
Introduction:
The International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use released the ICH M10 Bioanalytical Method Validation Guidelines to harmonize bioanalytical validation expectations across global regulatory agencies. For pharmaceutical companies, biotech innovators, and clinical research organizations, understanding the ICH M10 Bioanalytical Method Validation Guidelines is critical for successful drug development and regulatory approval.
Bioanalytical methods are essential for measuring drugs, metabolites, and biomarkers in biological matrices such as plasma, serum, urine, or tissues. Regulatory authorities rely heavily on bioanalytical data to evaluate pharmacokinetics, bioavailability, bioequivalence, and safety. Any inconsistency in analytical performance can compromise the reliability of clinical trial outcomes.
The ICH M10 guideline was developed to create a unified global framework that aligns requirements from agencies such as the United States Food and Drug Administration, European Medicines Agency, and Pharmaceuticals and Medical Devices Agency. This harmonization helps reduce duplicate studies, minimizes regulatory uncertainty, and streamlines international drug submissions.
For organizations developing pharmaceuticals, biologics, biosimilars, gene therapies, or complex formulations, understanding the practical implications of ICH M10 is essential for maintaining compliance and accelerating development timelines.
Summary:
- The Bioanalytical Method Validation guideline harmonizes global bioanalytical validation requirements across the US, Europe, and Japan.
- It establishes standardized expectations for assay validation, sample analysis, data integrity, and regulatory compliance.
- Drug developers must demonstrate accuracy, precision, selectivity, sensitivity, reproducibility, and stability of bioanalytical methods.
- The guideline applies to chromatographic, ligand-binding, and hybrid analytical methods used in pharmaceutical and biologic development.
- Proper implementation of ICH M10 reduces regulatory risk, supports smoother submissions, and improves global acceptance of bioanalytical data.
- Early collaboration with experienced bioanalytical laboratories helps companies avoid costly delays and failed validations.
1: What Are the ICH M10 Bioanalytical Method Validation Guidelines?
The ICH M10 Bioanalytical Method Validation Guidelines defines internationally accepted standards for validating bioanalytical methods used in regulatory submissions. The guideline establishes how laboratories must demonstrate that their analytical methods generate reliable, reproducible, accurate, and regulatory-compliant data when measuring drugs, metabolites, and biomarkers in biological samples.
In practical terms, the guideline explains how bioanalytical laboratories should validate methods used for pharmacokinetic, bioavailability, bioequivalence, toxicokinetic, and biomarker studies throughout drug development. It creates harmonized expectations across major regulatory agencies, helping pharmaceutical and biotechnology companies streamline global submissions.
The guideline covers several critical aspects of bioanalytical testing, including:
- Method development
- Full method validation
- Partial validation
- Cross-validation
- Sample analysis
- Incurred sample reanalysis (ISR)
- Stability assessments
- Documentation and reporting expectations
The ICH M10 guideline applies across a broad range of pharmaceutical and biotechnology applications:
| Applicable Areas | Examples |
|---|---|
| Small molecule drugs | Oral solid dosage forms, injectables |
| Biologics | Monoclonal antibodies, peptides |
| Biosimilars | Comparative PK studies |
| Gene and cell therapies | Biomarker quantification |
| Clinical trials | Phase I–IV studies |
| Bioequivalence studies | Generic drug development |
Because regulatory agencies increasingly emphasize data integrity and global harmonization, the ICH M10 Bioanalytical Method Validation Guidelines has become a critical framework for ensuring bioanalytical data quality and regulatory acceptance worldwide.
2: Why Were the ICH M10 Guidelines Introduced?
The primary purpose of the ICH M10 Bioanalytical Method Validation Guidelines is global harmonization of bioanalytical method validation requirements. The guideline was introduced to create a unified regulatory framework that aligns expectations across major international health authorities and reduces inconsistencies in bioanalytical data evaluation.
