA product-specific guidance for leuprolide emphasizes that PLGA-based injectable products must use the same polymer type and excipients at identical concentrations as the reference listed drug. This ensures predictable in vivo performance and consistent hormonal suppression for prostate cancer, endometriosis, and precocious puberty. Achieving equivalence requires both analytical characterization and reproducible manufacturing. Developers must also show that particle size, morphology, and drug distribution do not impact in vivo performance.
Pharmaceutical Equivalence Criteria for Lupron Depot
| Criterion | Regulatory Requirement | Strategic Implication for Reverse Engineering |
|---|---|---|
| Q1 (Qualitative) | Same inactive ingredients as RLD | Identifying specific PLGA grades, Type B gelatin, and D-mannitol |
| Q2 (Quantitative) | Same concentration of ingredients | Replicating the ≈ 88.3% PLGA and ≈ 1.5% gelatin composition |
| Q3 (Microstructural) | Same internal arrangement and performance | Matching porosity, drug distribution, and thermal behavior (Tg) |
While Q1 and Q2 sameness can be confirmed using analytical methods, Q3 microstructural equivalence is more challenging. Internal microstructure, including pore connectivity and spatial drug distribution, determines polymer erosion and drug diffusion. Reverse engineering therefore uses high-resolution imaging, thermal analysis, and modeling tools. These techniques help verify whether the generic microsphere behaves like the reference product. Achieving Q3 sameness supports consistent in vitro release and bioequivalence outcomes.
Technical Resource: Analytical Challenges in Leuprolide Depot Development
Polymer Matrix Deconstruction: The Role of PLGA Chemistry in Reverse Engineering Lupron Depot
Replicating the Lupron Depot release profile depends on selecting and characterizing the correct PLGA polymer matrix. PLGA is a copolymer made from lactic acid and glycolic acid that degrades into non-toxic metabolites. For a 1-month depot, the polymer must provide controlled delivery for about 30 days. Performance depends on lactide-to-glycolide ratio, molecular weight, and end-group chemistry. Each parameter affects water uptake, polymer mobility, and degradation kinetics. Careful polymer characterization is required to reproduce the intended release behavior.
LA:GA Ratio and Degradation Kinetics
The ratio of lactic acid to glycolic acid controls polymer hydrophobicity and glass transition temperature (Tg). Lactic acid increases hydrophobicity and slows water penetration compared with glycolic acid. Reverse engineering typically identifies a 75:25 LA:GA ratio for the 1-month depot. ResolveMass confirmed a composition of 74.3/25.7 using Proton Nuclear Magnetic Resonance. Maintaining this ratio is critical because small changes alter crystallinity and degradation. Accurate control ensures predictable hydration and release.
Molecular Weight Distribution and Polydispersity
Polymer molecular weight influences mechanical strength and timing of erosion. For Lupron Depot, the weight-average molecular weight is about 13.0\text{ kDa} and number-average molecular weight is about $8.7\text{ kDa}$. This results in a polydispersity index of approximately 1.5. A narrow distribution ensures uniform degradation across microspheres. Wider distributions may lead to inconsistent release behavior. Gel Permeation Chromatography is used to measure these values.
The Criticality of Acid End-Groups
Lupron Depot uses acid-capped PLGA with terminal carboxyl groups. These groups interact ionically with the basic leuprolide peptide. This interaction anchors the drug within the matrix and reduces initial burst release. Non-aqueous titration confirms an acid number near $12.9\text{ mg KOH/g}$. These interactions also improve peptide stability during storage. Replicating end-group chemistry is essential for matching release behavior.
