
Introduction:
The Sameness Evaluation of Synthetic Peptides for ANDA is a critical regulatory requirement that ensures generic synthetic peptide drugs are structurally and functionally equivalent to the Reference Listed Drug (RLD). FDA guidance emphasizes that peptides, due to their complexity, cannot be evaluated using a single analytical method. Instead, a comprehensive orthogonal analytical strategy is required.
For pharmaceutical developers like ResolveMass Laboratories Inc., this evaluation is not just a regulatory checkpoint—it is a scientific confirmation of identity, purity, and therapeutic equivalence. Modern peptide therapeutics demand high-resolution analytical techniques to detect even minor variations that may impact safety or efficacy.
Explore FDA peptide sameness study requirements for deeper regulatory insights.
See peptide sameness study for ANDA workflow approach.
Summary:
- Sameness Evaluation of Synthetic Peptides for ANDA ensures the generic peptide matches the Reference Listed Drug (RLD) in structure, purity, and biological relevance.
- FDA expects a multi-layered analytical approach combining LC-MS, HRMS, peptide mapping, and NMR.
- Orthogonal methods are essential to confirm identity, sequence integrity, and impurity profile.
- Regulatory approval depends on demonstrating structural and functional equivalence, not just compositional similarity.
- Advanced characterization reduces risk of rejection and accelerates ANDA approval.
Learn more about Sameness Evaluation in ANDA and regulatory expectations.
1: What is Sameness Evaluation of Synthetic Peptides for ANDA?
Sameness evaluation of synthetic peptides for ANDA is the scientific and regulatory process used to demonstrate that a generic synthetic peptide drug is structurally, chemically, and functionally equivalent to the Reference Listed Drug (RLD).
Learn more about active ingredient sameness in ANDA
Read: peptide sameness study for ANDA
In simple terms, it confirms that the generic peptide is essentially identical to the innovator product in all critical quality attributes.
It ensures:
- The same amino acid sequence as the RLD
- The same stereochemistry, folding, and higher-order structure
- The same impurity and degradation profile
- The same biological activity and functional performance
Why FDA requires this:
The FDA mandates sameness evaluation to ensure that there are no clinically meaningful differences between the generic peptide and the reference product. This is crucial because even minor structural variations in peptides can significantly impact safety, efficacy, and immunogenicity.
2: Why is Sameness Evaluation Critical in ANDA?
Sameness evaluation in ANDA is critical because synthetic peptides are highly complex molecules, and even small variations in their structure or composition can significantly impact their safety, efficacy, and therapeutic performance.
Related reading:
It is critical because:
- Even minor sequence variations can change biological activity and reduce therapeutic effectiveness
- Impurities or related substances may trigger unwanted immunogenic or adverse immune responses
- Structural modifications can alter pharmacokinetics, affecting absorption, distribution, and duration of action
- Regulatory approval depends on proven equivalence to the Reference Listed Drug (RLD) in both structure and function
Regulatory importance:
FDA requires robust sameness evaluation to ensure that the generic peptide product performs identically to the innovator drug in clinical settings.
Key risk:
Without comprehensive analytical validation, ANDA submissions may be rejected due to incomplete characterization or insufficient proof of equivalence, delaying product approval and market entry.
3: FDA Guidance on Synthetic Peptide Sameness
FDA guidance for synthetic peptide sameness in ANDA submissions follows a science-driven, evidence-based approach that requires comprehensive structural and analytical characterization to demonstrate equivalence with the Reference Listed Drug (RLD).
Supporting resources:
- FDA peptide sameness study requirements
- Orthogonal analytical techniques for ANDA sameness evaluation
Key FDA expectations include:
- Full primary sequence confirmation to ensure the correct amino acid order and integrity of the peptide backbone
- High-resolution mass analysis (HRMS) to accurately verify molecular weight and detect subtle structural differences
- Detailed impurity profiling to identify and quantify related substances, degradation products, and process-related impurities
- Comparative structural characterization with the RLD to confirm equivalence in identity, structure, and higher-order attributes
- Use of orthogonal analytical methods such as LC-MS, peptide mapping, and NMR to provide cross-validated evidence of sameness
Regulatory principle:
FDA explicitly does not accept single-method validation for synthetic peptides because of their structural complexity, sensitivity to manufacturing variations, and potential for subtle modifications that may impact safety or efficacy.