Before the implementation of ICH M10, regulatory agencies such as the United States Food and Drug Administration, European Medicines Agency, and Pharmaceuticals and Medical Devices Agency maintained separate bioanalytical validation guidance documents. As a result, pharmaceutical and biotechnology companies conducting multinational clinical studies often faced differing regional expectations for assay validation, sample analysis, and documentation.
This lack of consistency created several challenges, including:
- Duplicate validation studies
- Increased development timelines
- Higher operational costs
- Regulatory uncertainty
- Complicated global submissions
- Inconsistent data acceptance between regions
The ICH M10 Bioanalytical Method Validation Guidelines addresses these issues by establishing a single internationally accepted framework for bioanalytical method validation.
Key objectives of the guideline include:
- Creating globally accepted validation standards
- Reducing redundant validation activities
- Improving consistency in regulatory submissions
- Enhancing confidence in bioanalytical data
- Supporting efficient global drug approvals
- Strengthening data integrity and reproducibility
For pharmaceutical companies managing global clinical development programs, this harmonization significantly reduces both operational and regulatory burden. It also helps accelerate drug development by enabling bioanalytical data generated in one region to be more readily accepted by regulatory agencies worldwide.
Ultimately, the ICH M10 guideline improves the reliability, consistency, and scientific quality of bioanalytical data used to support drug safety and efficacy evaluations.
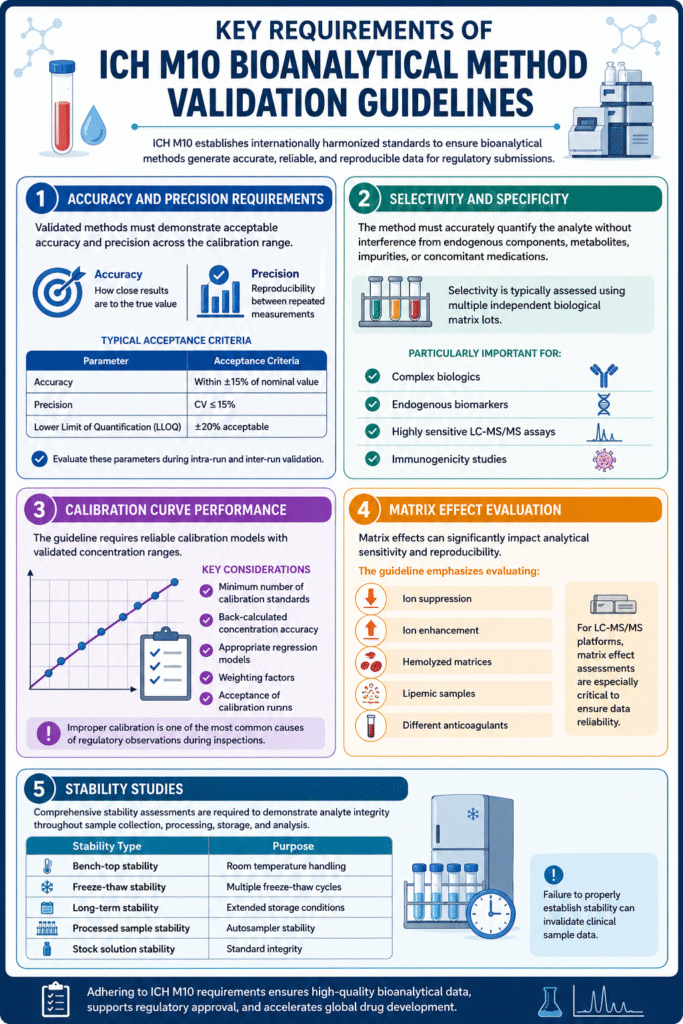
3: Key Requirements of ICH M10 Bioanalytical Method Validation Guidelines
The ICH M10 Bioanalytical Method Validation Guidelines establishes detailed validation requirements to ensure bioanalytical methods generate accurate, reproducible, and regulatory-compliant data throughout pharmaceutical development. These requirements apply to both small molecule and large molecule bioanalytical assays used in clinical and nonclinical studies.