Looking for development support? Review our Leuprolide Depot Generic Drug Development Services
PLGA Critical Quality Attributes (CQAs) for 1-Month Depot
| Attribute | Measured Value (RLD) | Impact on Performance |
|---|---|---|
| LA:GA Ratio | 74.3 ± 0.1 / 25.7 ± 0.1 | Controls hydrophobicity and Tg |
| Mw (Weight Avg) | 13.04 ± 0.06 kDa | Determines onset of bulk erosion |
| PDI (Mw/Mn) | ≈ 1.5 | Ensures uniform degradation across batches |
| End-Group | Carboxylic Acid (Acid-capped) | Enhances drug-polymer ionic interactions |
| Tg (Glass Trans.) | 48.6 ± 0.1 °C | Influences storage stability and release |
Manufacturing Engineering: Replicating the W1/O/W2 Double Emulsion in Reverse Engineering Lupron Depot
Replicating Lupron Depot requires understanding the W1/O/W2 double emulsion solvent evaporation process. In this method, the drug is dissolved in an internal aqueous phase, emulsified into a polymer organic phase, and then dispersed into a second aqueous phase. The process is sensitive to homogenization speed, temperature, and solvent removal rate. Small deviations can change particle size and morphology. Reverse engineering identifies the shear conditions and thermal environment used in the reference product. Careful control ensures reproducible microsphere formation.
The Viscosity-Enhancing Cooling Mechanism
A key step in Reverse Engineering Lupron Depot is cooling during primary emulsion formation. Because leuprolide acetate is highly water-soluble, it tends to diffuse into the external phase. Cooling increases viscosity and traps internal droplets. This reduces drug leakage and improves encapsulation efficiency. Although gelatin contributes to viscosity, cooling plays the dominant role. Implementing this step is essential for matching the RLD.
Homogenization Shear and Particle Size Distribution
The median particle size of Lupron Depot is about 11.4\ \mu\text{m}. This size supports injectability through 22-25 gauge needles. Achieving this distribution requires precise control of homogenization speed. Primary emulsification may exceed 17,500\text{ rpm} followed by controlled secondary emulsification. A wide distribution increases surface area and burst release. Maintaining a narrow span ensures predictable kinetics.
Need a development partner? Find the right CRO for Leuprolide Depot Development
Solvent Removal and “In-Water Drying” Kinetics
The rate of dichloromethane removal influences microsphere porosity. Rapid removal creates pores that promote uncontrolled diffusion. Lupron Depot uses gradual in-water drying where solvent diffuses slowly. This allows polymer annealing and formation of dense microspheres. Residual solvent levels remain below $1\text{ ppm}$. Controlled solvent removal is critical for achieving the correct microstructure.
Microstructural Q3 Equivalence in Reverse Engineering Lupron Depot
Microstructural equivalence involves matching spatial drug distribution and pore network. For complex generics, Q1 and Q2 sameness alone is not enough. Manufacturing leaves a structural fingerprint that controls release. Reverse engineering therefore uses advanced imaging techniques. These methods quantify pore connectivity and density gradients. Achieving similar morphology ensures comparable hydration and erosion.
Focused Ion Beam Scanning Electron Microscopy (FIB-SEM)
FIB-SEM provides high-resolution 3D imaging of microsphere interiors. A gallium ion beam mills thin layers while SEM captures images. These images are reconstructed to analyze pore distribution. Lupron Depot shows a dense polymer core with minimal voids. This supports near zero-order release. Matching these features is essential.
Synchrotron Radiation X-ray Microcomputed Tomography (SR-μCT)
SR-μCT provides non-destructive 3D imaging of large populations. High-energy X-rays reveal internal density variations. This allows statistical comparison between generic and RLD batches. The technique detects variability not visible by SEM. Combining SR-μCT with FIB-SEM strengthens characterization. Together they confirm microstructural sameness.
Thermal Analysis and Tg Shifts
Differential Scanning Calorimetry measures glass transition temperature. The 1-month depot shows a Tg around 48.6\text{°C}. This is higher than raw PLGA due to peptide-polymer interaction. Matching this shift confirms similar integration. Thermal analysis also predicts storage stability. Replicating Tg behavior is therefore critical.
