
Introduction:
Reverse Engineering Risperidone PLGA Microspheres is a critical approach in modern pharmaceutical development, particularly for long-acting injectable formulations. By understanding how polymer characteristics influence drug release, scientists can effectively control the lag phase and replicate or enhance reference products.
For a deeper understanding of polymer selection, refer to role of PLGA polymer grade in long-acting release formulation and PLGA (polylactic-co-glycolic acid) for parenteral use.
Risperidone PLGA microspheres are widely used in sustained-release formulations to treat psychiatric disorders. However, achieving the desired release profile—especially minimizing or controlling the lag phase—requires deep expertise in polymer science and analytical characterization. This blog explores how reverse engineering techniques enable precise control over these systems.
Summary:
- Reverse engineering risperidone PLGA microspheres helps optimize drug release and bioavailability.
- The lag phase is primarily controlled by polymer properties such as molecular weight, lactide:glycolide ratio, and end-group chemistry.
- Advanced characterization techniques enable precise replication and improvement of reference formulations (see PLGA polymer characterization for generics).
- Controlling the lag phase ensures consistent therapeutic performance and patient compliance.
- ResolveMass Laboratories Inc. leverages analytical expertise to decode complex microsphere systems efficiently.
1: What Are Risperidone PLGA Microspheres?
Risperidone PLGA microspheres are biodegradable polymer-based drug delivery systems designed to release risperidone over an extended period, enabling long-acting therapeutic effects.
These systems fall under long-acting injectable drug delivery technologies and are widely used in chronic therapies.
These microspheres are engineered using a controlled matrix system that gradually degrades in the body, allowing for sustained and predictable drug release.
Composition of Risperidone PLGA Microspheres
- PLGA (Poly(lactic-co-glycolic acid)) polymer matrix – acts as the biodegradable carrier controlling drug release
- Encapsulated risperidone drug – the active pharmaceutical ingredient
- Stabilizers and excipients – enhance formulation stability and manufacturability
Key Benefits
- Sustained drug release over several weeks, reducing frequent dosing
- Improved patient compliance, especially in chronic psychiatric conditions
- Reduced dosing frequency, minimizing fluctuations in drug levels
Challenges
- Complex formulation design, requiring precise control over multiple variables
- Initial lag phase before drug release, which can delay therapeutic onset
- High sensitivity to polymer properties, impacting consistency and performance
These characteristics make risperidone PLGA microspheres a powerful yet technically demanding drug delivery system, emphasizing the importance of advanced formulation strategies and reverse engineering approaches.
For formulation insights, see PLGA microsphere formulation development.
2: What Is the Lag Phase in PLGA Microspheres?
The lag phase is the initial delay in drug release after administration, during which little to no drug is released before significant diffusion or polymer erosion begins.
Understanding this phase is essential in PLGA reverse engineering for ANDA submissions.
This phase is a critical aspect of long-acting injectable formulations, as it directly influences the onset of therapeutic action.
Why Does the Lag Phase Occur?
- Limited water penetration into the polymer matrix – slows the initiation of polymer hydrolysis
- Slow polymer degradation – PLGA requires time to break down before releasing the drug
- Drug trapped within dense polymer regions – delays diffusion until structural changes occur
Impact on Therapy
- Delayed therapeutic action, which may affect early treatment outcomes
- Risk of sub-therapeutic drug levels during the initial period
- Need for loading doses or oral supplementation in some formulations to bridge the delay
Understanding and controlling the lag phase is essential in Reverse Engineering Risperidone PLGA Microspheres, as it ensures consistent drug release profiles and optimal clinical performance.
3: Why Reverse Engineering Risperidone PLGA Microspheres Is Important
Reverse Engineering Risperidone PLGA Microspheres is essential because it uncovers the critical formulation and polymer parameters that govern drug release, enabling developers to match or improve the performance of reference products.
