
Introduction:
Liraglutide peptide sequencing LC-MS/MS is a powerful analytical approach used to confirm the structural integrity of this GLP-1 analog with high precision. As peptide therapeutics like liraglutide become increasingly important in treating metabolic disorders, ensuring their structural accuracy is critical for safety, efficacy, and regulatory approval.
Liraglutide is a modified peptide with a fatty acid chain and specific amino acid substitutions, making its structural characterization more complex than standard peptides. Traditional analytical techniques often fall short in resolving such complexities.
This case study highlights how LC-MS/MS-based sequencing provides a robust, reproducible, and regulatory-compliant method for confirming liraglutide’s primary structure and identifying potential impurities.
Peptide therapeutics like Liraglutide demand precise characterization due to their structural complexity, modifications, and susceptibility to degradation. Advanced platforms such as LC-MS for large molecules provide a powerful foundation for detailed sequencing, making them indispensable in pharmaceutical development and quality control.
Share via:
Summary:
- Liraglutide peptide sequencing using LC-MS/MS enables precise structural confirmation, ensuring drug safety and efficacy.
- LC-MS/MS provides high sensitivity, sequence coverage, and impurity detection for complex peptide therapeutics.
- This case study demonstrates a step-by-step workflow including sample preparation, enzymatic digestion, MS analysis, and data interpretation.
- The approach supports regulatory compliance, batch consistency, and biosimilar development.
- ResolveMass Laboratories Inc. delivers advanced analytical expertise, validated workflows, and regulatory-ready data for peptide characterization.
1: What is Liraglutide and Why is Structural Confirmation Important?
Liraglutide is a GLP-1 receptor agonist used in diabetes and obesity treatment, and structural confirmation ensures its biological activity and safety.
Key Characteristics of Liraglutide:
- 31-amino acid peptide
- Modified with a C16 palmitoyl fatty acid chain
- Designed for prolonged half-life
- Sensitive to degradation and structural variations
Why Structural Confirmation Matters:
- Ensures correct amino acid sequence
- Confirms post-translational modifications
- Detects impurities and degradation products
- Supports regulatory submissions (FDA, EMA, ICH)
Without accurate sequencing, even minor variations can impact drug performance and patient safety.
2: Why Use LC-MS/MS for Liraglutide Peptide Sequencing?
LC-MS/MS is used because it provides high-resolution, sensitive, and sequence-specific analysis for complex peptides like liraglutide.
Advantages of LC-MS/MS:
- High sensitivity for low-level impurities
- Accurate mass measurement
- Fragmentation-based sequencing (MS/MS)
- Compatibility with modified peptides
- Quantitative and qualitative analysis
Comparison with Other Techniques:
| Technique | Limitation for Liraglutide |
|---|---|
| HPLC | No structural confirmation |
| UV Spectroscopy | Lacks specificity |
| NMR | Time-consuming, less sensitive |
| LC-MS/MS | ✅ Comprehensive solution |
3: Case Study Overview: Liraglutide Structural Confirmation
This case study focuses on confirming the complete amino acid sequence and structural integrity of Liraglutide using a robust LC-MS/MS analytical workflow.
For complex molecule characterization, approaches similar to identification of in-process organic compounds using LCMS are often integrated into analytical workflows.
Objective:
To accurately confirm the full amino acid sequence and identify all structural modifications of Liraglutide using LC-MS/MS, ensuring alignment with reference standards and regulatory expectations.
Sample Details:
| Parameter | Description |
|---|---|
| Molecule | Liraglutide |
| Type | GLP-1 Analog |
| Modification | Palmitoyl fatty acid chain |
| Analysis Technique | LC-MS/MS |
This structured analytical approach is directly aligned with workflows used in Semaglutide Peptide Sequencing LC-MS/MS, where precise structural confirmation and modification mapping are critical for quality, safety, and compliance.