Below are the major validation parameters drug developers and bioanalytical laboratories must address under ICH M10.
1. Accuracy and Precision Requirements
The guideline requires validated methods to demonstrate acceptable accuracy and precision across the calibration range.
Accuracy measures how close results are to the true value, while precision evaluates reproducibility between repeated measurements.
Typical acceptance criteria include:
| Parameter | Acceptance Criteria |
|---|---|
| Accuracy | Within ±15% of nominal value |
| Precision | CV ≤15% |
| Lower Limit of Quantification (LLOQ) | ±20% acceptable |
Drug developers must evaluate these parameters during intra-run and inter-run validation.
2. Selectivity and Specificity
The method must accurately quantify the analyte without interference from endogenous matrix components, metabolites, impurities, or concomitant medications.
Laboratories typically assess selectivity using multiple independent biological matrix lots.
This is particularly important for:
- Complex biologics
- Endogenous biomarkers
- Highly sensitive LC-MS/MS assays
- Immunogenicity studies
3. Calibration Curve Performance
The guideline requires reliable calibration models with validated concentration ranges.
Key calibration considerations include:
- Minimum number of calibration standards
- Back-calculated concentration accuracy
- Appropriate regression models
- Weighting factors
- Acceptance of calibration runs
Improper calibration remains one of the most common causes of regulatory observations during inspections.
4. Matrix Effect Evaluation
Matrix effects can significantly impact analytical sensitivity and reproducibility.
The ICH M10 guideline emphasizes evaluating:
- Ion suppression
- Ion enhancement
- Hemolyzed matrices
- Lipemic samples
- Different anticoagulants
For LC-MS/MS platforms, matrix effect assessments are especially critical to ensure data reliability.
5. Stability Studies
The guideline requires comprehensive stability assessments to demonstrate analyte integrity throughout sample handling and storage.
Required stability studies may include:
| Stability Type | Purpose |
|---|---|
| Bench-top stability | Room temperature handling |
| Freeze-thaw stability | Multiple freeze-thaw cycles |
| Long-term stability | Extended storage conditions |
| Processed sample stability | Autosampler stability |
| Stock solution stability | Standard integrity |
Failure to properly establish stability can invalidate clinical sample data.
4: Incurred Sample Reanalysis (ISR): A Major Focus Area
One of the most critical components of the ICH M10 Bioanalytical Method Validation Guidelines is Incurred Sample Reanalysis (ISR). ISR is performed to confirm that a validated bioanalytical method can consistently reproduce results when testing actual clinical or nonclinical study samples rather than artificially prepared quality control samples.
Unlike spiked QC samples prepared in controlled laboratory conditions, incurred samples contain real biological matrix complexities, metabolites, binding interactions, and patient-specific variables. ISR therefore provides an additional level of confidence in the reliability and robustness of the analytical method during real-world study execution.
The primary objective of ISR is to verify that the assay maintains reproducibility throughout sample analysis and supports the integrity of pharmacokinetic and bioequivalence data submitted to regulatory agencies.
The ICH M10 Bioanalytical Method Validation Guidelines generally recommends:
- Reanalysis of study samples near Cmax and elimination phases
- Selection of representative study samples
- Evaluation across different concentration ranges
- Acceptance criteria based on percentage differences between original and repeat results
- Assessment of assay reproducibility under routine analytical conditions
ISR is especially important for:
| Application Area | Importance of ISR |
|---|---|
| Pharmacokinetic studies | Confirms concentration reproducibility |
| Bioequivalence studies | Supports regulatory acceptance |
| Biologics and biosimilars | Evaluates complex matrix behavior |
| Large clinical trials | Demonstrates long-term assay consistency |
| LC-MS/MS assays | Confirms analytical robustness |
Regulatory agencies use ISR results to assess whether the analytical method performs consistently during actual study sample analysis. Poor ISR performance may indicate issues such as matrix effects, analyte instability, extraction variability, or method reproducibility problems.