A multi-technique analytical strategy is therefore essential to establish robust scientific proof of sameness and support successful ANDA approval.
4: Analytical Strategy for Sameness Evaluation
A robust analytical strategy for sameness evaluation ensures reliable confirmation that a synthetic peptide in an ANDA product is structurally and functionally equivalent to the Reference Listed Drug (RLD). This is achieved by integrating multiple complementary, orthogonal analytical techniques that together provide complete molecular characterization.
Explore: Analytical strategies for sameness study
Core Analytical Techniques
| Technique | Purpose | Outcome |
|---|---|---|
| LC-MS | Molecular weight confirmation | Verifies peptide mass accuracy and overall identity |
| HRMS | High-resolution structural analysis | Detects subtle mass differences and structural variations |
| Peptide Mapping | Sequence verification | Confirms amino acid order and primary structure integrity |
| 1D/2D NMR | Structural conformation analysis | Confirms 3D folding, spatial arrangement, and structural integrity |
| LC-MS/MS | Fragment analysis | Validates sequence, modifications, and site-specific changes |
Why this strategy is important:
Each technique provides a different layer of structural insight, and when combined, they eliminate analytical blind spots that may arise from using a single method.
Together, these methods create a comprehensive structural fingerprint of the peptide, ensuring high confidence in sameness evaluation and supporting regulatory acceptance in ANDA submissions.
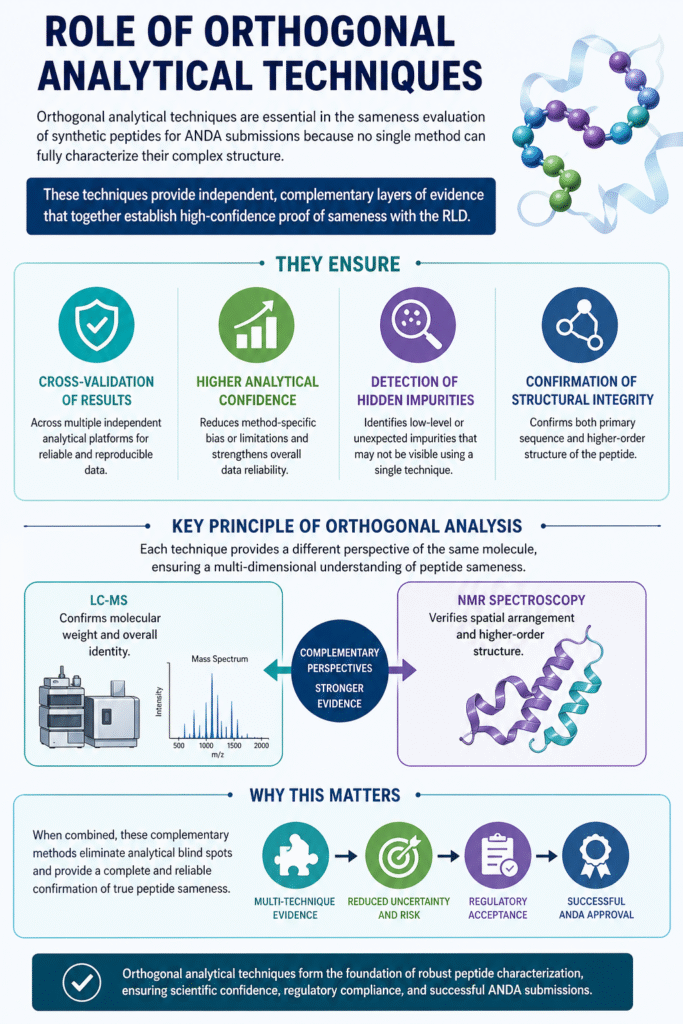
5: Role of Orthogonal Analytical Techniques
Orthogonal analytical techniques play a crucial role in sameness evaluation because no single analytical method is capable of fully characterizing the complex structure of synthetic peptides used in ANDA submissions.