Case Study Highlights: Explore the Leuprolide Depot Case Study on Sustained Release
Release Kinetics: Mastering the Triphasic Profile in Reverse Engineering Lupron Depot
Drug release follows a triphasic pattern consisting of burst, lag, and erosion phases. Each phase is governed by diffusion, hydration, and degradation mechanisms. Reverse engineering must reproduce all three phases. Minor deviations can alter release timing. In vitro release testing evaluates prototype performance. Matching the full profile ensures therapeutic consistency.
Phase I: The Initial Burst Management
The initial burst occurs within 24 hours. It represents about 20\text{–}25% of the total dose. Surface-associated drug and pore diffusion drive this phase. Controlling porosity reduces burst magnitude. Particle size and solvent removal also influence release. Proper control ensures safety and efficacy.
Phase II: The Lag Phase and Hydration
The lag phase shows minimal release while polymer hydration begins. Its duration depends on molecular weight and LA:GA ratio. Higher molecular weight extends the lag phase. Incorrect lag duration may disrupt hormonal suppression. Polymer selection must match the RLD. Hydration kinetics are critical.
Phase III: Secondary Erosion-Controlled Release
The final phase is governed by bulk erosion. Polymer chains hydrolyze and release drug steadily. This phase spans Day 7 to Day 30. Matching polymer composition ensures consistent erosion. This phase delivers most of the dose. Accurate replication is essential.
Expert Guidance: Learn How to Develop Generic Leuprolide Depot
Mechanisms of the Triphasic Release Profile
| Release Phase | Contribution | Primary Mechanism | Controlling Factors |
|---|---|---|---|
| Initial Burst | 20–25% | Surface desorption and pore diffusion | Surface area, porosity, end-groups |
| Lag Phase | Minimal | Polymer hydration and chain cleavage | LA:GA ratio, Mw, Tg |
| Sustained Phase | 50–60% | Matrix erosion and bulk degradation | Mw distribution, microclimate pH |
Peptide Stability and Drug-Polymer Interactions
Leuprolide stability inside PLGA microspheres is a major concern. As PLGA degrades, acidic byproducts reduce internal pH. This can lead to peptide degradation and acylation. Such reactions may reduce efficacy. Stability assessment is therefore essential. Formulation design must minimize acid-induced degradation.
Serine Acylation and Mitigation Strategies
Serine acylation occurs when polymer fragments react with peptide residues. This is common in PLGA systems. Reverse engineering identifies impurity profiles. Controlling molecular weight helps manage acid generation. Additives may reduce acylation risk. Monitoring impurities ensures stability.
Mitigating Risks: Nitrosamine Risk Assessment in Generic Drugs
Ion Exchange and Peptide Retention
Acidic PLGA end-groups interact with the peptide through ion exchange. This improves retention and reduces diffusion. Ionic anchoring stabilizes the drug during lag phase. It also reduces burst release. Replicating this interaction is essential. Proper chemistry ensures consistent performance.
Analytical Method Validation and Solvent Extraction
Accurate analytical methods are required for Reverse Engineering Lupron Depot. Microsphere matrices can interfere with assays. Polymer-drug interactions reduce recovery. Specialized extraction methods are needed. Validation ensures reliable quantification. Multiple techniques are typically used.
The Challenge of Multiple Extractions
Strong ionic interactions reduce extraction efficiency. Single extraction may recover only 85\text{–}90%. Multiple sequential extractions are required. Acidic buffers displace bound peptide. Up to 11 extractions may be needed. This ensures accurate quantification.
Polymer Isolation and Purification
Polymer isolation is needed for PLGA characterization. Microspheres are suspended in cold water. D-mannitol and gelatin are removed. The core is dissolved in THF. Peptide is removed by centrifugation. Polymer is precipitated and analyzed.
