Introduction: Why Critical Quality Attributes (CQAs) in Biosimilars Need Advanced Analysis
The evaluation of Critical Quality Attributes (CQAs) in Biosimilars is one of the most important parts of biosimilar development. It is not only a regulatory requirement but also a scientific process that helps prove biosimilarity, product safety, and long-term therapeutic performance. Biosimilars are highly complex biological products, and even small structural changes can affect their efficacy, stability, or immunogenicity. Because of this complexity, detailed analytical characterization is essential during every stage of development.
Traditional analytical techniques such as HPLC, capillary electrophoresis, and UV spectroscopy are still valuable for routine testing. However, these methods often cannot provide the deep molecular-level information required for modern biosimilar evaluation. Regulatory agencies now expect more detailed analytical evidence for Critical Quality Attributes (CQAs) in Biosimilars, especially for complex biologics like monoclonal antibodies and fusion proteins. Mass spectrometry has become one of the most powerful technologies for meeting these expectations because it offers high sensitivity, accuracy, and detailed structural information.
Explore our comprehensive guide on Biosimilar Characterization Using Mass Spectrometry to see how advanced workflows streamline regulatory approval.
Share via:
Article Summary:
- Critical Quality Attributes (CQAs) in Biosimilars include important structural, functional, and post-translational characteristics that must be carefully analyzed to confirm similarity with the reference biologic product. Accurate evaluation of these attributes is essential for demonstrating biosimilar quality, safety, and performance.
- Mass spectrometry (MS) is widely recognized as one of the most advanced analytical technologies for detailed biosimilar characterization. It provides deep molecular-level insight, ranging from amino acid sequence verification to detailed glycan and glycoform analysis.
- Important CQAs commonly assessed using MS include glycosylation profiles, charge heterogeneity, oxidation levels, deamidation, disulfide bond connectivity, and higher-order structural properties studied through Hydrogen-Deuterium Exchange Mass Spectrometry (HDX-MS).
- Regulatory authorities such as the FDA and EMA are increasingly encouraging the use of Multi-Attribute Monitoring (MAM) workflows supported by advanced MS platforms during biosimilar development and comparability studies.
- Strong MS-based CQA assessment helps minimize the risk of immunogenicity, potency variation, and product inconsistency between the biosimilar and the reference biologic throughout the product lifecycle.
- ResolveMass Laboratories utilizes advanced LC-MS/MS, native MS, and HDX-MS technologies to provide high-quality biosimilar comparability data designed to support global regulatory expectations and development goals.
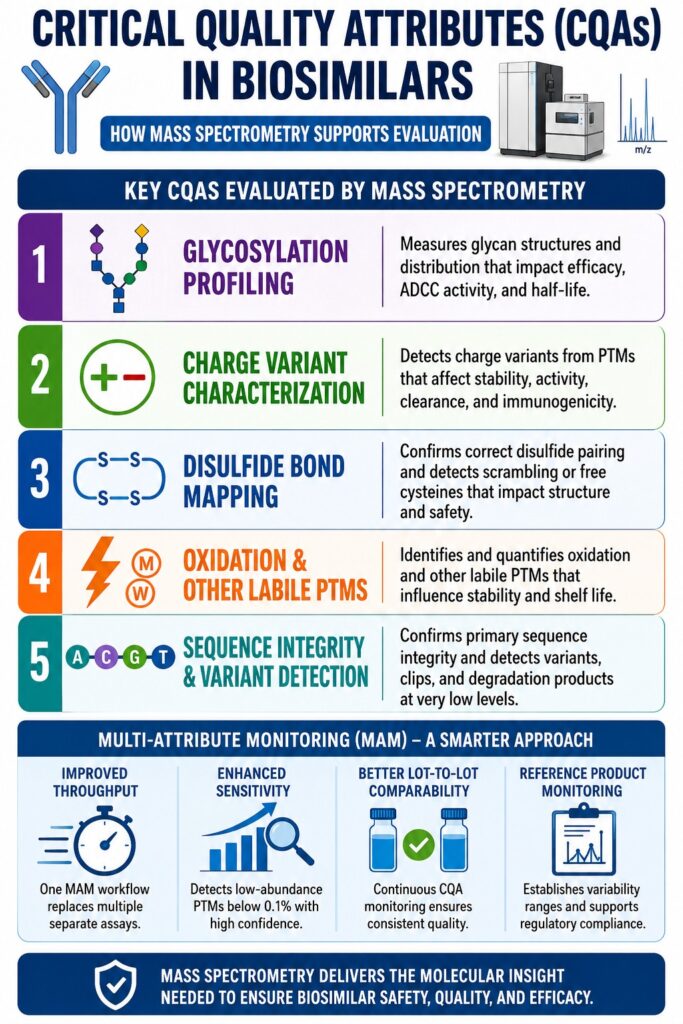
The CQA Landscape in Biosimilars: Key Attributes Evaluated by Mass Spectrometry
The most important Critical Quality Attributes (CQAs) in Biosimilars vary depending on the product structure, manufacturing process, and therapeutic mechanism. Some CQAs directly affect biological activity, while others influence stability, safety, or shelf life. Mass spectrometry plays a major role in evaluating these complex attributes with high precision.