For this reason, well-designed ISR programs are considered essential for maintaining data integrity and ensuring compliance with global bioanalytical validation expectations.
5: Partial Validation and Cross Validation Requirements
The ICH M10 Bioanalytical Method Validation Guidelines clearly explains when partial validation and cross validation are necessary to maintain the reliability, consistency, and regulatory acceptability of bioanalytical data. These validation approaches help ensure that method modifications or analytical differences do not compromise study results.
Both processes are essential for maintaining continuity throughout pharmaceutical development, especially during global clinical programs and method transfers.
Partial Validation:
Partial validation is performed when a previously validated bioanalytical method undergoes modifications that may impact analytical performance. Instead of repeating a full validation, laboratories evaluate only the affected parameters to confirm continued method suitability.
The extent of partial validation depends on the nature and significance of the method change.
Partial validation may be required when:
- Changing analytical instruments
- Modifying extraction methods
- Extending calibration ranges
- Changing anticoagulants
- Transferring methods between laboratories
- Altering sample preparation procedures
- Updating chromatographic conditions
- Introducing new biological matrices
Typical parameters evaluated during partial validation include:
| Validation Parameter | Purpose |
|---|---|
| Accuracy | Confirm result reliability |
| Precision | Evaluate reproducibility |
| Selectivity | Assess matrix interference |
| Stability | Verify analyte integrity |
| Calibration performance | Confirm concentration range suitability |
The ICH M10 Bioanalytical Method Validation Guidelines emphasizes that all method changes should be scientifically justified and properly documented to maintain regulatory compliance.
Cross Validation:
Cross validation is required when comparing analytical results generated using different bioanalytical methods, platforms, or laboratories. The purpose is to demonstrate that results remain comparable regardless of where or how the analysis was performed.
Cross validation is particularly important when multiple laboratories or analytical technologies are involved in the same development program.
This process is especially critical during:
- Global clinical studies
- CRO transfers
- Platform transitions
- Combination analytical approaches
- Bridging studies between methods
- Multi-site clinical trial analysis
Common cross-validation scenarios include:
| Scenario | Example |
|---|---|
| Method transfer | Sponsor lab to CRO |
| Platform comparison | LC-MS/MS vs ligand-binding assay |
| Multi-regional studies | Different global testing sites |
| Hybrid assays | Combining analytical technologies |
Proper cross-validation ensures:
- Consistency across datasets
- Comparable pharmacokinetic results
- Reliable regulatory submissions
- Reduced inter-laboratory variability
- Improved global data acceptance
Regulatory agencies expect scientifically sound cross-validation strategies whenever analytical results from multiple methods or laboratories contribute to the same study or submission package.
By implementing robust partial and cross-validation practices, pharmaceutical companies can maintain data integrity while adapting to evolving analytical and operational requirements throughout drug development.