These techniques provide independent, complementary layers of evidence that together establish high-confidence proof of sameness with the Reference Listed Drug (RLD).
Detailed guide: Orthogonal analytical techniques for ANDA sameness evaluation
They ensure:
- Cross-validation of results across multiple independent analytical platforms
- Higher analytical confidence by reducing method-specific bias or limitations
- Detection of hidden impurities that may not be visible in a single analytical approach
- Confirmation of structural integrity at both primary and higher-order structural levels
Key principle of orthogonal analysis:
Each technique provides a different perspective of the same molecule. For example:
- LC-MS confirms molecular weight and overall identity
- NMR spectroscopy verifies spatial arrangement and higher-order structure
Why this matters:
When combined, these complementary methods eliminate analytical blind spots and provide a complete and reliable confirmation of true peptide sameness, which is essential for regulatory acceptance in ANDA submissions.
6: Peptide Mapping in Sameness Evaluation
Peptide mapping is a cornerstone analytical technique in the sameness evaluation of synthetic peptides for ANDA submissions. It provides detailed confirmation of the primary structure by breaking the peptide into smaller, analyzable fragments and comparing them with the Reference Listed Drug (RLD).
Related studies:
How peptide mapping works:
- The peptide is enzymatically digested into specific fragments using proteolytic enzymes
- These fragments are then separated using high-performance chromatography (typically LC-based systems)
- The resulting fragment pattern is compared with the RLD peptide map to identify structural consistency
What peptide mapping ensures:
- Complete sequence coverage across the entire peptide chain
- Detection of post-translational or chemical modifications such as oxidation or deamidation
- Identification of missing, altered, or mis-incorporated residues that may affect functionality
Why it is important:
Peptide mapping provides high-resolution confirmation of primary structure, making it one of the most reliable tools for demonstrating structural sameness in regulatory ANDA submissions.
7: Impurity Profiling in Synthetic Peptides
Impurity profiling is a critical component of sameness evaluation in synthetic peptide-based ANDA submissions. It involves the identification, characterization, and quantification of related substances that may be present in the peptide drug product alongside the desired active pharmaceutical ingredient.
Learn more:
What impurity profiling does:
It systematically analyzes the peptide sample to detect any structurally related or process-derived variants that could impact safety, efficacy, or regulatory acceptance.
Common impurity types include:
- Truncated sequences resulting from incomplete peptide synthesis
- Deamidated forms arising from chemical instability of amino acid residues
- Oxidized variants caused by exposure to oxidative conditions during manufacturing or storage
- Aggregates and dimers formed due to peptide self-association or improper folding
Analytical approaches used:
Advanced high-sensitivity techniques such as HRMS (High-Resolution Mass Spectrometry) and LC-MS (Liquid Chromatography–Mass Spectrometry) are commonly employed to detect, characterize, and quantify impurities, even at trace levels.
Why it is important:
Comprehensive impurity profiling ensures:
- Product safety and tolerability
- Compliance with regulatory quality standards
- Accurate comparison with the Reference Listed Drug (RLD)
- Reduced risk of immunogenicity or reduced therapeutic performance
Robust impurity analysis is therefore essential to support reliable sameness evaluation and successful ANDA approval.

