
Introduction:
Sameness Evaluation in ANDA is the scientific and regulatory process of proving that a generic drug’s active ingredient is equivalent to its reference listed drug (RLD).
For pharmaceutical companies pursuing generic drug approval, this is not just a compliance requirement—it is the foundation of ANDA success. Any gaps in demonstrating sameness can lead to regulatory delays, complete response letters (CRLs), or even rejection.
For real-world case studies, refer to:
At ResolveMass Laboratories Inc., we specialize in delivering high-resolution analytical characterization that ensures your API meets stringent regulatory expectations with confidence and precision.
Summary:
- Sameness Evaluation in ANDA ensures a generic drug matches the reference listed drug (RLD) in identity, strength, quality, purity, and performance.
- It is a regulatory requirement by the FDA for Abbreviated New Drug Application (ANDA) approval.
- Critical components include:
- Peptide/chemical structure confirmation
- Impurity profiling
- Physicochemical characterization
- Analytical method validation
- Advanced tools such as LC-MS, HRMS, and NMR are essential for demonstrating sameness.
- Early and robust sameness evaluation significantly reduces regulatory risk and accelerates approval timelines.
1: What Does “Sameness” Mean for Active Ingredients in ANDA?
Sameness Evaluation in ANDA for APIs means that the generic drug substance must be scientifically demonstrated to be the same as the reference listed drug (RLD) in identity, molecular structure, and critical quality attributes.
In regulatory terms, this is not a superficial comparison—it is a data-driven confirmation using advanced analytical characterization.
Regulatory Definition of API Sameness
Regulators define API sameness as equivalence across all fundamental molecular and quality attributes that influence safety and efficacy.
For regulatory expectations, refer to:
FDA Peptide Sameness Study Requirements
To establish sameness, the API must demonstrate:
- Same chemical structure or peptide sequence
→ The exact molecular framework, including bonding and sequence (for peptides), must match the RLD without deviation. - Same stereochemistry and isomeric form
→ Chirality, geometric isomerism, and spatial configuration must be identical, as even minor differences can alter biological activity. - Comparable impurity profile
→ The types and levels of impurities should be similar and within acceptable regulatory thresholds, with no unexpected or unsafe impurities. - Equivalent physicochemical properties
→ Properties such as solubility, pKa, and partition coefficient should align closely to ensure consistent in vivo performance.
Important Clarification
Sameness Evaluation in ANDA is strictly limited to the active pharmaceutical ingredient and does not extend to other aspects of the drug product.
Sameness does NOT require:
- Same manufacturing process
→ Generic manufacturers can use different synthetic routes, provided the final API meets all sameness criteria. - Same excipients
→ Non-active ingredients may differ, as long as they do not affect safety or efficacy. - Same formulation
→ The dosage form design can vary, provided bioequivalence is demonstrated separately.
It strictly refers to the active pharmaceutical ingredient (drug substance).
