
Introduction:
Toxicological Assessment in Extractables & Leachables Studies is a critical scientific process used to evaluate whether chemical compounds migrating from packaging systems, medical devices, manufacturing components, or delivery systems could harm patients. These studies are essential in pharmaceutical development because even trace levels of contaminants may impact product safety, efficacy, or regulatory approval.
As pharmaceutical formulations become increasingly complex, regulators worldwide expect robust extractables and leachables (E&L) programs supported by scientifically justified toxicological risk assessments. Companies developing injectable drugs, inhalation products, biologics, ophthalmics, and combination devices must establish acceptable safety limits for potential leachables before commercialization. Organizations seeking guidance on ICH Q3E Extractables and Leachables Study Requirements and ICH Q3E Guideline for Extractables and Leachables are increasingly focusing on risk-based toxicological evaluations aligned with global regulatory expectations.
At ResolveMass Laboratories Inc., toxicological evaluation is integrated with advanced analytical characterization to help pharmaceutical and medical device manufacturers identify, quantify, and assess chemical risks throughout product development and regulatory submission processes.
Summary:
- Toxicological Assessment in Extractables & Leachables Studies helps determine whether chemicals migrating from pharmaceutical packaging, medical devices, or manufacturing systems pose patient safety risks.
- Extractables are compounds that can be pulled from materials under aggressive laboratory conditions, while leachables are compounds that actually migrate into the drug product during normal storage or use.
- Safety limits are established using toxicological risk assessment methods such as PDE (Permitted Daily Exposure), TTC (Threshold of Toxicological Concern), and safety margin calculations.
- Regulatory agencies including the U.S. Food and Drug Administration, European Medicines Agency, and International Council for Harmonisation expect scientifically justified toxicological evaluations for E&L programs.
- Modern E&L studies combine analytical chemistry, toxicology, risk assessment, and regulatory expertise to ensure patient safety and regulatory compliance.
- Toxicological assessments are essential for pharmaceuticals, biologics, combination products, inhalation therapies, ophthalmics, and medical devices.
- Companies preparing global regulatory submissions often review requirements for Extractables and Leachables E&L Requirements for U.S. Market Authorization and Extractables and Leachables E&L Testing in the United States.
1: What Are Extractables and Leachables?
Extractables and leachables are chemical compounds that originate from materials used in pharmaceutical packaging, processing equipment, medical devices, or delivery systems. Advanced studies involving EL Testing for Pre-Filled Syringes and Packaging Leachables Nitrosamine E&L Studies are increasingly important because modern drug delivery systems involve complex polymeric materials and packaging components.
Extractables:
Extractables are compounds that can be released from materials under aggressive laboratory extraction conditions such as:
- Elevated temperatures
- Strong solvents
- Extended extraction times
- Extreme pH conditions
These studies are designed to identify potential chemical migrants that could appear during the product lifecycle.
Common Sources of Extractables
| Material Source | Examples |
|---|---|
| Plastic packaging | Bottles, tubing, bags |
| Elastomers | Stoppers, seals, gaskets |
| Medical devices | Catheters, inhalers |
| Manufacturing systems | Filters, single-use systems |
| Coatings and adhesives | Labels, laminated materials |
Leachables:
Leachables are compounds that actually migrate into the drug product during normal manufacturing, storage, or administration conditions.
Unlike extractables, leachables directly impact patient exposure because they are present in the final product consumed by patients.
Common Leachable Risks
- Drug degradation
- Toxicity concerns
- Product instability
- Immunogenicity
- Regulatory non-compliance
2: Why Toxicological Assessment in Extractables & Leachables Studies Is Important
The primary purpose of Toxicological Assessment in Extractables & Leachables Studies is to establish whether detected compounds are safe at observed exposure levels.
Even low-level chemical contaminants may create risks depending on:
- Route of administration
- Duration of exposure
- Patient population
- Dose frequency
- Compound toxicity profile
For example:
| Product Type | Toxicological Concern |
|---|---|
| Inhalation products | Pulmonary toxicity |
| Injectables | Systemic toxicity |
| Ophthalmics | Ocular irritation |
| Biologics | Protein interaction |
| Pediatric drugs | Increased sensitivity |
Regulatory agencies expect manufacturers to demonstrate that identified leachables remain below toxicological safety thresholds throughout the product shelf life. Manufacturers developing biologics and advanced therapies frequently require specialized support for Extractables and Leachables in Biologics and ATMPs and Extractables and Leachables in Emerging Biologics and Advanced Therapies.