1. Glycosylation Profiling: A Critical Quality Attribute in Biosimilars
Glycosylation is one of the most important CQAs in biosimilar characterization. A single glycosylation site may contain many glycan structures, and even small differences in glycoform distribution can affect Fc receptor binding, serum half-life, and antibody-dependent cellular cytotoxicity (ADCC). Because of this, accurate glycan analysis is essential for regulatory approval and product consistency.
Mass spectrometry supports glycosylation analysis using several advanced techniques:
Released Glycan Analysis (PNGase F + LC-MS)
This method identifies and measures individual N-glycan structures with very high sensitivity. It is especially useful for evaluating core fucosylation levels that influence ADCC activity. Researchers can compare glycan profiles between the biosimilar and reference product with excellent accuracy.
Intact Glycoprotein Mass Spectrometry
Intact MS provides a complete overview of glycoform distribution under native or denaturing conditions. This approach helps scientists directly compare the molecular profile of the biosimilar with the innovator product and identify even small structural differences.
Site-Specific Glycopeptide Mapping
This technique evaluates glycosylation occupancy at specific glycosylation sites. It is especially valuable for products with multiple glycosylation regions because each site may have different regulatory importance and functional impact.
For monoclonal antibodies, glycoforms such as G0F, G1F, G2F, and afucosylated species must remain within defined comparability ranges. High-resolution mass spectrometry is one of the few technologies capable of providing this level of detailed characterization.
Learn more about our specialized Glycosylation Analysis of Biosimilars
and Intact Mass Analysis services.
2. Charge Variant Characterization in Critical Quality Attributes (CQAs) in Biosimilars
Charge heterogeneity may result from deamidation, lysine clipping, pyroglutamate formation, or changes in sialylation. These charge variants can influence product stability, biological activity, clearance rate, and immunogenicity. Accurate characterization is important for maintaining biosimilar consistency.
| Charge Variant Type | MS Detection Method | Regulatory Relevance |
|---|---|---|
| Deamidation (Asn → Asp/isoAsp) | Peptide mapping LC-MS/MS | Influences binding and stability |
| C-terminal Lys clipping | Intact mass analysis | Alters charge distribution |
| Pyroglutamate formation | N-terminal peptide MS | May impact potency |
| Succinimide intermediates | Middle-down MS | Indicates structural instability |
| Sialylation variants | Glycopeptide MS | Modulates serum half-life |
Multi-attribute monitoring (MAM) workflows based on LC-MS/MS allow simultaneous monitoring of several charge-related CQAs in one analytical run. This approach improves efficiency while delivering highly specific analytical data.
Discover Mass Spectrometry Approaches for Charge Variant Analysis to manage molecular heterogeneity.
3. Disulfide Bond Mapping and Scrambling Detection
Correct disulfide bond formation is critical for biosimilar structural integrity. Problems such as incomplete pairing, disulfide scrambling, or free cysteines may affect protein folding and increase the risk of immunogenicity. These issues are particularly important in complex biologics where structural stability directly affects therapeutic function.
Mass spectrometry-based peptide mapping under non-reducing conditions can:
- Confirm proper intra-chain and inter-chain disulfide pairing
- Detect very low levels of disulfide scrambling below 1%
- Identify free cysteine variants linked to oxidative stress or process conditions
This level of analytical precision is especially valuable for biologics such as etanercept and abatacept, where disulfide architecture is closely connected to biological performance.
Access detailed insights on Peptide Mapping in Biosimilars for structural verification.
4. Monitoring Oxidation and Other Labile PTMs
Oxidation of methionine and tryptophan residues is a major stress-related CQA that may impact biosimilar stability and shelf life. Oxidation inside complementarity-determining regions (CDRs) can also reduce binding affinity and biological activity. Monitoring these changes is essential during stability studies and product development.