In Vitro Release Testing (IVRT) and Accelerated Methods
In vitro release testing evaluates triphasic kinetics. Real-time studies take weeks. Accelerated methods reduce timelines. Both approaches support formulation optimization. IVRT also supports quality control. Method validation is essential.
Real-Time Release in Biorelevant Media
Real-time IVRT is conducted in PBS at pH 7.4 and 37°C. Sink conditions must be maintained. Large volumes or flow-through cells are used. Continuous agitation improves reproducibility. These conditions simulate subcutaneous environment. Consistent sampling ensures accuracy.
Accelerated Release and the Arrhenius Equation
Accelerated release is performed at higher temperatures. Increased temperature accelerates degradation. The Arrhenius equation predicts release constants. Studies at 55\text{°C} reduce timelines to a few days. Correlation with real-time data is required. Validation ensures predictive accuracy.
Advanced Modeling: The Importance of IVIVC for Leuprolide Depot
Correlation of Real-Time vs. Accelerated Release (PBS pH 7.4)
| Temperature | Study Duration | Correlation to 37°C | Mechanism |
|---|---|---|---|
| 37°C | ≈ 30 Days | Baseline (1.0) | Real-time hydration and erosion |
| 50°C | ≈ 5.0 Days | High | Increased hydration rate |
| 55°C | ≈ 3.5 Days | Very High | Optimal for Q3 differentiation |
| 60°C | ≈ 2.2 Days | Moderate | Risk of pore closure / mechanism change |
Bioequivalence Challenges and Clinical Correlations
The goal of Reverse Engineering Lupron Depot is demonstrating clinical bioequivalence. Long-acting injectables require PK and PD endpoints. Matching in vitro release alone is insufficient. Clinical performance must confirm equivalence. Regulatory expectations are strict. Accurate replication is essential.
Pharmacokinetic Endpoints (C{max}, AUC{0-t}, AU{7-t})
Confidence intervals for PK parameters must fall within 80\text{–}125%. The AUC_{7-t} evaluates sustained exposure. Failure indicates premature erosion. Matching metrics ensures comparable exposure. PK analysis supports approval. Careful formulation control is required.
Pharmacodynamic Suppression of Testosterone
Clinical trials monitor testosterone suppression. Medical castration is defined as < 50\text{ ng/dL}. The reference product maintains steady levels. Generic products must replicate suppression. Matching in vivo degradation is critical. PD endpoints confirm equivalence.
Conclusion: Reverse Engineering Lupron Depot for Complex Generic Excellence
Reverse Engineering Lupron Depot microspheres is a multidisciplinary challenge involving polymer chemistry, manufacturing engineering, and advanced analytical characterization. Success requires understanding the Q1, Q2, and Q3 framework and controlling microstructural variables. Replicating acid-capped PLGA, double emulsion processing, and morphology is essential. Imaging and release testing confirm equivalence. A well-executed Reverse Engineering Lupron Depot strategy ensures consistent therapeutic performance. The final product must function as a pharmaceutical mirror of the innovator.
Next Steps for Developers: Why there is No Generic for Lupron Depot and how to change that
To discuss how ResolveMass Laboratories Inc. can support your complex generic development through expert de-formulation and microstructural characterization, please visit our contact page: https://resolvemass.ca/contact/
FAQs on Reverse Engineering Lupron Depot
The main challenge is the highly complex PLGA microsphere delivery system, where performance depends on internal microstructure rather than just the active drug. Small changes in polymer chemistry or manufacturing conditions can alter the triphasic release profile. Developers must also match strict pharmacokinetic limits and long-acting release behavior. Achieving this level of control makes regulatory approval extremely difficult.
Acid-capped PLGA contains terminal carboxyl groups that interact ionically with the basic leuprolide peptide. This interaction improves drug retention inside the microspheres and reduces the initial burst release. Ester-capped polymers lack these binding sites and are more hydrophobic. As a result, they typically produce higher burst release and different degradation behavior.