Case studies like dexamethasone implant PLGA characterization and exenatide PLGA microsphere characterization demonstrate how analytical insights improve formulation success.
In long-acting injectable formulations, even minor variations in polymer characteristics or processing conditions can significantly impact the lag phase and overall release kinetics. Reverse engineering provides a structured, data-driven pathway to decode these complexities.
Key Objectives
- Replicate reference listed drug (RLD) performance to achieve bioequivalence
- Optimize lag phase and release kinetics for consistent therapeutic outcomes
- Ensure regulatory compliance with stringent pharmaceutical guidelines
Benefits
- Faster product development by minimizing guesswork
- Reduced trial-and-error experimentation, saving time and cost
- Improved formulation robustness with better reproducibility and scalability
By applying advanced analytical techniques and formulation expertise, Reverse Engineering Risperidone PLGA Microspheres enables pharmaceutical developers to create reliable, high-quality long-acting drug delivery systems with predictable performance.
4: Key Polymer Characteristics That Control Lag Phase
The lag phase in PLGA microspheres is primarily governed by polymer properties such as molecular weight, lactide:glycolide ratio, end-group chemistry, and crystallinity, all of which influence water uptake and degradation rate.
To understand sourcing and quality, refer to PLGA supplier benefits and GMP PLGA requirements.
A precise understanding of these parameters is essential in Reverse Engineering Risperidone PLGA Microspheres, as small variations can significantly alter drug release behavior.
1. Molecular Weight
- Higher molecular weight → longer polymer chain length → slower degradation → extended lag phase
- Lower molecular weight → shorter chains → faster erosion → reduced lag phase
Insight: Molecular weight directly affects how quickly the polymer matrix breaks down, making it one of the most critical parameters to control.
2. Lactide:Glycolide Ratio
- Higher lactide content → increased hydrophobicity → slower water penetration → longer lag phase
- Higher glycolide content → increased hydrophilicity → faster degradation → shorter lag phase
Insight: Adjusting this ratio is a powerful way to fine-tune degradation kinetics and drug release timing.
3. End-Group Chemistry
- Ester-terminated PLGA → less reactive → slower hydrolysis → extended lag phase
- Acid-terminated PLGA → more reactive → faster degradation → reduced lag phase
Insight: End-group functionality influences how readily the polymer undergoes hydrolytic cleavage.
4. Polymer Crystallinity
- More crystalline structure → tightly packed chains → reduced water uptake → slower drug release
- More amorphous structure → loosely packed chains → enhanced diffusion and degradation → faster release
Insight: Crystallinity affects both diffusion pathways and polymer erosion behavior.
Why These Factors Matter
Controlling these polymer characteristics allows formulation scientists to:
- Precisely tune the lag phase duration
- Achieve consistent batch-to-batch performance
- Improve in vitro–in vivo correlation (IVIVC)
In Reverse Engineering Risperidone PLGA Microspheres, detailed polymer characterization ensures accurate replication of reference formulations and supports the development of optimized long-acting injectable therapies.