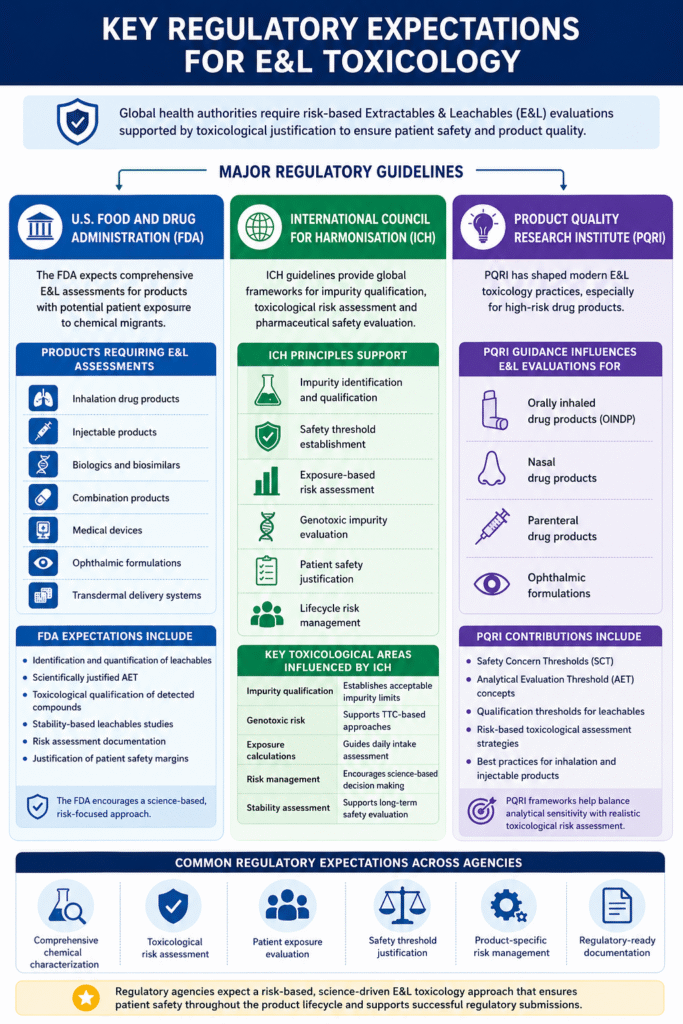
3: Key Regulatory Expectations for E&L Toxicology
Global health authorities require scientifically justified, risk-based extractables and leachables (E&L) evaluations to ensure patient safety and product quality. Regulatory agencies expect pharmaceutical and medical device manufacturers to identify potential leachables, assess toxicological risks, establish acceptable exposure limits, and demonstrate that detected compounds remain within safe limits throughout the product lifecycle.
Regulatory expectations generally focus on:
- Comprehensive chemical characterization
- Toxic
- Patient exposure evaluation
- Safety threshold justification
- Product-specific risk management
- Documentation suitable for regulatory submissions
As drug delivery systems and packaging technologies become more complex, regulatory scrutiny surrounding E&L toxicology continues to increase, especially for high-risk dosage forms such as inhalation products, injectables, biologics, and combination devices.
Organizations preparing submissions for regulated markets often review:
- ICH Q3E Extractables Leachables Risk Assessment
- FDA Extractables and Leachables Case Studies
- E&L Testing USA vs Europe
These resources help manufacturers understand evolving regulatory expectations for toxicological qualification, analytical thresholds, and risk management strategies.