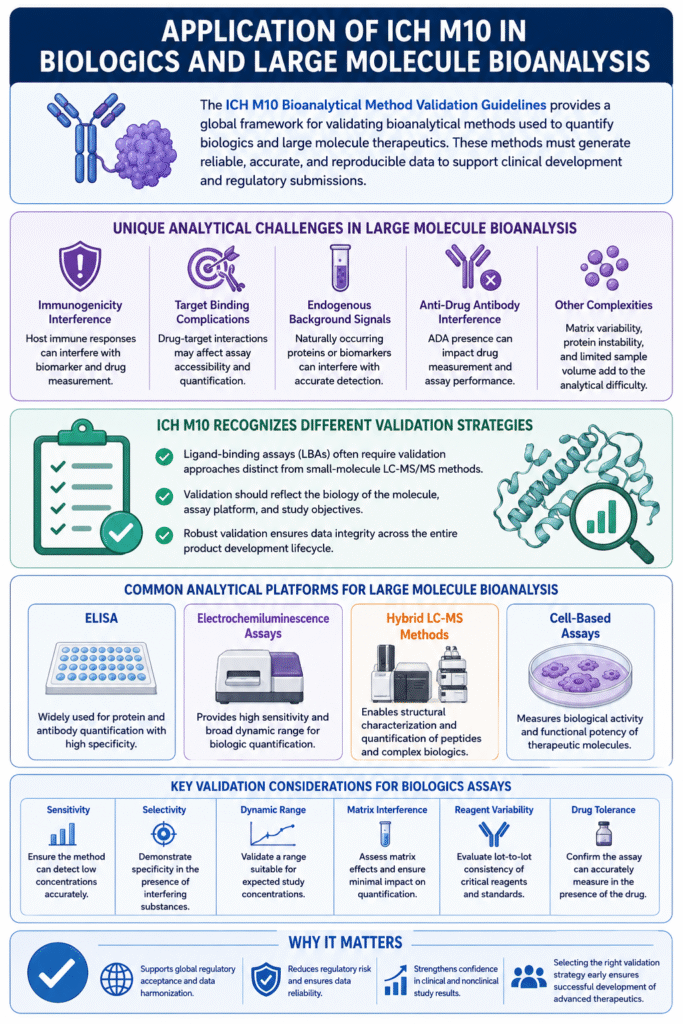
6: Application of ICH M10 in Biologics and Large Molecule Bioanalysis
The ICH M10 Bioanalytical Method Validation Guidelines plays a critical role in biologics and large molecule bioanalysis by establishing validation expectations for complex therapeutic products such as monoclonal antibodies, peptides, proteins, biosimilars, and advanced biologic therapies.
Unlike small molecule drugs, large molecule therapeutics often exhibit complex biological interactions that can significantly affect analytical performance. As a result, bioanalytical methods for biologics require specialized validation approaches to ensure reliable and reproducible data throughout drug development.
Large molecule assays frequently present unique analytical challenges, including:
- Immunogenicity interference
- Target binding complications
- Endogenous background signals
- Anti-drug antibody (ADA) interference
- Limited assay selectivity
- Matrix variability in biological samples
These factors can impact assay sensitivity, specificity, and quantification accuracy, particularly during long-term clinical studies.
The ICH M10 Bioanalytical Method Validation Guidelines recognizes that ligand-binding assays (LBAs) often require different validation strategies compared with traditional small-molecule Liquid Chromatography–Mass Spectrometry methods.
Common analytical platforms used in large molecule bioanalysis include:
| Analytical Platform | Typical Application |
|---|---|
| ELISA | Protein and antibody quantification |
| Electrochemiluminescence assays | High-sensitivity biologic analysis |
| Hybrid LC-MS methods | Peptide and complex biologic characterization |
| Cell-based assays | Functional bioactivity evaluation |
Each platform presents distinct validation considerations related to:
- Sensitivity
- Selectivity
- Dynamic range
- Matrix interference
- Reagent variability
- Drug tolerance
For example, ligand-binding assays may require careful evaluation of:
- Critical reagent stability
- Hook effect assessments
- Parallelism studies
- Dilution linearity
- Anti-drug antibody interference
Meanwhile, hybrid LC-MS approaches may require enhanced matrix effect investigations and optimized sample preparation strategies.
The increasing development of advanced therapeutics such as:
- Monoclonal antibodies
- Antibody-drug conjugates (ADCs)
- Cell and gene therapies
- RNA therapeutics
- Biosimilars
has made robust bioanalytical validation more important than ever.
For advanced biologic programs, selecting appropriate validation strategies early in development is essential for minimizing regulatory risk, ensuring data integrity, and supporting successful global submissions under the ICH M10 Bioanalytical Method Validation Guidelines.