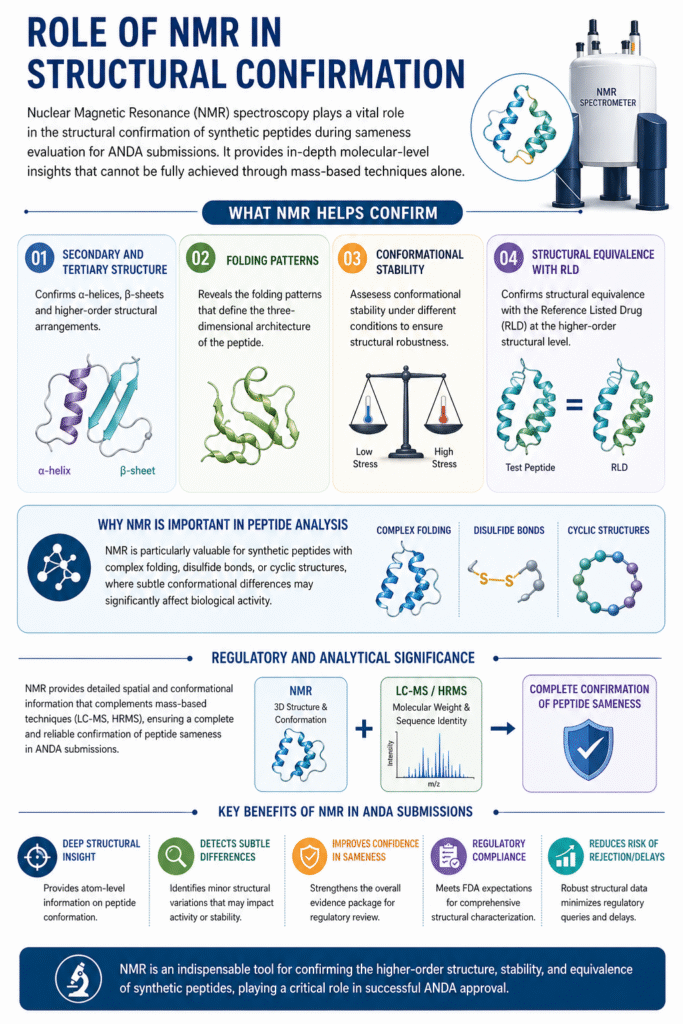
8: Role of NMR in Structural Confirmation
Nuclear Magnetic Resonance (NMR) spectroscopy plays a vital role in the structural confirmation of synthetic peptides during sameness evaluation for ANDA submissions. It provides in-depth molecular-level insights that cannot be fully achieved through mass-based techniques alone.
Connected analytical support:
What NMR helps confirm:
- Secondary and tertiary structure of peptides, including α-helices, β-sheets, and higher-order arrangements
- Folding patterns that define the three-dimensional architecture of the peptide
- Conformational stability under different conditions, ensuring structural robustness
- Structural equivalence with the Reference Listed Drug (RLD) at the higher-order structural level
Why NMR is important in peptide analysis:
NMR is particularly valuable for synthetic peptides with complex folding, disulfide bonds, or cyclic structures, where subtle conformational differences may significantly affect biological activity.
Regulatory and analytical significance:
By providing detailed spatial and conformational information, NMR complements techniques like LC-MS and HRMS, ensuring a complete and reliable confirmation of peptide sameness in ANDA submissions.
9: Challenges in Sameness Evaluation of Synthetic Peptides
Sameness evaluation of synthetic peptides in ANDA submissions is scientifically complex because peptides exhibit high structural sensitivity and require highly precise analytical control to ensure equivalence with the Reference Listed Drug (RLD).
Real-world regulatory challenges discussed in:
Key Challenges
- High structural variability
Synthetic peptides can exist in multiple micro-variants due to slight changes in synthesis, purification, or storage conditions. - Sensitivity to degradation
Peptides are prone to chemical instability, including oxidation, hydrolysis, and deamidation, which can alter their quality profile. - Difficulty in impurity separation
Structurally similar impurities often co-elute or overlap, making separation and identification analytically challenging. - Complex post-translational or chemical modifications
Modifications such as disulfide bond formation, glycosylation (in some cases), or side-chain alterations add analytical complexity. - Limited reference material availability
Access to high-quality RLD material can be restricted, making direct comparative analysis more difficult.