Major Regulatory Guidelines:
U.S. Food and Drug Administration
The FDA expects comprehensive E&L assessments for:
- Inhalation drug products
- Injectable products
- Biologics
- Combination products
- Medical devices
International Council for Harmonisation
ICH guidelines support toxicological qualification principles including:
- Impurity assessment
- Safety thresholds
- Exposure calculations
- Risk management
Product Quality Research Institute
PQRI established influential safety threshold concepts for:
- Orally inhaled products
- Nasal drug products
- Parenteral and ophthalmic drug products
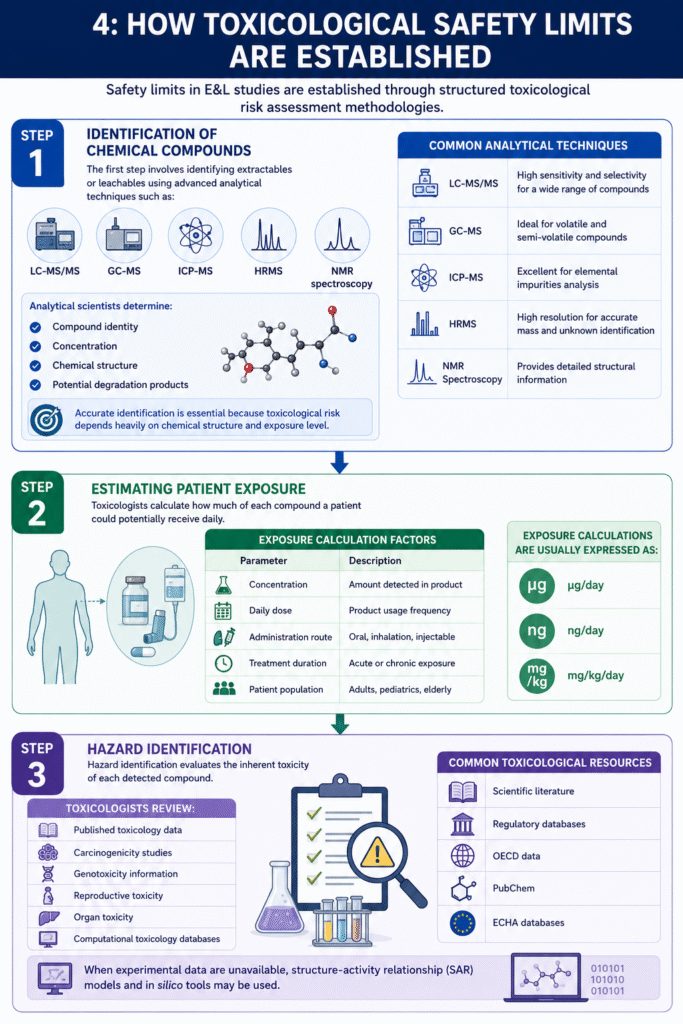
4: How Toxicological Safety Limits Are Established
Safety limits in E&L studies are established through structured toxicological risk assessment methodologies.
Manufacturers pursuing regulatory approvals such as NDA and ANDA submissions often require scientifically justified safety evaluations through Extractables and Leachables E&L Testing for Drug Safety for NDA ANDA Submissions.
Step 1: Identification of Chemical Compounds
The first step involves identifying extractables or leachables using advanced analytical techniques such as:
- LC-MS/MS
- GC-MS
- ICP-MS
- HRMS
- NMR spectroscopy
Analytical scientists determine:
- Compound identity
- Concentration
- Chemical structure
- Potential degradation products
Accurate identification is essential because toxicological risk depends heavily on chemical structure and exposure level. This is particularly important in Extractables and Leachables Carcinogenicity Testing where trace-level genotoxic or carcinogenic impurities require careful evaluation.
Step 2: Estimating Patient Exposure
Toxicologists calculate how much of each compound a patient could potentially receive daily.
Exposure Calculation Factors
| Parameter | Description |
|---|---|
| Concentration | Amount detected in product |
| Daily dose | Product usage frequency |
| Administration route | Oral, inhalation, injectable |
| Treatment duration | Acute or chronic exposure |
| Patient population | Adults, pediatrics, elderly |
Exposure calculations are usually expressed as:
- µg/day
- of/day
- mg/kg/day
Step 3: Hazard Identification
Hazard identification evaluates the inherent toxicity of each detected compound.
Toxicologists review:
- Published toxicology data
- Carcinogenicity studies
- Genotoxicity information
- Reproductive toxicity
- Organ toxicity
- Computational toxicology databases
Common Toxicological Resources
- Scientific literature
- Regulatory databases
- OECD date
- PubChem
- ECHA databases
When experimental data are unavailable, structure-activity relationship (SAR) models and in silico tools may be used.