Peptide mapping MS combined with forced degradation studies helps researchers:
- Identify oxidation-sensitive regions in the biosimilar sequence
- Quantify oxidation levels at individual sites with high accuracy
- Establish scientifically justified acceptance criteria for stability testing
This detailed analysis helps maintain product quality throughout manufacturing and storage.
See how we handle Post-Translational Modifications (PTMs)
and Forced Degradation Studies to ensure long-term stability.
5. Sequence Integrity and Variant Detection
A biosimilar must maintain the same primary amino acid sequence as the reference product. LC-MS/MS peptide mapping with more than 95% sequence coverage is considered the gold standard for confirming sequence integrity and detecting unexpected variants.
Mass spectrometry can also identify:
- Low-level amino acid substitutions caused by misincorporation
- N-terminal and C-terminal heterogeneity
- Clip sites and degradation-related fragments
This high sensitivity allows manufacturers to detect even very small sequence variations that may affect product quality or clinical performance.
Need to Prove Biosimilarity Using LC-MS? Consult our sequence integrity experts.
Multi-Attribute Monitoring (MAM) for Critical Quality Attributes (CQAs) in Biosimilars
Regulatory agencies such as the FDA and EMA increasingly recognize the value of MAM workflows for biosimilar characterization. In many cases, MAM has become an important part of analytical comparability packages because it allows several CQAs to be monitored together in one workflow.
MAM replaces the traditional “one test for one attribute” approach by using a single LC-MS/MS platform to monitor multiple post-translational modifications in a single run. This improves efficiency while maintaining high analytical confidence.
Key Benefits of MAM in Biosimilar Development
Improved Throughput
A single MAM workflow can replace several separate analytical assays. This reduces testing time and supports faster biosimilar development.
Enhanced Sensitivity
Low-abundance PTMs below 0.1% can be detected routinely, reducing the possibility of missing clinically relevant differences.
Better Lot-to-Lot Comparability
MAM allows continuous monitoring of CQA changes across manufacturing batches. This helps manufacturers maintain process consistency and regulatory compliance.
Reference Product Monitoring
Ongoing analysis of innovator product lots helps establish scientifically justified variability ranges and acceptance criteria.
The strong alignment of MAM with ICH Q6B guidance and FDA analytical frameworks makes it both a scientific and strategic advantage in biosimilar development.
Get a complete overview of Impurity Profiling of Biosimilars using multi-attribute workflows.
Higher-Order Structure (HOS) Assessment in Critical Quality Attributes (CQAs) in Biosimilars
Biosimilar characterization extends beyond primary sequence analysis. Regulatory agencies also expect evidence that secondary, tertiary, and quaternary structures are comparable to those of the reference product. Structural differences at higher-order levels may affect protein folding, stability, and biological function.
Hydrogen-Deuterium Exchange Mass Spectrometry (HDX-MS) provides highly detailed structural information by measuring deuterium incorporation into protein backbone amide bonds. This helps scientists evaluate conformational flexibility and solvent accessibility with residue-level precision.
HDX-MS is particularly useful for:
- Detecting subtle Fc-region conformational changes
- Assessing epitope comparability in antigen-binding regions
- Evaluating glycosylation-related effects on protein dynamics
Native mass spectrometry further supports HOS evaluation by preserving non-covalent interactions during analysis. This enables characterization of protein complexes, subunit assembly, and ligand-binding states.
Converting Mass Spectrometry Data into Regulatory-Ready Evidence
Generating high-quality analytical data is only part of successful biosimilar characterization. The information must also be translated into a clear scientific and regulatory narrative that demonstrates comparability and product quality.
This process often includes:
- Statistical similarity evaluation using equivalence testing and glycan distribution analysis
- Risk-based prioritization of CQAs based on their effect on safety and efficacy
- Bridging studies that connect analytical findings with functional assay outcomes
Modern biosimilar development programs use mass spectrometry as part of a complete CQA strategy that supports process development, manufacturing control, and regulatory submissions.
Streamline your path to market with our Biosimilar Comparability Studies.
Conclusion: Mass Spectrometry as the Foundation of Critical Quality Attributes (CQAs) in Biosimilars
The evaluation of Critical Quality Attributes (CQAs) in Biosimilars has become increasingly detailed and scientifically demanding. Conventional analytical methods alone are no longer sufficient for understanding complex glycan heterogeneity, charge variants, oxidation pathways, disulfide bond integrity, and higher-order structures.