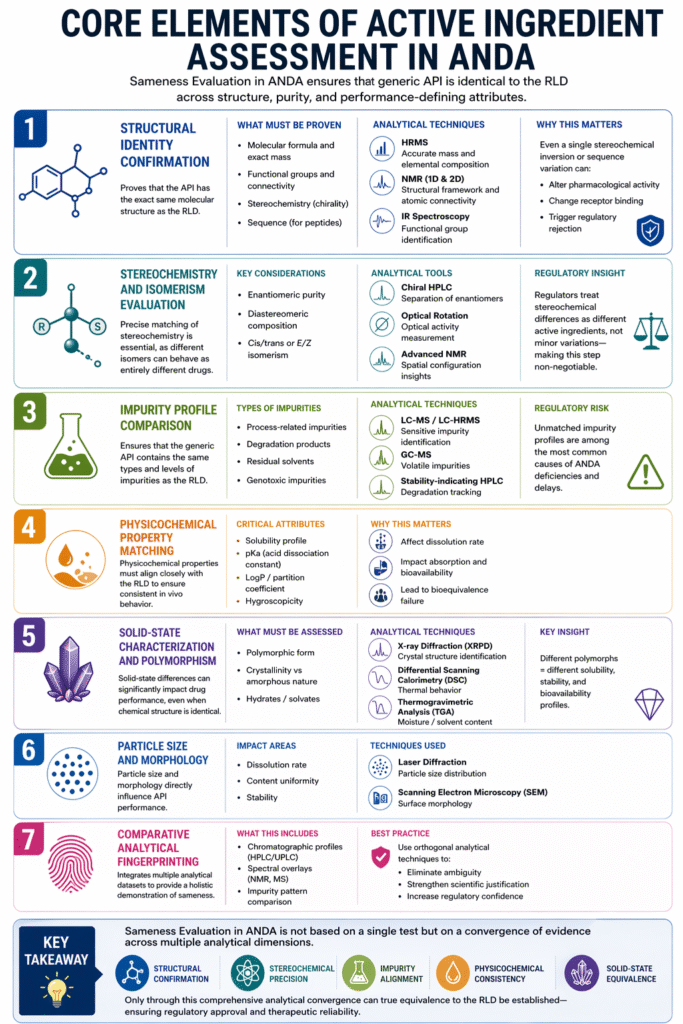
2: Core Elements of Active Ingredient Assessment in ANDA
The core elements of active ingredient assessment in Sameness Evaluation in ANDA ensure that the generic API is identical to the RLD across structure, purity, and performance-defining attributes.
Each element provides a different layer of scientific evidence, and together they form a comprehensive, regulator-ready demonstration of sameness.
For peptide-specific regulatory gaps, see:
Peptide Sameness Study Deficiencies
1. Structural Identity Confirmation
Structural identity confirmation proves that the API has the exact same molecular structure as the RLD.
What Must Be Proven?
- Molecular formula and exact mass → Confirms elemental composition
- Functional groups and connectivity → Ensures correct bonding framework
- Stereochemistry (chirality) → Verifies 3D arrangement
- Sequence (for peptides) → Confirms amino acid order
Analytical Techniques Used
- High-Resolution Mass Spectrometry (HRMS) → Accurate mass and elemental composition
- NMR (1D & 2D) → Structural framework and atomic connectivity
- Infrared Spectroscopy (IR) → Functional group identification
Why This Matters
Even a single stereochemical inversion or sequence variation can:
- Alter pharmacological activity
- Change receptor binding
- Trigger regulatory rejection
2. Stereochemistry and Isomerism Evaluation
Sameness Evaluation in ANDA requires precise matching of stereochemistry, as different isomers can behave as entirely different drugs.
Key Considerations
- Enantiomeric purity → Ratio of optical isomers
- Diastereomeric composition → Multiple chiral centers
- Cis/trans or E/Z isomerism → Geometric configuration
Analytical Tools
- Chiral HPLC → Separation of enantiomers
- Optical rotation → Optical activity measurement
- Advanced NMR techniques → Spatial configuration insights
Regulatory Insight
Regulators treat stereochemical differences as different active ingredients, not minor variations—making this step non-negotiable.
3. Impurity Profile Comparison
Impurity profiling ensures that the generic API contains the same types and levels of impurities as the RLD.
Types of API Impurities
- Process-related impurities
- Degradation products
- Residual solvents
- Genotoxic impurities
Key Requirements
- Identification of impurities above reporting thresholds
- Qualification of unknown impurities
- Direct comparison with RLD impurity spectrum
Analytical Techniques
- LC-MS / LC-HRMS → Sensitive impurity identification
- GC-MS → Volatile impurities
- Stability-indicating HPLC → Degradation tracking
Regulatory Risk
Unmatched impurity profiles are among the most common causes of ANDA deficiencies and delays.
4. Physicochemical Property Matching
Physicochemical properties must align closely with the RLD to ensure consistent in vivo behavior.