5: Threshold of Toxicological Concern (TTC)
One of the most widely used approaches in Toxicological Assessment in Extractables & Leachables Studies is the Threshold of Toxicological Concern (TTC).
The TTC concept establishes exposure limits below which compounds are considered to present negligible risk.
Why TTC Is Important
TTC is especially useful for:
- Unknown compounds
- Trace-level impurities
- Early-stage risk assessment
- Compounds lacking extensive toxicity data
Typical TTC Thresholds
| Compound Type | Typical Threshold |
|---|---|
| Genotoxic compounds | Very low thresholds |
| Non-genotoxic compounds | Higher thresholds |
| Inhalation products | Route-specific limits |
| Parenteral products | Product-specific thresholds |
TTC helps prioritize compounds requiring further toxicological qualification.
6: Permitted Daily Exposure (PDE)
Permitted Daily Exposure (PDE) is another critical toxicological concept used to establish acceptable intake levels.
What Is PDE?
PDE represents the maximum acceptable daily intake of a compound without appreciable health risk.
PDE calculations incorporate:
- NOAEL data (No Observed Adverse Effect Level)
- Uncertainty factors
- Body weight adjustments
- Exposure duration
- Species extrapolation
A scientifically justified PDE provides strong regulatory support for E&L submissions.
7: Analytical Evaluation Threshold (AET)
The Analytical Evaluation Threshold (AET) determines which compounds must be identified and evaluated toxicologically.
Purpose of AET
AET helps laboratories:
- Prioritize analytical efforts
- Avoid unnecessary over-reporting
- Focus on clinically relevant compounds
AET Depends On
| Factor | Impact |
|---|---|
| Dose volume | Higher dose lowers AET |
| Safety threshold | Product-specific |
| Route of administration | Impacts acceptable exposure |
| Patient population | Pediatric products may require stricter thresholds |
Establishing a justified AET is essential for efficient E&L study design.
8: Toxicological Risk Assessment Process
A complete toxicological risk assessment integrates analytical data with toxicological interpretation.
Core Risk Assessment Components:
1. Hazard Identification
Determines whether a compound can cause harm.
2. Dose-Response Assessment
Evaluates toxicity relative to exposure level.
3. Exposure Assessment
Calculates actual patient exposure.
4. Risk Characterization
Determines whether observed exposure presents acceptable risk.
9: Special Considerations for High-Risk Drug Products
Certain pharmaceutical products require especially stringent E&L toxicological evaluations.
1. Inhalation Products
Inhalation therapies demand extremely sensitive assessments because lungs are highly vulnerable to contaminants.
Key Concerns
- Pulmonary toxicity
- Respiratory irritation
- Chronic exposure risks
2. Injectable Products
Parenteral products bypass natural protective barriers, increasing toxicological sensitivity.
Important Factors
- Systemic exposure
- Sterility concerns
- Low safety thresholds
3. Biologics
Biologic formulations may interact differently with leachables compared to small-molecule drugs.
Potential Risks
- Protein aggregation
- Immunogenicity
- Stability reduction
The increasing complexity of biologics and advanced therapies has accelerated industry focus on Future of Extractables and Leachables Testing and evolving regulatory frameworks for high-risk pharmaceutical products.
10: Challenges in Toxicological Assessment
Despite advances in analytical science, several challenges remain in E&L toxicology.
Major Challenges:
1. Unknown Compounds
Some detected compounds lack sufficient toxicological data.
2. Ultra-Trace Detection
Modern instruments detect compounds at extremely low concentrations that may not have clinical significance.
3. Complex Mixtures
Products may contain multiple interacting leachables.
4. Regulatory Variability
Different regulatory agencies may expect different reporting thresholds and risk approaches.
11: Why Partner With Specialized E&L Laboratories
Comprehensive E&L programs require expertise across multiple scientific disciplines.
A specialized laboratory provides:
- Advanced analytical instrumentation
- Toxicological expertise
- Regulatory guidance
- Method development
- Stability study support
- Comprehensive reporting
Companies often benefit from Outsourcing Extractables and Leachables E&L Testing Laboratory United States to access specialized instrumentation, regulatory expertise, and experienced scientific teams.