Mass spectrometry provides the molecular-level resolution, sensitivity, and quantitative accuracy required for modern biosimilar development. Technologies such as LC-MS/MS, native MS, HDX-MS, and MAM workflows deliver the detailed characterization needed to support regulatory approval and ensure biosimilar safety, quality, and efficacy.
At ResolveMass Laboratories Inc., our analytical scientists specialize in advanced biosimilar characterization using LC-MS/MS, native MS, HDX-MS, and MAM technologies. We understand that every analytical result contributes to a broader scientific and regulatory framework where accuracy, interpretation, and compliance are equally important.
Frequently Asked Questions (FAQs)
Certain Critical Quality Attributes (CQAs) carry a higher regulatory risk because they can directly affect the safety and effectiveness of a biosimilar. Differences in glycosylation patterns, especially afucosylation levels linked to ADCC activity, are among the most closely monitored attributes. Aggregation-related issues are also a major concern because they may increase immunogenicity risk and impact product stability. Regulatory agencies often request additional comparability studies when these attributes fall outside acceptable ranges.
Mass spectrometry is one of the most powerful tools for evaluating biosimilar CQAs, but it cannot completely replace all other analytical methods. Regulatory agencies still expect orthogonal techniques such as SPR for binding analysis, SEC for aggregation testing, and cell-based assays for potency evaluation. Each method provides different scientific information that supports a complete comparability package. Using multiple analytical approaches helps ensure accurate and reliable biosimilar characterization.
Traditional peptide mapping mainly focuses on confirming amino acid sequence identity and identifying major modifications within the protein structure. Multi-Attribute Monitoring (MAM) goes further by allowing simultaneous monitoring and quantification of many post-translational modifications during a single analytical run. MAM also uses reference standards and automated workflows to improve consistency and efficiency. This makes it highly useful for both product development and routine quality monitoring.
A full mass spectrometry-based characterization study usually requires around 1–5 mg of purified biosimilar protein. This amount supports detailed testing such as glycan profiling, intact mass analysis, peptide mapping, and HDX-MS studies. However, advances in analytical sensitivity now allow some workflows to operate with much smaller sample quantities. The exact requirement depends on the complexity of the molecule and the scope of the analytical program.
Glycan heterogeneity can influence how the immune system reacts to a biosimilar product. Certain non-human glycan structures, such as alpha-gal or Neu5Gc, may trigger anti-drug antibody responses in patients. These immune reactions can affect treatment safety and therapeutic performance. Mass spectrometry-based glycan profiling helps detect these glycan species at very low levels before the product enters clinical development.
Yes, HDX-MS is recognized by the FDA as a scientifically appropriate method for evaluating higher-order structure comparability in biosimilars. The technique provides detailed information about protein folding, conformational stability, and structural dynamics at the residue level. It is particularly valuable when differences are observed using other structural analysis methods. HDX-MS supports a deeper understanding of biosimilar structural similarity.
The importance of charge variants depends on how strongly they affect biological activity and product performance. Deamidation within complementarity-determining regions (CDRs) is generally considered highly critical because it can directly influence antigen binding. C-terminal lysine clipping is usually viewed as lower risk since it has limited impact on function. The effect of N-terminal pyroglutamate formation varies depending on the specific monoclonal antibody.
Intact mass analysis provides a broad overview of the biosimilar’s molecular weight and overall modification profile. It is useful for quickly identifying large structural changes or major post-translational modifications. Peptide mapping, on the other hand, offers detailed site-specific information about individual amino acid modifications. Both techniques are complementary and are commonly included together in comprehensive biosimilar characterization studies.
Reference:
- National Center for Biotechnology Information. (2024). Reverse engineering and characterization of long-acting injectable microspheres: Current approaches and challenges. PubMed Central. https://pmc.ncbi.nlm.nih.gov/articles/PMC10892806/
- European Medicines Agency. (2025). Reflection paper on a tailored clinical approach in biosimilar development (EMA/CHMP/BMWP/60916/2025). https://www.ema.europa.eu/en/documents/other/reflection-paper-tailored-clinical-approach-biosimilar-development_en.pdf
- National Center for Biotechnology Information. (2023). Recent advances in long-acting injectable microsphere formulations: Design, characterization, and challenges. PubMed Central. https://pmc.ncbi.nlm.nih.gov/articles/PMC10432323/