Critical Attributes
- Solubility profile
- pKa (acid dissociation constant)
- LogP / partition coefficient
- Hygroscopicity
Why This Matters
Variations in these properties can:
- Affect dissolution rate
- Impact absorption and bioavailability
- Lead to bioequivalence failure
5. Solid-State Characterization and Polymorphism
Solid-state differences can significantly impact drug performance, even when chemical structure is identical.
What Must Be Assessed?
- Polymorphic form
- Crystallinity vs amorphous nature
- Hydrates/solvates
Analytical Techniques
- X-ray diffraction (XRPD) → Crystal structure identification
- Differential scanning calorimetry (DSC) → Thermal behavior
- Thermogravimetric analysis (TGA) → Moisture/solvent content
Key Insight
Different polymorphs = different solubility, stability, and bioavailability profiles.
6. Particle Size and Morphology
Particle size and morphology directly influence API performance and must be comparable to the RLD.
Impact Areas
- Dissolution rate
- Content uniformity
- Stability
Techniques Used
- Laser diffraction → Particle size distribution
- Scanning Electron Microscopy (SEM) → Surface morphology
7. Comparative Analytical Fingerprinting
Comparative fingerprinting integrates multiple analytical datasets to provide a holistic demonstration of sameness.
What This Includes
- Chromatographic profiles (HPLC/UPLC)
- Spectral overlays (NMR, MS)
- Impurity pattern comparison
Best Practice
Use orthogonal analytical techniques (multiple independent methods) to:
- Eliminate ambiguity
- Strengthen scientific justification
- Increase regulatory confidence
Key Takeaway
Sameness Evaluation in ANDA is not based on a single test but on a convergence of evidence across multiple analytical dimensions.
A robust active ingredient assessment integrates structure, impurities, physicochemical properties, and solid-state characteristics to prove true equivalence with the RLD.
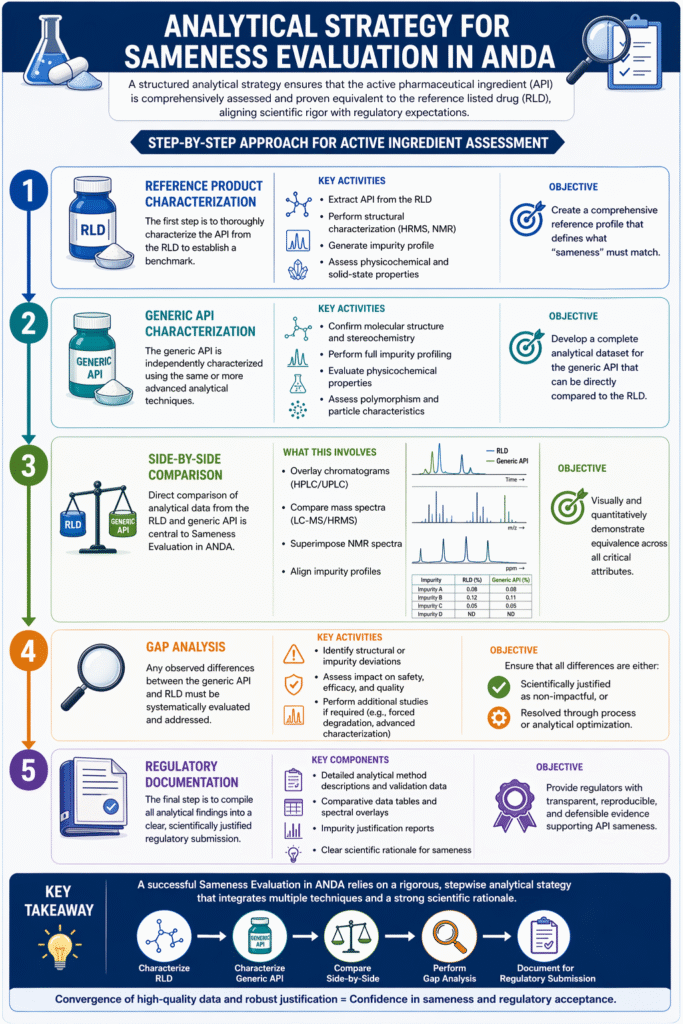
3: Analytical Strategy for Sameness Evaluation in ANDA
A structured analytical strategy is essential for Sameness Evaluation in ANDA to ensure that the active pharmaceutical ingredient (API) is comprehensively assessed and proven equivalent to the reference listed drug (RLD).