7: Regulatory Inspection Readiness Under ICH M10
Regulatory inspection readiness is a critical component of compliance under the ICH M10 Bioanalytical Method Validation Guidelines. Global health authorities increasingly scrutinize bioanalytical laboratories to ensure that analytical data used in pharmaceutical development is accurate, traceable, reproducible, and fully compliant with regulatory expectations.
The ICH M10 guideline places strong emphasis on:
- Data integrity
- Audit trails
- SOP compliance
- Documentation practices
- Chain of custody
- Instrument qualification
- Analyst training
Inspection findings frequently involve:
- Incomplete documentation
- Poor chromatographic integration practices
- Uncontrolled method changes
- Insufficient stability data
- Inadequate ISR performance
Organizations should maintain inspection-ready quality systems throughout development.
8: Challenges Drug Developers Commonly Face
Implementing the ICH M10 Bioanalytical Method Validation Guidelines can be complex, particularly for pharmaceutical and biotechnology companies developing advanced therapeutics under aggressive timelines. Although the guideline provides a harmonized global framework, drug developers still face several scientific, operational, and regulatory challenges during bioanalytical method development and validation.
Addressing these challenges early is essential for maintaining data integrity, avoiding regulatory delays, and ensuring successful clinical development.
1. Complex Biological Matrices
Biological samples contain proteins, phospholipids, metabolites, and endogenous compounds that can interfere with analysis.
Advanced extraction techniques and optimized chromatography are often required.
2. Ultra-Low Quantification Requirements
Modern therapeutics may require picogram-level sensitivity.
Achieving reliable lower limits of quantification requires highly sensitive instrumentation and optimized sample preparation.
3. Global Regulatory Expectations
Multinational development programs must ensure data acceptability across multiple health authorities.
ICH M10 significantly simplifies this process but still requires expert interpretation and implementation.
4. Rapid Timelines
Pharmaceutical companies frequently face accelerated development schedules.
Delays in method validation can directly impact clinical timelines and regulatory submissions.
9: Best Practices for Successful ICH M10 Compliance
Successful implementation of the ICH M10 Bioanalytical Method Validation Guidelines requires more than simply meeting validation requirements. Pharmaceutical and biotechnology companies must establish scientifically robust, inspection-ready, and globally compliant bioanalytical practices throughout the drug development lifecycle.
By following proven best practices, drug developers can reduce regulatory risk, improve data reliability, and accelerate global submissions.
1. Engage Bioanalytical Experts Early
Early involvement of experienced bioanalytical scientists significantly improves the success of method development and validation programs.
Developing robust analytical methods during the early stages of pharmaceutical development helps identify potential challenges before formal validation begins.
Benefits of early bioanalytical planning include:
- Improved assay robustness
- Reduced validation failures
- Faster troubleshooting
- Better regulatory preparedness
- Optimized sample preparation strategies
- Enhanced analytical sensitivity and selectivity
Early scientific collaboration is particularly important for:
- Complex biologics
- Cell and gene therapies
- Biomarker assays
- Ultra-low concentration therapeutics
- Multi-analyte studies
Proactive planning reduces the likelihood of costly redevelopment later in clinical development.
2. Use Risk-Based Validation Strategies
The ICH M10 Bioanalytical Method Validation Guidelines supports scientifically justified, risk-based approaches to bioanalytical validation.
Rather than applying identical validation intensity to every parameter, laboratories should prioritize critical assay performance characteristics and high-risk analytical variables.
Risk-based validation focuses on factors that may significantly impact:
- Patient safety
- Pharmacokinetic interpretation
- Regulatory submissions
- Data reproducibility
- Assay reliability
Examples of high-risk areas include:
| High-Risk Variable | Potential Impact |
|---|---|
| Matrix effects | Quantification variability |
| Stability limitations | Invalid clinical data |
| Low concentration sensitivity | Poor assay reproducibility |
| Carryover effects | False positive results |
| Critical reagent variability | Inconsistent assay performance |
A structured risk-based strategy helps laboratories allocate resources efficiently while maintaining regulatory compliance.