Leuprolide acetate is highly water-soluble and can diffuse out during emulsification. Cooling the primary emulsion increases viscosity of the polymer phase and helps trap drug droplets. This reduces drug leakage into the external phase and improves encapsulation efficiency. The step is essential for forming dense microspheres with controlled release.
AUC₇–t measures drug exposure from Day 7 to the end of the dosing interval. This period represents the sustained erosion-controlled release phase rather than the initial burst. Matching this parameter ensures the generic maintains long-term therapeutic levels. It helps confirm comparable hormonal suppression across the dosing period.
Porosity determines how quickly water penetrates and drug diffuses from the microsphere matrix. High porosity can increase the initial burst and shorten the lag phase. A dense core with controlled pore distribution promotes gradual hydration and erosion. This structure supports the characteristic triphasic release profile.
Accelerated testing at elevated temperatures speeds up polymer degradation and drug diffusion. When properly validated, these studies can correlate with real-time release behavior. For example, higher temperatures can compress a month-long study into a few days. However, the method must remain sensitive to formulation differences.
Type B gelatin increases viscosity of the internal aqueous phase during emulsification. This helps stabilize droplets and supports uniform microsphere formation. Although cooling drives encapsulation efficiency, gelatin contributes to microstructural consistency. Using the same gelatin grade is important for matching morphology.
Orthogonal imaging techniques are commonly used to evaluate internal structure. FIB-SEM provides high-resolution 3D visualization of pore networks and drug distribution. Synchrotron micro-CT allows non-destructive analysis of large particle populations. Together, these methods confirm structural similarity.
Reference:
- Zhou, Z., Zhou, Y., Yan, W., Feng, T., & Liang, Z. (2024). Comparison of the efficacy and safety profiles of generic and branded leuprorelin acetate microspheres in patients with prostate cancer. Oncology Letters, 28(1), 319. https://pmc.ncbi.nlm.nih.gov/articles/PMC10888034/
- Zhou, J., Schutzman, R., Shi, N.-Q., Ackermann, R., Olsen, K., Wang, Y., & Schwendeman, S. P. (2023). Influence of encapsulation variables on formation of leuprolide-loaded PLGA microspheres. Journal of Colloid and Interface Science, 636, 401–412. https://pubmed.ncbi.nlm.nih.gov/36640551/
- U.S. Food and Drug Administration. (2023). Product-specific guidance for industry: Leuprolide acetate injection (depot microspheres). U.S. Department of Health and Human Services. https://www.fda.gov/media/165544/download
- Zhou, J., Hirota, K., Ackermann, R., Walker, J., Wang, Y., Choi, S., Schwendeman, A., & Schwendeman, S. P. (2018). Reverse engineering the 1-month Lupron Depot®. AAPS Journal, 20(6), 105. https://pubmed.ncbi.nlm.nih.gov/30280294/
- Zhang, S., Wu, D., & Zhou, L. (2020). Characterization of controlled release microspheres using FIB-SEM and image-based release prediction. AAPS PharmSciTech, 21(5), 194. https://pubmed.ncbi.nlm.nih.gov/32666160/
- Clark, A. G., Wang, R., Qin, Y., Wang, Y., Zhu, A., Lomeo, J., Bao, Q., Burgess, D. J., Chen, J., Qin, B., Zou, Y., & Zhang, S. (2022). Assessing microstructural critical quality attributes in PLGA microspheres by FIB-SEM analytics. Journal of Controlled Release, 349, 580–591. https://pubmed.ncbi.nlm.nih.gov/35803326/
- Jiang, C., Kuang, L., Merkel, M. P., Yue, F., Cano-Vega, M. A., Narayanan, N., Kuang, S., & Deng, M. (2015). Biodegradable polymeric microsphere-based drug delivery for inductive browning of fat. Frontiers in Endocrinology, 6, 169. https://pmc.ncbi.nlm.nih.gov/articles/PMC4326979/