Mitigation Strategies
To address these challenges, a multi-layered analytical approach is applied:
- High-resolution instrumentation (HRMS, UHPLC)
Enables precise mass detection and improved separation of closely related variants. - Orthogonal analytical validation
Combines multiple independent techniques (LC-MS, NMR, peptide mapping) to confirm results and reduce analytical uncertainty. - Forced degradation studies
Helps understand degradation pathways and identify potential stability-related impurities. - Advanced bioinformatics for sequence validation
Supports accurate interpretation of peptide mapping and sequence confirmation data.
Overall impact:
By combining advanced instrumentation with orthogonal and computational approaches, these challenges can be effectively managed, ensuring robust and regulatory-compliant sameness evaluation in ANDA submissions.
10: Importance of Regulatory Compliance in ANDA Submissions
Regulatory compliance in ANDA submissions is essential to ensure a smooth approval process, minimize regulatory queries, and avoid unnecessary delays in product commercialization. For synthetic peptides, where structural complexity is high, compliance becomes even more critical.
Regulatory support services:
Why regulatory compliance matters:
It ensures that the submitted data is scientifically robust, reproducible, and fully aligned with FDA expectations for demonstrating sameness with the Reference Listed Drug (RLD).
FDA expectations include:
- Comprehensive analytical data package covering identity, purity, structure, and impurities
- Justification of analytical methods demonstrating suitability, sensitivity, and reliability for peptide characterization
- Cross-validation using multiple analytical techniques (orthogonal approaches such as LC-MS, HRMS, NMR, and peptide mapping)
- Clear comparative assessment with the RLD to establish structural and functional equivalence
Regulatory risk:
Failure to meet these requirements can lead to:
- Potential rejection or resubmission requirements due to insufficient evidence of sameness
- Complete Response Letters (CRLs) requesting additional data or clarification
- Extended review timelines, delaying approval and market entry
11: Role of ResolveMass Laboratories in Sameness Evaluation
ResolveMass Laboratories Inc. plays a critical role in supporting pharmaceutical companies with advanced peptide characterization.
With expertise in:
- High-resolution LC-MS
- Peptide mapping workflows
- HRMS-based impurity profiling
- 1D/2D NMR structural validation
The organization ensures that Sameness Evaluation of Synthetic Peptides for ANDA meets FDA expectations with scientific precision and regulatory reliability.
12: Why Multi-Technique Approach is Essential
A single analytical method is not sufficient to establish sameness of synthetic peptides in ANDA submissions because peptides are structurally complex and can exhibit subtle variations that may go undetected by one technique alone.
A multi-technique (orthogonal) analytical approach is therefore essential to achieve complete and reliable characterization.
Related scientific framework:
What a multi-technique approach ensures:
- Structural accuracy
Confirms primary, secondary, and higher-order structures using complementary methods such as LC-MS, HRMS, and NMR. - Functional equivalence
Ensures that the peptide behaves similarly to the Reference Listed Drug (RLD) in terms of biological activity and stability. - Regulatory compliance
Meets FDA expectations for comprehensive, multi-layered evidence rather than single-method validation. - Risk reduction in approval process
Minimizes the chances of missed impurities, structural misinterpretation, or incomplete characterization that could lead to regulatory queries or rejection.
Why this approach is critical:
Each analytical technique has limitations on its own, but when combined, they provide overlapping and confirming evidence that strengthens data reliability and scientific confidence.
Key takeaway:
The multi-technique strategy forms the foundation of modern peptide ANDA submissions, ensuring robust sameness evaluation, higher regulatory acceptance probability, and reduced development risk.
13: Future of Synthetic Peptide Analytics in ANDA
The future of synthetic peptide analytics in ANDA submissions is rapidly evolving toward higher precision, automation, and data-driven decision-making. These advancements aim to improve the accuracy of sameness evaluation while significantly reducing analysis time and regulatory uncertainty.