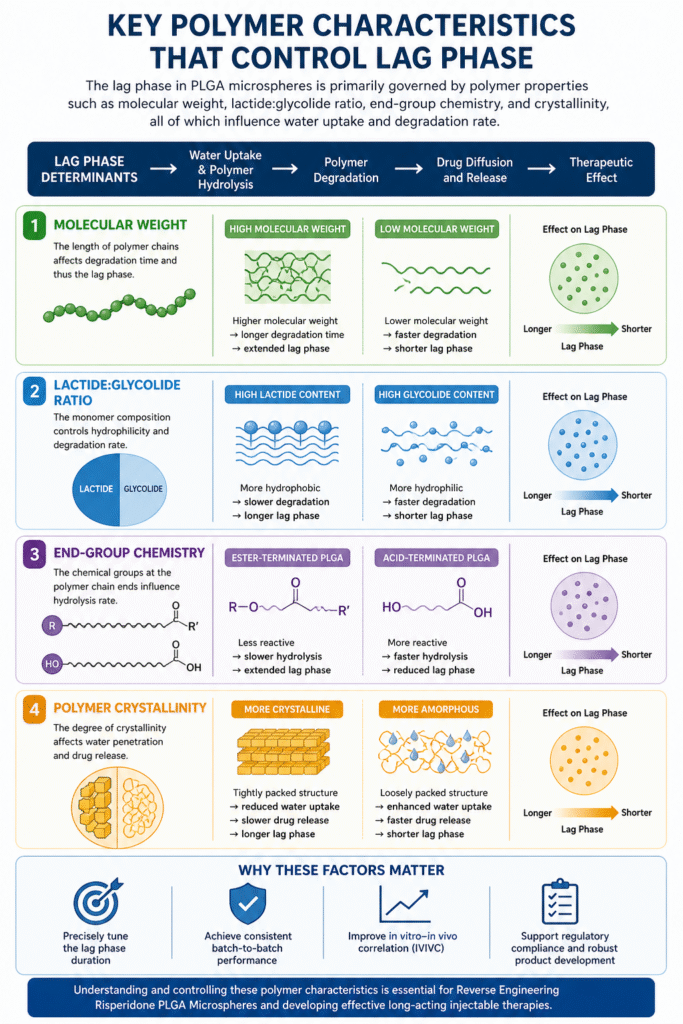
Table: Polymer Properties vs Lag Phase Behavior
| Polymer Property | Effect on Lag Phase | Mechanism |
|---|---|---|
| High Molecular Weight | Increases lag phase | Slower degradation |
| Low Molecular Weight | Decreases lag phase | Faster erosion |
| High Lactide Ratio | Longer lag phase | Hydrophobic nature |
| High Glycolide Ratio | Shorter lag phase | Hydrophilic nature |
| Acid End Groups | Reduced lag phase | Faster hydrolysis |
| Ester End Groups | Extended lag phase | Slower hydrolysis |
This table highlights how subtle variations in polymer characteristics can significantly influence the lag phase, making polymer selection and characterization critical in Reverse Engineering Risperidone PLGA Microspheres.
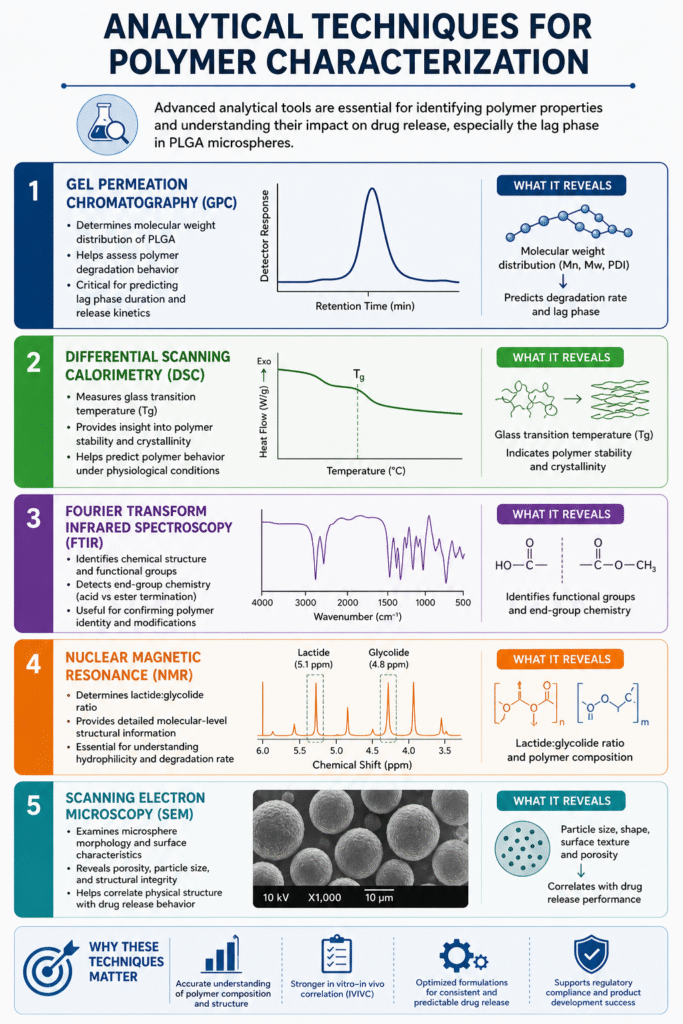
5: Analytical Techniques for Polymer Characterization
Advanced analytical techniques are essential for identifying polymer properties and understanding how they influence drug release, especially the lag phase in PLGA microspheres.