For end-to-end execution, explore:
Peptide Sameness Study Services in United States
Peptide Sameness Study Services in Canada
This strategy follows a stepwise, evidence-based approach, aligning scientific rigor with regulatory expectations.
Step-by-Step Approach for Active Ingredient Assessment
1. Reference Product Characterization
The first step in Sameness Evaluation in ANDA is to thoroughly characterize the API from the RLD to establish a benchmark.
Key Activities
- Extract API from the reference listed drug (RLD)
- Perform structural characterization (HRMS, NMR)
- Generate impurity profile
- Assess physicochemical and solid-state properties
Objective
Create a comprehensive reference profile that defines what “sameness” must match.
2. Generic API Characterization
The generic API is then independently characterized using the same or more advanced analytical techniques.
Key Activities
- Confirm molecular structure and stereochemistry
- Perform full impurity profiling
- Evaluate physicochemical properties
- Assess polymorphism and particle characteristics
Objective
Develop a complete analytical dataset for the generic API that can be directly compared to the RLD.
3. Side-by-Side Comparison
Direct comparison of analytical data from the RLD and generic API is central to Sameness Evaluation in ANDA.
What This Involves
- Overlay chromatograms (HPLC/UPLC)
- Compare mass spectra (LC-MS/HRMS)
- Superimpose NMR spectra
- Align impurity profiles
Objective
Visually and quantitatively demonstrate equivalence across all critical attributes.
4. Gap Analysis
Any observed differences between the generic API and RLD must be systematically evaluated and addressed.
Key Activities
- Identify structural or impurity deviations
- Assess impact on safety, efficacy, and quality
- Perform additional studies if required (e.g., forced degradation, advanced characterization)
Objective
Ensure that all differences are either:
- Scientifically justified as non-impactful, or
- Resolved through process or analytical optimization
5. Regulatory Documentation
The final step is to compile all analytical findings into a clear, scientifically justified regulatory submission.
Key Components
- Detailed analytical method descriptions and validation data
- Comparative data tables and spectral overlays
- Impurity justification reports
- Clear scientific rationale for sameness
Objective
Provide regulators with transparent, reproducible, and defensible evidence supporting API sameness.
4: Challenges in Active Ingredient Sameness Evaluation
Active ingredient assessment in Sameness Evaluation in ANDA presents multiple scientific and regulatory challenges that can directly impact approval timelines.
For real-world regulatory issues, refer to:
Peptide Sameness Study Deficiencies
These challenges arise from the need to prove molecular-level equivalence with high precision, often under constraints such as limited reference material and complex API structures.
Common Issues in Sameness Evaluation in ANDA
1. Trace-Level Impurity Detection
Detecting and identifying impurities at very low levels is one of the most critical challenges.
- Impurities may exist at ppm or sub-ppm levels
- Some impurities may be unknown or structurally similar to the API
- Regulatory thresholds (ICH) require accurate identification and quantification
Impact: Undetected or unqualified impurities can lead to regulatory queries or rejection.
2. Isomer Differentiation
Distinguishing between structurally similar isomers is complex but essential for API sameness.
- Enantiomers may have different biological activity
- Diastereomers can affect stability and efficacy
- Geometric isomers (E/Z, cis/trans) require precise identification
Impact: Failure to correctly identify and control isomers can result in the API being considered non-equivalent.
3. Limited Access to RLD API
Obtaining sufficient quantities of the reference listed drug (RLD) API for analysis is often a major limitation.
- API must be extracted from finished dosage forms
- Extraction can introduce artifacts or degradation
- Limited sample quantity restricts repeat testing and method development
Impact: Incomplete characterization of the RLD can weaken the entire sameness strategy.