Emerging peptide applications:
Key emerging trends:
- AI-assisted spectral interpretation
Artificial intelligence and machine learning tools are increasingly being used to interpret complex LC-MS, HRMS, and NMR data, improving accuracy and reducing human bias in structural analysis. - Ultra-high-resolution mass spectrometry
Next-generation MS platforms provide enhanced sensitivity and resolution, enabling detection of ultra-trace impurities and minor structural variants that were previously undetectable. - Automated peptide mapping systems
Automation in sample preparation, digestion, and chromatographic analysis improves reproducibility, reduces manual errors, and accelerates workflow efficiency. - Integrated multi-omics analytical platforms
The convergence of proteomics, metabolomics, and advanced structural analytics enables a more holistic understanding of peptide behavior, stability, and equivalence.
Overall impact:
These technological advancements are expected to significantly reduce ambiguity in sameness evaluation, improve data reliability, and streamline regulatory submissions.
Conclusion:
The Sameness Evaluation of Synthetic Peptides for ANDA is a scientifically rigorous process that ensures generic peptides match their reference products in structure, purity, and function. FDA guidance strongly emphasizes the use of orthogonal analytical techniques, including LC-MS, HRMS, peptide mapping, and NMR, to establish complete equivalence.
For organizations like ResolveMass Laboratories Inc., this evaluation represents a critical intersection of analytical chemistry and regulatory science. By applying advanced characterization strategies, manufacturers can confidently demonstrate sameness, reduce regulatory risk, and ensure successful ANDA submissions.
Ultimately, robust sameness evaluation is not just a regulatory requirement—it is a cornerstone of patient safety and therapeutic consistency.
Learn more about peptide sameness study for ANDA
Frequently Asked Questions:
Sameness evaluation is the regulatory process used to confirm that a synthetic peptide in a generic drug is structurally, chemically, and functionally identical to the Reference Listed Drug (RLD). It ensures that the generic product matches the innovator in sequence, structure, purity, and biological performance.
It is important because even small structural differences in peptides can affect safety, efficacy, and immunogenicity. FDA requires proof of sameness to ensure that the generic peptide performs the same as the RLD with no clinically meaningful differences.
A single method cannot fully characterize the complex structure of synthetic peptides. Different techniques are needed to analyze sequence, mass, 3D structure, and impurities. This is why FDA requires a multi-technique approach.
Peptide mapping confirms the primary amino acid sequence by breaking the peptide into fragments and comparing them with the Reference Listed Drug. It helps detect sequence errors, modifications, or missing residues.
Impurity profiling identifies and quantifies related substances such as truncated peptides, oxidized forms, and aggregates. This ensures product safety, quality, and regulatory compliance while minimizing risk of immunogenic reactions.
Reference
- Wu L. Regulatory considerations for peptide therapeutics.https://books.rsc.org/books/edited-volume/801/chapter/540098
- De Groot AS, Roberts BJ, Mattei A, Lelias S, Boyle C, Martin WD. Immunogenicity risk assessment of synthetic peptide drugs and their impurities. Drug Discovery Today. 2023 Oct 1;28(10):103714.https://www.sciencedirect.com/science/article/pii/S1359644623002301
- Jois SD. Regulatory Issues for Peptide Drugs. InPeptide Therapeutics: Fundamentals of Design, Development, and Delivery 2022 Sep 27 (pp. 287-305). Cham: Springer International Publishing.https://link.springer.com/chapter/10.1007/978-3-031-04544-8_9
- Mitra MS, DeMarco S, Holub B, Thiruneelakantapillai L, Thackaberry EA. Development of peptide therapeutics: A nonclinical safety assessment perspective. Regulatory Toxicology and Pharmacology. 2020 Nov 1;117:104766.https://www.sciencedirect.com/science/article/pii/S0273230020301926
- Giri T, Sakharwade S, Subbappa P, Chinnakadoori SR, Sharma N. Regulatory Considerations in Synthetic Peptide Characterization: Techniques and Compliance. Separation Science Plus. 2025 Jun;8(6):e70057.https://analyticalsciencejournals.onlinelibrary.wiley.com/doi/abs/10.1002/sscp.70057