Explore detailed methods in PLGA characterization methods and PLGA reverse engineering CRO services.
For peptide-related systems, see PLGA peptide delivery.
In Reverse Engineering Risperidone PLGA Microspheres, these tools provide critical insights into polymer composition, structure, and performance, enabling precise formulation development.
Common Techniques Used
1. Gel Permeation Chromatography (GPC)
- Determines molecular weight distribution of PLGA
- Helps assess polymer degradation behavior
- Critical for predicting lag phase duration and release kinetics
2. Differential Scanning Calorimetry (DSC)
- Measures glass transition temperature (Tg)
- Provides insight into polymer stability and crystallinity
- Helps predict how the polymer will behave under physiological conditions
3. Fourier Transform Infrared Spectroscopy (FTIR)
- Identifies chemical structure and functional groups
- Detects end-group chemistry (acid vs ester termination)
- Useful for confirming polymer identity and modifications
4. Nuclear Magnetic Resonance (NMR)
- Determines lactide:glycolide ratio
- Provides detailed molecular-level structural information
- Essential for understanding hydrophilicity and degradation rate
5. Scanning Electron Microscopy (SEM)
- Examines microsphere morphology and surface characteristics
- Reveals porosity, particle size, and structural integrity
- Helps correlate physical structure with drug release behavior
Why These Techniques Matter
Using these analytical methods allows scientists to:
- Accurately decode polymer composition and structure
- Establish strong in vitro–in vivo correlations (IVIVC)
- Optimize formulations for consistent and predictable drug release
In Reverse Engineering Risperidone PLGA Microspheres, combining these techniques ensures a comprehensive understanding of the system, enabling better control over the lag phase and overall therapeutic performance.
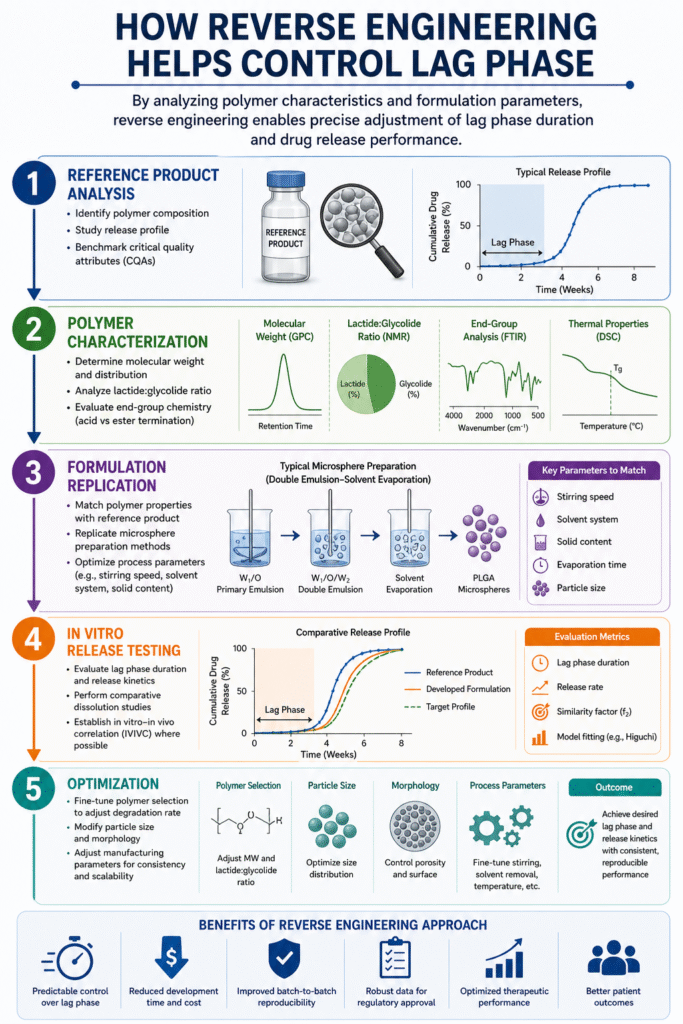
6: How Reverse Engineering Helps Control Lag Phase
Reverse Engineering Risperidone PLGA Microspheres enables precise control of the lag phase by systematically analyzing polymer characteristics and formulation parameters, then replicating and optimizing them to achieve the desired drug release profile.