3. Maintain Strong Documentation
Comprehensive and traceable documentation is essential for regulatory inspections, data integrity, and global submission readiness.
Regulatory agencies increasingly evaluate not only analytical results but also the quality systems supporting those results.
Critical documentation elements include:
- Standard operating procedures (SOPs)
- Validation protocols
- Analytical run records
- Audit trail reviews
- Instrument maintenance logs
- Deviation investigations
- Stability study records
- Training documentation
Proper documentation ensures:
- Full analytical traceability
- Reproducibility of results
- Regulatory transparency
- Inspection readiness
- Strong data integrity compliance
Incomplete or inconsistent records remain one of the most common causes of regulatory observations during inspections.
4. Conduct Pre-Validation Optimisation
Extensive method optimization before formal validation greatly improves long-term assay reliability and reduces the risk of validation failure.
Pre-validation optimization helps laboratories refine analytical conditions under controlled development settings before executing regulatory validation studies.
Optimization activities may include:
- Sample extraction refinement
- Chromatographic optimization
- Internal standard selection
- Matrix effect reduction
- Sensitivity enhancement
- Stability investigations
- Calibration range selection
Well-optimized methods typically demonstrate:
- Better reproducibility
- Lower variability
- Improved selectivity
- Greater robustness during routine analysis
Investing time in early optimization often saves substantial time and cost during later clinical phases.
5. Partner With Experienced Laboratories
Working with experienced bioanalytical laboratories provides :
- Regulatory expertise
- Advanced instrumentation
- Established SOP systems
- Faster troubleshooting
- Inspection-ready operations
10: How ResolveMass Laboratories Supports ICH M10 Compliance
ResolveMass Laboratories Inc. supports pharmaceutical and biotechnology companies with advanced bioanalytical testing services aligned with global regulatory expectations.
The laboratory supports:
- LC-MS/MS method development
- Bioanalytical method validation
- Stability studies
- Pharmacokinetic sample analysis
- Biomarker quantification
- Large molecule bioanalysis
- Regulatory-compliant documentation
With increasing regulatory scrutiny, partnering with experienced analytical scientists helps drug developers reduce risk and maintain high-quality data throughout development programs.
11: Future Impact of ICH M10 on Drug Development
The ICH M10 Bioanalytical Method Validation Guidelines will continue shaping pharmaceutical development worldwide.
As advanced therapeutics evolve, bioanalytical methods must adapt to increasingly complex analytical requirements. Areas expected to grow include:
- Cell and gene therapy bioanalysis
- Biomarker-driven clinical trials
- Hybrid analytical technologies
- High-resolution mass spectrometry
- AI-assisted data review
- Ultra-sensitive quantification techniques
Global harmonization under ICH M10 will remain central to ensuring reliable and reproducible bioanalytical data.
Conclusion:
The ICH M10 Bioanalytical Method Validation Guidelines establishes a globally harmonized framework for validating bioanalytical methods used in pharmaceutical and biologic development.
For drug developers, understanding these requirements is essential for regulatory compliance, successful submissions, and efficient global clinical development. The guideline emphasizes accuracy, precision, selectivity, stability, data integrity, and reproducibility to ensure trustworthy bioanalytical data.
Organizations that proactively implement robust validation strategies, maintain strong quality systems, and collaborate with experienced analytical laboratories are better positioned to reduce regulatory risk and accelerate development timelines.
As pharmaceutical science advances toward increasingly complex therapeutics, the role of compliant and scientifically rigorous bioanalysis will continue to grow.
Frequently Asked Questions:
ICH M10 helps pharmaceutical and biotechnology companies align their bioanalytical validation practices with global regulatory expectations. Before harmonization, companies often faced different regional requirements, increasing complexity and costs. The guideline simplifies multinational drug development by improving consistency in regulatory submissions. It also reduces duplicate validation work and strengthens confidence in analytical data. Ultimately, ICH M10 helps accelerate global drug approvals and improve compliance readiness.