4. Complex Peptide or Synthetic APIs
Highly complex molecules significantly increase the difficulty of sameness evaluation.
- Peptides require sequence confirmation and PTM analysis
- Synthetic APIs may have multiple chiral centers and impurities
- Structural complexity demands advanced, multi-technique approaches
Impact: Higher risk of undetected variations and increased regulatory scrutiny.
Mitigation Strategies for Successful Sameness Evaluation
1. Use High-Resolution Instrumentation
Advanced analytical tools improve sensitivity, accuracy, and confidence in results.
- HRMS for exact mass and impurity identification
- 2D NMR for structural confirmation
- High-resolution chromatography for better separation
2. Apply Forced Degradation Studies
Forced degradation helps uncover potential impurities and degradation pathways.
- Stress testing under heat, light, pH, and oxidation
- Identification of degradation products
- Supports development of stability-indicating methods
3. Develop Sensitive and Selective Methods
Robust analytical methods are essential to detect subtle differences between APIs.
- Optimize method sensitivity for trace detection
- Ensure specificity to separate closely related compounds
- Validate methods for accuracy, precision, and reproducibility
Key Takeaway
Challenges in active ingredient sameness evaluation are inevitable, but they are manageable with the right analytical strategy.
Organizations that invest in high-resolution techniques and early risk assessment are far more likely to achieve successful and timely ANDA approvals.
5: Special Considerations for Complex APIs
For peptides, complex molecules, or highly functional APIs, Sameness Evaluation in ANDA requires deeper and more sophisticated analytical assessment beyond conventional approaches.
These APIs exhibit structural complexity, conformational variability, and modification sensitivity, making sameness evaluation more demanding and risk-prone.
Relevant Case Studies
Why Complex APIs Require Additional Scrutiny
Complex APIs cannot be fully characterized using a single technique and often require multi-dimensional analysis.
Key Challenges
- Presence of multiple structural variants
- Sensitivity to minor sequence or chemical modifications
- Potential for conformational changes affecting activity
- Increased likelihood of process-related and degradation impurities
Impact:
Even subtle differences can significantly alter biological function, stability, and immunogenicity, leading to regulatory concerns.
Additional Requirements
- Sequence confirmation
- Post-translational modification (PTM) analysis
- Conformational assessment
Advanced Techniques for Complex API Characterization
A combination of advanced analytical tools is required to fully assess complex APIs.
1. Peptide Mapping
Peptide mapping provides detailed insight into sequence and modification patterns.
- Enzymatic digestion followed by separation
- Identification of peptide fragments
- Detection of sequence variations and PTMs
2. Enzymatic Digestion + LC-MS
This approach enables high-resolution analysis of peptide fragments and modifications.
- Precise mass measurement of fragments
- Localization of modifications within sequence
- High sensitivity for trace-level variants
3. 2D NMR
Two-dimensional NMR offers detailed structural and conformational insights.
- Atomic-level connectivity and spatial relationships
- Detection of subtle structural differences
- Complementary to MS-based techniques
6: Regulatory Expectations: What Reviewers Look For
In Sameness Evaluation in ANDA, regulators expect clear, reproducible, and scientifically justified evidence demonstrating that the active pharmaceutical ingredient (API) is truly equivalent to the reference listed drug (RLD).
Reviewers are not only assessing data—they are evaluating the quality, consistency, and scientific credibility of the entire analytical package.
For detailed regulatory alignment, see:
FDA Peptide Sameness Study Requirements
Key Reviewer Focus Areas in Sameness Evaluation in ANDA
1. Consistency Across Analytical Techniques
Regulators expect alignment of results across multiple orthogonal analytical methods.
- Structural data from HRMS, NMR, and IR should be consistent
- Chromatographic profiles should support spectral findings
- No contradictions between different analytical datasets
Why it matters:
Consistency across techniques increases confidence in the accuracy and reliability of the results.
2. Justification for Any Observed Differences
Any differences between the generic API and RLD must be clearly explained and scientifically justified.