A strong example is reverse engineering of PLGA polymer in Lupron Depot.
This structured approach reduces uncertainty and allows scientists to fine-tune formulations with high accuracy.
Step-by-Step Approach
1. Reference Product Analysis
- Identify polymer composition and excipient profile
- Study the drug release profile, including lag phase duration
- Benchmark critical quality attributes (CQAs)
2. Polymer Characterization
- Determine molecular weight and distribution
- Analyze lactide:glycolide ratio
- Evaluate end-group chemistry (acid vs ester termination)
3. Formulation Replication
- Match polymer properties with the reference product
- Replicate microsphere preparation methods (e.g., solvent evaporation)
- Optimize process parameters such as stirring speed and solvent system
4. In Vitro Release Testing
- Evaluate lag phase duration and overall release kinetics
- Perform comparative dissolution studies
- Establish in vitro–in vivo correlation (IVIVC) where possible
5. Optimization
- Fine-tune polymer selection to adjust degradation rate
- Modify particle size and morphology
- Adjust manufacturing parameters for consistency and scalability
Why This Approach Works
- Enables predictable control over lag phase
- Reduces development time and cost
- Improves batch-to-batch reproducibility
- Supports regulatory approval through robust data
In Reverse Engineering Risperidone PLGA Microspheres, this systematic methodology ensures that the lag phase is not just understood—but precisely controlled—leading to reliable, high-performance long-acting injectable formulations.
7: Strategies to Minimize or Control Lag Phase
The lag phase in PLGA microspheres can be effectively minimized or controlled by optimizing polymer properties and refining formulation techniques to accelerate water uptake and polymer degradation.
In Reverse Engineering Risperidone PLGA Microspheres, applying the right combination of strategies ensures a more predictable and therapeutically effective drug release profile.
For oncology applications, see PLGA for oncology implant and formulating highly potent APIs using PLGA microspheres.
Effective Strategies
- Use lower molecular weight PLGA
→ Promotes faster polymer degradation and reduces lag phase duration - Increase glycolide content
→ Enhances hydrophilicity, allowing quicker water penetration and erosion - Incorporate pore-forming agents
→ Creates channels within microspheres, enabling earlier drug diffusion - Optimize particle size distribution
→ Smaller particles increase surface area, leading to faster release initiation - Modify solvent evaporation techniques
→ Controls internal structure and porosity, influencing release kinetics
Advanced Approaches
- Polymer blending
→ Combines different PLGA grades to fine-tune degradation behavior - Surface modification
→ Alters microsphere surface to improve water interaction and reduce lag time - Multi-layer microsphere design
→ Enables staged or controlled release profiles with reduced initial delay
Why These Strategies Matter
- Enable precise control over lag phase duration
- Improve drug release consistency and predictability
- Support bioequivalence with reference formulations
- Enhance overall therapeutic performance
By implementing these strategies, scientists can significantly reduce the lag phase and optimize long-acting injectable formulations, making Reverse Engineering Risperidone PLGA Microspheres more efficient and clinically effective.