The ICH M10 Bioanalytical Method Validation Guidelines is supported by major international regulatory agencies including the United States Food and Drug Administration, European Medicines Agency, and Pharmaceuticals and Medical Devices Agency. These agencies collaborated through the International Council for Harmonisation to create unified validation standards. The guideline helps ensure that bioanalytical data generated in one region is more readily accepted globally. This harmonization benefits multinational clinical programs and regulatory submissions.
Bioanalytical method validation is required for studies that involve measuring drugs, metabolites, or biomarkers in biological samples. Common applications include pharmacokinetic studies, bioavailability and bioequivalence studies, toxicokinetic testing, and clinical trials. It is also essential for biologics, biosimilars, and biomarker-driven development programs. Validation ensures that analytical methods perform consistently throughout the study lifecycle. Reliable bioanalytical data is critical for regulatory approval and patient safety evaluations.
Incurred Sample Reanalysis (ISR) involves retesting actual study samples to confirm the reproducibility of a validated analytical method. Unlike quality control samples, incurred samples contain real biological matrix complexities and patient-specific variables. ISR helps demonstrate that the assay performs consistently during routine clinical sample analysis. Regulatory agencies consider ISR an important measure of analytical reliability and data integrity. Poor ISR performance may indicate issues with method robustness or sample handling.
Cross validation is used to compare results generated by different analytical methods, laboratories, or platforms. It confirms that analytical data remains consistent regardless of where or how the samples are analyzed. Cross validation is especially important during global clinical studies, CRO transfers, or platform transitions. Regulatory agencies require this process when multiple datasets contribute to the same submission. Proper cross validation improves data comparability and global regulatory acceptance.
Experienced bioanalytical laboratories provide scientific expertise, advanced instrumentation, and established quality systems to support regulatory-compliant testing. These laboratories help pharmaceutical companies develop robust analytical methods and avoid costly validation failures. They also assist with stability studies, ISR programs, matrix effect evaluations, and regulatory documentation. Inspection-ready operations and experienced analysts improve data reliability and regulatory confidence. Partnering with a qualified laboratory can significantly streamline global drug development programs.
Reference
- Vazvaei-Smith F, Wickremsinhe E, Woolf E, Yu C. ICH M10 bioanalytical method validation guideline-1 year Later. The AAPS Journal. 2024 Sep 12;26(5):103.https://link.springer.com/article/10.1208/s12248-024-00974-y
- Guideline IH. Bioanalytical method validation and study sample analysis M10. ICH Harmonised Guideline: Geneva, Switzerland. 2022.https://www.inotiv.com/hubfs/ICH-Harmonized-Guideline-for-Bioanalytical-Method-Validation-and-Study-Sample-Analysis-M10.pdf
- Guideline IH. Bioanalytical method validation M10. European Medicines Agency: Amsterdam, The Netherlands. 2019 Feb.https://www.wrib.org/PDFs/ICH_M10_BMV_Draft_Guideline-190227.pdf
- Gu M, Gehman A, Nifong B, Mayer AP, Li V, Birchler M, Wang K, Tang H. From guidelines to implementation: a case study on applying ICH M10 for bioanalytical assay cross-validation. The AAPS Journal. 2025 Feb 28;27(2):54.https://link.springer.com/article/10.1208/s12248-025-01038-5
- Timmerman P, White S, Adcock N, Arfvidsson C, Barfield M, Cowan K, Ferrari L, Golob M, Goodwin L, Hughes R, Ivanova T. Feedback from a workshop by the European Bioanalysis Forum on assay validation requirements for in vitro assays following the publication of ICH M12 guideline–a plea for context-of-use over ICH M10 standards.https://www.tandfonline.com/doi/abs/10.1080/17576180.2025.2468596