- Minor impurity differences must be within acceptable limits
- Structural or physicochemical variations must be shown to have no impact on safety or efficacy
- Additional studies may be required to support justification
Why it matters:
Unexplained differences are one of the most common reasons for regulatory queries and delays.
3. Compliance with ICH Impurity Guidelines
Impurity profiling must fully comply with international regulatory standards.
Key Expectations
- Identification of impurities above reporting thresholds
- Qualification of impurities above qualification thresholds
- Control of genotoxic impurities at very low levels
Why it matters:
Impurities directly impact patient safety, making this a high-priority review area.
4. Robust Analytical Method Validation
All analytical methods used in sameness evaluation must be fully validated and fit for purpose.
Validation Parameters
- Accuracy → Correctness of results
- Precision → Reproducibility
- Specificity → Ability to distinguish analytes
- Linearity → Response proportionality
- Robustness → Stability under varying conditions
Why it matters:
Without validated methods, even high-quality data may be considered unreliable or unacceptable.
7: Best Practices for API Sameness Evaluation in ANDA
Following best practices significantly improves approval success rates.
Recommended Approach
- Start API characterization early in development
- Use multiple orthogonal analytical methods
- Maintain high-quality reference standards
- Document all findings with regulatory clarity
- Collaborate with experienced analytical laboratories
Learn more about our Peptide Sameness Study for ANDA solutions.
Conclusion:
Sameness Evaluation in ANDA is fundamentally an exercise in deep scientific validation of the active ingredient.
A successful ANDA hinges on proving that the API is not just similar—but truly identical in structure, purity, and critical attributes to the reference listed drug.
By focusing on comprehensive active ingredient assessment, leveraging advanced analytical techniques, and aligning with regulatory expectations, pharmaceutical companies can significantly reduce risk and accelerate approval timelines.
By leveraging advanced analytical techniques and regulatory expertise, companies can significantly reduce risk and accelerate approval timelines.
For specialized support, explore:
Frequently Asked Questions:
Key parameters include:
-Molecular structure and identity
-Stereochemistry and isomerism
-Impurity profile
-Physicochemical properties
-Solid-state characteristics
Commonly used techniques include:
-LC-MS / HRMS for structure and impurities
-NMR (1D & 2D) for structural confirmation
-HPLC/UPLC for purity and profiling
-XRPD and DSC for solid-state analysis
Major challenges include:
-Detecting trace-level impurities
-Differentiating isomers
-Limited access to RLD API
-Characterizing complex APIs like peptides
Impurities are identified, quantified, and compared with the RLD using advanced analytical methods, ensuring compliance with ICH guidelines and safety thresholds.
Differences must be scientifically justified. If they impact safety, efficacy, or quality, they can lead to regulatory queries or rejection.
Reference
- Cohen EM, Lin LY. Active Pharmaceutical Ingredients. InGeneric Drug Product Development 2014 Oct 31 (pp. 39-52). CRC Press.https://api.taylorfrancis.com/content/chapters/edit/download?identifierName=doi&identifierValue=10.1201/9781420030419-5&type=chapterpdf
- Stevens RE, Gray V, Dorantes A, Gold L, Pham L. Scientific and regulatory standards for assessing product performance using the similarity factor, f2. The AAPS journal. 2015 Mar;17(2):301-6.https://link.springer.com/article/10.1208/s12248-015-9723-y
- Faria EC, Bercu JP, Dolan DG, Morinello EJ, Pecquet AM, Seaman C, Sehner C, Weideman PA. Using default methodologies to derive an acceptable daily exposure (ADE). Regulatory Toxicology and Pharmacology. 2016 Aug 15;79:S28-38.https://www.sciencedirect.com/science/article/pii/S0273230016301398
- Pawar J, Hegde N, Sharma S. Regulatory frameworks and filing discrepancies in generic drug approvals: A cross-regional study with analysis of FDA ANDA deficiencies. InAnnales Pharmaceutiques Françaises 2025 Mar 10. Elsevier Masson.https://www.sciencedirect.com/science/article/pii/S0003450925000434