8: Role of ResolveMass Laboratories Inc.
ResolveMass Laboratories Inc. plays a critical role in Reverse Engineering Risperidone PLGA Microspheres by delivering advanced analytical and formulation insights that enable precise control over lag phase and drug release performance.
Their expertise is evident in projects like PLGA characterization Lupron Depot and addressing leuprolide depot formulation challenges.
With deep expertise in complex drug delivery systems, the organization supports pharmaceutical companies in decoding intricate microsphere formulations and accelerating development timelines.
Expertise Includes:
- Comprehensive polymer characterization
→ Detailed analysis of molecular weight, composition, and end-group chemistry - Microsphere formulation analysis
→ Evaluation of particle size, morphology, and internal structure - Bioequivalence support
→ Ensuring formulations match reference listed drug (RLD) performance - Regulatory-ready documentation
→ Preparation of high-quality data packages aligned with global guidelines
Why Choose ResolveMass?
- Proven experience in complex generics
→ Strong track record in handling long-acting injectable formulations - State-of-the-art analytical tools
→ Advanced instrumentation for accurate and reproducible results - Strong focus on data integrity and compliance
→ Ensuring reliability and regulatory acceptance
Impact on Lag Phase Control
Through a combination of analytical precision and formulation expertise, ResolveMass Laboratories Inc. enables:
- Accurate decoding of polymer characteristics
- Effective optimization of lag phase duration
- Development of robust and scalable formulations
Their integrated approach ensures that Reverse Engineering Risperidone PLGA Microspheres is not only efficient but also aligned with regulatory and therapeutic expectations.
9: Challenges in Reverse Engineering PLGA Microspheres
Reverse engineering PLGA microspheres is challenging due to inherent polymer variability, complex drug release mechanisms, and limitations in analytical characterization.
These challenges are often addressed through structured workflows in PLGA polymer characterization for generics.
In Reverse Engineering Risperidone PLGA Microspheres, overcoming these challenges is essential to achieve consistent performance and regulatory acceptance.
Key Challenges
- Batch-to-batch polymer variability
→ Differences in molecular weight, end-group chemistry, and composition can significantly impact degradation rate and lag phase - Incomplete knowledge of proprietary formulations
→ Limited access to exact formulation details makes accurate replication difficult - Difficulty in replicating manufacturing processes
→ Parameters such as solvent systems, emulsification conditions, and drying techniques are often not fully disclosed - Need for extensive in vitro–in vivo correlation (IVIVC)
→ Establishing a reliable relationship between lab testing and clinical performance is complex but critical
Why These Challenges Matter
- Can lead to inconsistent drug release profiles
- Increase development time and cost
- Complicate regulatory approval processes
Addressing These Challenges
Advanced analytical tools and a systematic approach help mitigate these issues by:
- Improving polymer characterization accuracy
- Enabling data-driven formulation development
- Strengthening predictability of drug release behavior
Successfully navigating these challenges is a key component of Reverse Engineering Risperidone PLGA Microspheres, ensuring the development of reliable, high-quality long-acting injectable products.
10: Future Trends in Microsphere Reverse Engineering
Emerging technologies are improving the precision and efficiency of reverse engineering processes.
Innovations Include:
- AI-driven formulation modeling
- Advanced imaging techniques
- Predictive polymer degradation models
- Continuous manufacturing approaches
These advancements are expected to significantly improve control over lag phase and overall drug delivery performance.
Conclusion:
Reverse Engineering Risperidone PLGA Microspheres plays a vital role in developing effective long-acting injectable formulations. By understanding and controlling polymer characteristics, scientists can precisely manage the lag phase and ensure consistent therapeutic outcomes.