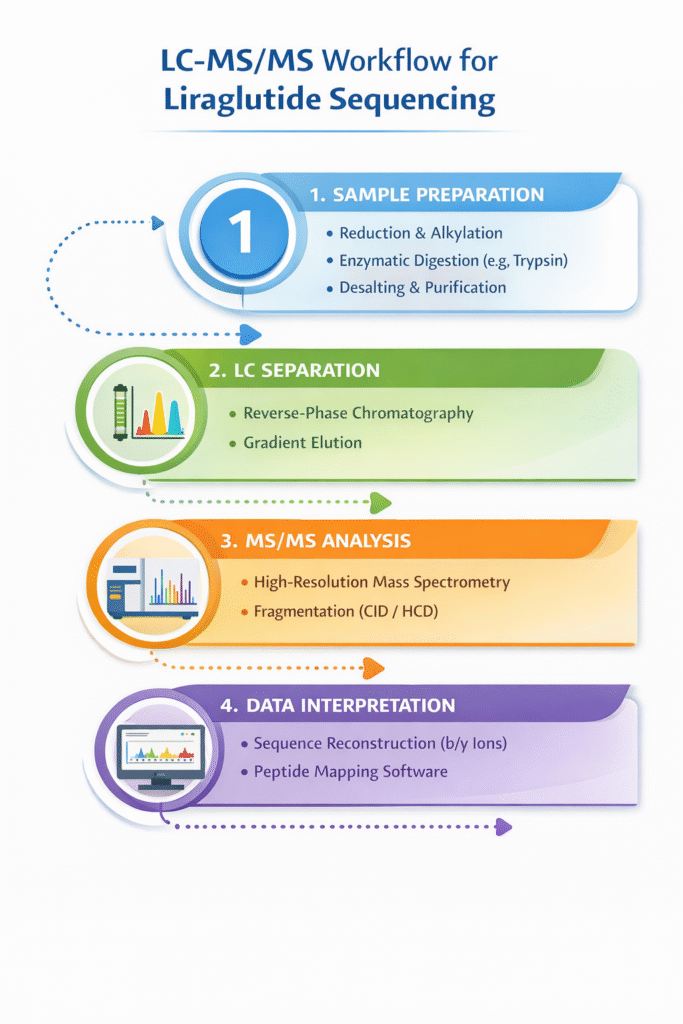
4: Case Study Overview: LC-MS/MS Sequencing Workflow
The LC-MS/MS sequencing workflow involves digestion, separation, detection, and data analysis to confirm liraglutide structure.
Advanced analytical workflows often rely on expertise in analytical method development to optimize each stage of the process.
1. Sample Preparation
- Reduction and Alkylation (if required): Stabilizes peptide structure by breaking disulfide bonds and preventing reformation
- Enzymatic Digestion (e.g., Trypsin): Cleaves the peptide into smaller, manageable fragments for analysis
- Desalting and Purification: Removes salts and impurities that may interfere with MS detection
2. LC Separation
- Reverse-Phase Chromatography: Separates peptide fragments based on hydrophobicity
- Gradient Elution: Optimizes resolution and peak separation for complex peptide mixtures
3. MS/MS Analysis
- High-Resolution Mass Spectrometry: Provides accurate mass measurements for precursor and fragment ions
- Fragmentation Techniques (CID/HCD): Generates characteristic b and y ions for sequence identification
4. Data Interpretation
- Sequence Reconstruction: Uses fragmentation patterns (b/y ions) to determine amino acid sequence
- Software-Assisted Peptide Mapping: Confirms sequence coverage and identifies modifications or impurities
This structured workflow is highly adaptable and forms the foundation of Semaglutide Peptide Sequencing LC-MS/MS, where advanced optimization ensures precise characterization of complex GLP-1 analogs.