At ResolveMass Laboratories Inc., multidisciplinary teams support pharmaceutical and medical device companies with scientifically rigorous extractables and leachables testing programs aligned with global regulatory expectations.
Conclusion:
Toxicological Assessment in Extractables & Leachables Studies plays a vital role in ensuring pharmaceutical and medical device safety. By combining analytical chemistry with toxicological science, manufacturers can identify potential contaminants, evaluate patient exposure, and establish scientifically justified safety limits.
Approaches such as TTC, PDE, AET, and structured toxicological risk assessment frameworks help ensure that detected compounds remain within acceptable safety margins throughout the product lifecycle.
As regulatory expectations continue to evolve, robust E&L toxicology programs are becoming increasingly important for successful product development, regulatory approval, and patient protection.
Organizations seeking reliable E&L testing and toxicological assessment support benefit from partnering with experienced analytical laboratories capable of delivering scientifically defensible, regulatory-ready solutions.
Frequently Asked Questions:
Safety limits are established using toxicological risk assessment approaches such as TTC (Threshold of Toxicological Concern), PDE (Permitted Daily Exposure), and AET (Analytical Evaluation Threshold). Toxicologists evaluate compound identity, patient exposure levels, toxicological data, and safety margins. The goal is to ensure detected compounds remain below harmful exposure levels.
The Threshold of Toxicological Concern (TTC) is a risk assessment concept that establishes exposure limits below which compounds are considered unlikely to cause harm. TTC is especially useful for unknown compounds or impurities with limited toxicological data. It allows toxicologists to prioritize compounds requiring further evaluation.
Major regulatory organizations include the U.S. Food and Drug Administration, International Council for Harmonisation, and Product Quality Research Institute. These organizations provide frameworks for impurity assessment, safety thresholds, toxicological qualification, and risk management in E&L programs.
Toxicological assessment provides scientific justification that detected leachables are safe at observed exposure levels. Regulatory agencies expect comprehensive documentation including compound identification, exposure calculations, toxicological evaluation, and risk characterization. A strong E&L toxicology program improves regulatory readiness and supports product approval.
Toxicological Assessment in Extractables & Leachables Studies evaluates whether chemicals migrating from packaging, devices, or manufacturing systems pose safety risks to patients. It combines analytical chemistry with toxicology to determine acceptable exposure levels. These assessments help ensure pharmaceutical products remain safe throughout their shelf life. Regulatory agencies require such evaluations for many drug products and medical devices.
Reference
- Broschard TH, Glowienke S, Bruen US, Nagao LM, Teasdale A, Stults CL, Li KL, Iciek LA, Erexson G, Martin EA, Ball DJ. Assessing safety of extractables from materials and leachables in pharmaceuticals and biologics–current challenges and approaches. Regulatory Toxicology and Pharmacology. 2016 Nov 1;81:201-11.https://www.sciencedirect.com/science/article/pii/S027323001630232X
- Kuzmič S, Zlobec T, Dolenc MS, Roškar R, Lušin TT. Extractables and Leachables in Pharmaceutical Products: Potential Adverse Effects and Toxicological Risk Assessment. Toxics. 2026 Jan 20;14(1):92.https://www.mdpi.com/2305-6304/14/1/92
- Jenke D. Identification, analysis and safety assessment of leachables and extractables. TrAC Trends in Analytical Chemistry. 2018 Apr 1;101:56-65.https://www.sciencedirect.com/science/article/pii/S0165993617303321
- Jahn M. Leachables and extractables: from regulatory expectations to laboratory assessment. InChallenges in protein product development 2018 Jun 21 (pp. 337-351). Cham: Springer International Publishing.https://link.springer.com/chapter/10.1007/978-3-319-90603-4_16
- Parris P, Martin EA, Stanard B, Glowienke S, Dolan DG, Li K, Binazon O, Giddings A, Whelan G, Masuda-Herrera M, Bercu J. Considerations when deriving compound-specific limits for extractables and leachables from pharmaceutical products: Four case studies. Regulatory Toxicology and Pharmacology. 2020 Dec 1;118:104802.https://www.sciencedirect.com/science/article/pii/S0273230020302282