Through advanced analytical techniques and formulation expertise, organizations like ResolveMass Laboratories Inc. are leading the way in decoding complex microsphere systems. Their approach not only accelerates product development but also ensures regulatory compliance and patient safety.
Frequently Asked Questions:
Reverse Engineering Risperidone PLGA Microspheres is the process of analyzing a reference formulation to identify its polymer composition, structure, and drug release behavior. This helps in replicating or improving long-acting injectable products. It involves advanced analytical techniques to decode critical parameters like molecular weight and polymer ratio. The goal is to achieve similar or optimized therapeutic performance. It is widely used in generic drug development and formulation optimization.
The lag phase determines how quickly the drug begins to release after administration. A long lag phase can delay therapeutic effects, while a short lag phase may lead to rapid drug release. Controlling this phase ensures consistent drug levels in the body. It is especially important for long-acting injectables like risperidone. Proper lag phase management improves treatment outcomes and patient compliance.
The lag phase can be controlled by adjusting polymer properties such as molecular weight, lactide:glycolide ratio, and end-group chemistry. Formulation techniques like particle size optimization and pore-forming agents also help. Advanced strategies include polymer blending and surface modification. These approaches improve water penetration and polymer degradation rates. Together, they allow precise tuning of drug release profiles.
Common techniques include Gel Permeation Chromatography (GPC), Differential Scanning Calorimetry (DSC), FTIR, NMR, and SEM. These methods help determine molecular weight, polymer composition, and structural properties. They also provide insights into degradation behavior and release kinetics. Using multiple techniques ensures accurate characterization. This is essential for replicating and optimizing formulations.
Polymer characterization helps understand how properties like molecular weight and composition affect drug release. It ensures consistent performance and reproducibility. Accurate characterization is essential for controlling the lag phase. It also supports regulatory compliance and bioequivalence. Without it, achieving predictable drug delivery is difficult.
Reverse engineering reduces development time and minimizes trial-and-error experimentation. It helps achieve bioequivalence with reference products. Companies can optimize formulations for better performance and stability. It also supports regulatory submissions with strong analytical data. Overall, it improves efficiency and reduces costs.
Reference
- Wan B, Bao Q, Zou Y, Wang Y, Burgess DJ. Effect of polymer source variation on the properties and performance of risperidone microspheres. International Journal of Pharmaceutics. 2021 Dec 15;610:121265.https://www.sciencedirect.com/science/article/pii/S0378517321010711
- Chaurasia S, Mounika K, Bakshi V, Prasad V. 3-month parenteral PLGA microsphere formulations of risperidone: Fabrication, characterization and neuropharmacological assessments. Materials Science and Engineering: C. 2017 Jun 1;75:1496-505.https://www.sciencedirect.com/science/article/pii/S0928493116313613
- Son YJ, Yun TH, Lee JG, Bang KH, Kim KS. Development and Characterization of Long-Acting Injectable Risperidone Microspheres Using Biodegradable Polymers: Formulation Optimization and Release Kinetics. Processes. 2024 Dec 13;12(12):2858.https://www.mdpi.com/2227-9717/12/12/2858
- Hua Y, Wang Z, Wang D, Lin X, Liu B, Zhang H, Gao J, Zheng A. Key factor study for generic long-acting PLGA microspheres based on a reverse engineering of Vivitrol®. Molecules. 2021 Feb 25;26(5):1247.https://www.mdpi.com/1420-3049/26/5/1247
- Su ZX, Shi YN, Teng LS, Li X, Wang LX, Meng QF, Teng LR, Li YX. Biodegradable poly (D, L-lactide-co-glycolide)(PLGA) microspheres for sustained release of risperidone: Zero-order release formulation. Pharmaceutical Development and Technology. 2011 Aug 1;16(4):377-84.https://www.tandfonline.com/doi/abs/10.3109/10837451003739297