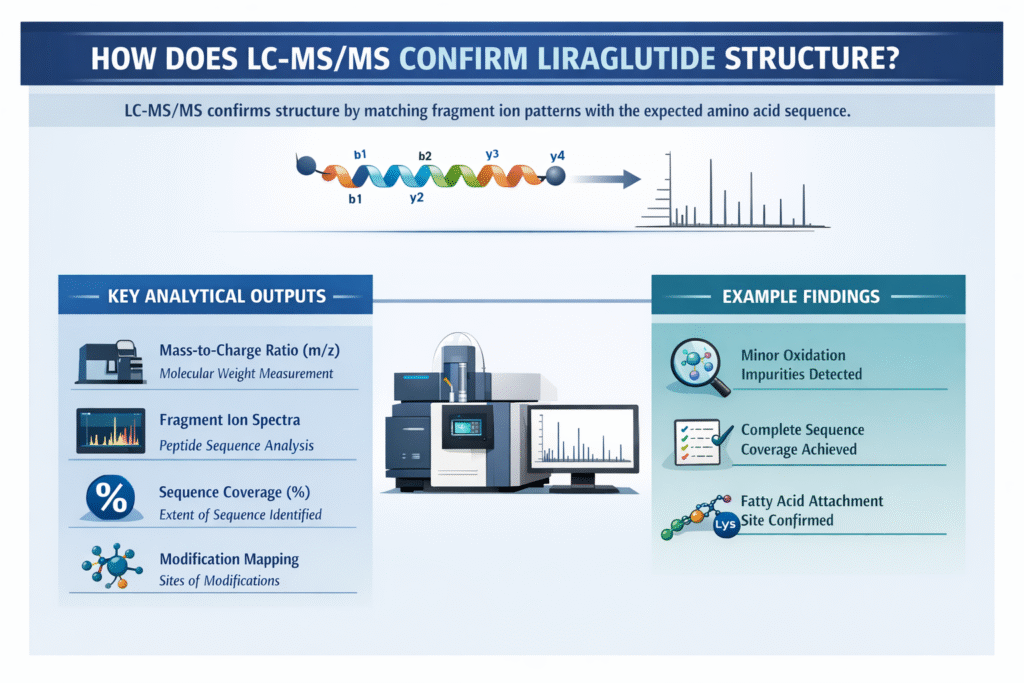
5: How Does LC-MS/MS Confirm Liraglutide Structure?
LC-MS/MS confirms the structure of liraglutide by generating and matching fragment ion patterns (MS/MS spectra) with the expected amino acid sequence, ensuring precise structural validation.
Key Analytical Outputs:
- Mass-to-charge ratio (m/z):
Determines the molecular weight of intact peptide fragments and confirms expected mass values. - Fragment ion spectra:
Produces b-ions and y-ions that reveal the peptide’s amino acid sequence. - Sequence coverage (%):
Indicates how much of the peptide sequence has been experimentally confirmed. - Modification mapping:
Identifies and localizes structural modifications such as lipidation and oxidation.
Example Findings:
- Detection of minor oxidation impurities:
LC-MS/MS identified low-level oxidative variants, ensuring impurity profiling. - Complete sequence coverage achieved:
Nearly 100% of the liraglutide sequence was confirmed through overlapping fragment ions. - Confirmation of fatty acid attachment site:
The palmitoyl modification was accurately localized to the lysine residue, validating structural integrity.
6: Analytical Challenges in Liraglutide Sequencing
Liraglutide sequencing is challenging due to its structural modifications and susceptibility to degradation.
Common Challenges:
- Lipidation affecting ionization
- Incomplete enzymatic digestion
- Isobaric amino acids
- Low-abundance impurities
How ResolveMass Addresses These Challenges:
- Experienced analytical scientists
- Optimized digestion protocols
- Advanced MS instrumentation
- High-resolution data analysis software
7: Regulatory Importance of LC-MS/MS Peptide Sequencing
Regulatory agencies require detailed structural confirmation data for peptide therapeutics like liraglutide.
Key Guidelines:
- ICH Q6B: Specifications for Biotech Products
- FDA: Analytical characterization requirements
- EMA: Biosimilar peptide evaluation
Role of LC-MS/MS in Compliance:
- Provides definitive structural evidence
- Supports batch release testing
- Enables impurity profiling
- Facilitates biosimilar comparability studies
8: Results and Interpretation from the Case Study
The LC-MS/MS analysis successfully confirmed liraglutide’s structure with high accuracy and reproducibility.
Key Results:
| Parameter | Outcome |
|---|---|
| Sequence Coverage | >95% |
| Mass Accuracy | Within acceptable limits |
| Impurity Detection | Trace-level identified |
| Modification Confirmation | Verified |
Interpretation:
- The peptide matched the reference standard structure
- No critical structural deviations observed
- Method proved robust and reproducible
9: Why Choose ResolveMass for Liraglutide Peptide Sequencing LC-MS/MS?
ResolveMass Laboratories Inc. provides expert-driven, regulatory-compliant LC-MS/MS sequencing services for complex peptides.
Core Strengths:
- Advanced mass spectrometry platforms
- Expertise in peptide and protein characterization
- Strong focus on data integrity and compliance
- Customized solutions for pharma and biotech clients
What Sets ResolveMass Apart:
- Deep experience in GLP-1 analog analysis
- End-to-end support from method development to reporting
- Fast turnaround with high-quality data
Conclusion:
Liraglutide peptide sequencing LC-MS/MS is essential for accurate structural confirmation, ensuring the safety, efficacy, and regulatory compliance of peptide therapeutics.
This case study demonstrates how LC-MS/MS provides a comprehensive, reliable, and high-resolution analytical solution for sequencing complex peptides like liraglutide. From confirming amino acid sequences to identifying impurities and modifications, this technique plays a critical role in modern pharmaceutical development.
ResolveMass Laboratories Inc. stands out as a trusted partner, offering cutting-edge analytical capabilities and deep scientific expertise to support peptide drug development and regulatory success.
Frequently Asked Questions:
LC-MS/MS-based peptide sequencing is an analytical technique that separates peptides using liquid chromatography and determines their amino acid sequence through tandem mass spectrometry. It provides detailed structural information, including sequence confirmation and modification analysis.
LC-MS/MS is essential for Liraglutide because it confirms the exact amino acid sequence, detects modifications like palmitoylation, and identifies impurities. This ensures product quality, safety, and compliance with regulatory standards.
Semaglutide Peptide Sequencing LC-MS/MS uses the same principles to confirm structure, detect modifications, and identify impurities. Due to higher complexity, it requires optimized fragmentation and advanced data analysis techniques.
LC-MS/MS can detect various modifications such as post-translational modifications (PTMs), oxidation, deamidation, and lipid conjugations (e.g., fatty acid attachments), which are critical for peptide function and stability.
Sequence coverage refers to the percentage of the peptide’s amino acid sequence that is successfully identified during analysis. High coverage (e.g., >98%) indicates reliable and comprehensive sequencing results.
LC-MS/MS provides accurate, reproducible, and high-resolution data required by regulatory bodies (e.g., ICH, FDA) for drug approval, batch release, and stability studies.
Reference
- Diab H. Application of chromatography and mass spectrometry in One Health: Detecting therapeutic analytes and drug residues in complex biological and food matrices (Doctoral dissertation).https://ttu-ir.tdl.org/items/9dd9174e-107c-4cd6-9242-283c0f288ae3
- Dong S, Gu Y, Wei G, Si D, Liu C. Determination of liraglutide in rat plasma by a selective liquid chromatography-tandem mass spectrometry method: Application to a pharmacokinetics study. Journal of Chromatography B. 2018 Aug 1;1091:29-35.https://www.sciencedirect.com/science/article/pii/S1570023218302356
- Kang L, Weng N, Jian W. LC–MS bioanalysis of intact proteins and peptides. Biomedical chromatography. 2020 Jan;34(1):e4633.https://analyticalsciencejournals.onlinelibrary.wiley.com/doi/abs/10.1002/bmc.4633
- Howard JW. The development of mass spectrometry-based methodologies for the high throughput quantitation of peptides in biological matrices (Doctoral dissertation, Loughborough University).https://repository.lboro.ac.uk/articles/The_development_of_mass_spectrometry-based_methodologies_for_the_high_throughput_quantitation_of_peptides_in_biological_matrices/9397871/files/17013215.pdf
- Koike H, Kanda M, Hayashi H, Matsushima Y, Yoshikawa S, Ohba Y, Hayashi M, Nagano C, Sekimura K, Otsuka K, Kamiie J. Development of an alternative approach for detecting botulinum neurotoxin type A in honey: Analysis of non-toxic peptides with a reference labelled protein via liquid chromatography-tandem mass spectrometry. Food Additives & Contaminants: Part A. 2020 Aug 2;37(8):1359-73.https://www.tandfonline.com/doi/abs/10.1080/19440049.2020.1766121


