
Introduction:
Octreotide PLGA Characterization plays a pivotal role in the development and regulatory approval of generic long-acting injectable depots. For complex formulations such as octreotide PLGA microspheres, proving polymer sameness is one of the most important requirements for Abbreviated New Drug Application (ANDA) success.
Octreotide depot formulations utilize poly(lactic-co-glycolic acid) (PLGA) polymers to achieve sustained drug release over extended periods. Even minor differences in polymer composition, molecular weight distribution, lactide:glycolide ratio, end-group chemistry, or residual solvents can significantly alter release kinetics and clinical performance.
Understanding the role of PLGA polymer grade in long-acting release formulation is therefore essential during formulation development and reverse engineering activities.
As regulatory scrutiny for complex generics continues to increase, pharmaceutical developers require robust analytical characterization strategies that establish a high degree of equivalence between the reference listed drug (RLD) and the proposed generic product. This case study discusses how advanced polymer characterization supports successful development of octreotide PLGA depot formulations.
Additional insights regarding PLGA characterization methods and PLGA reverse engineering CRO services further support analytical strategies for complex generic depots.
Summary
- Octreotide PLGA Characterization is critical for demonstrating polymer sameness in generic long-acting injectable (LAI) formulations.
- PLGA polymer properties directly influence drug release, stability, degradation, and therapeutic performance.
- Regulatory agencies require extensive physicochemical characterization for ANDA approval of complex depot products.
- Advanced analytical techniques such as GPC/SEC, NMR, LC-MS, DSC, FTIR, and residual solvent analysis are essential for establishing equivalence.
- Comprehensive polymer characterization reduces formulation risk and supports successful bioequivalence strategies.
- PLGA polymer characterization for generics and PLGA reverse engineering for ANDA are becoming increasingly important for complex injectable development programs.
- ResolveMass Laboratories Inc. provides analytical expertise for PLGA-based depot systems using advanced mass spectrometry and polymer characterization platforms.
1: Why Is Octreotide PLGA Characterization Important?
Octreotide PLGA Characterization is essential because PLGA polymer properties directly control drug release behavior, stability, and overall product performance. For long-acting injectable depot formulations, even small variations in polymer composition or molecular weight can significantly impact therapeutic outcomes and regulatory acceptance.
Octreotide depot formulations are highly sensitive polymeric systems in which the PLGA matrix functions as a critical release-controlling component. The polymer directly influences:
- Drug encapsulation efficiency
- Initial burst release
- Long-term release profile
- Polymer degradation rate
- Stability during storage
- Microsphere morphology
- Bioavailability
Because PLGA is a complex copolymer, identical formulation performance cannot be assumed solely based on matching excipient lists or nominal polymer specifications. Comprehensive physicochemical characterization is required to establish polymer sameness and support ANDA submissions for generic depot products.
Developers working with PLGA microsphere formulation development and PLGA peptide delivery systems must carefully evaluate these polymer-dependent performance characteristics.
Key Regulatory Concerns for ANDA Submissions
Regulatory agencies such as the FDA require extensive comparative characterization data demonstrating that the proposed generic product closely matches the Reference Listed Drug (RLD). The focus is not only on composition but also on functional equivalence of the polymer system.
| Characterization Parameter | Regulatory Importance |
|---|---|
| Molecular weight distribution | Controls polymer degradation and drug release |
| Lactide:glycolide ratio | Influences hydrophobicity, erosion, and release kinetics |
| Polymer end groups | Affects degradation behavior and stability |
| Residual monomers and solvents | Impacts product safety and quality |
| Thermal behavior | Indicates polymer consistency and processing characteristics |
| Particle morphology | Influences injectability and release performance |
| Impurity profile | Supports product quality and regulatory compliance |
Failure to adequately characterize PLGA polymers can lead to several development and regulatory risks, including:
- Bioequivalence challenges
- Inconsistent release profiles
- Stability failures
- Regulatory deficiencies
- Delayed ANDA approvals
For this reason, advanced analytical characterization using orthogonal techniques such as GPC/SEC, NMR, DSC, FTIR, LC-MS, and GC-MS has become a critical component of successful generic octreotide depot development.
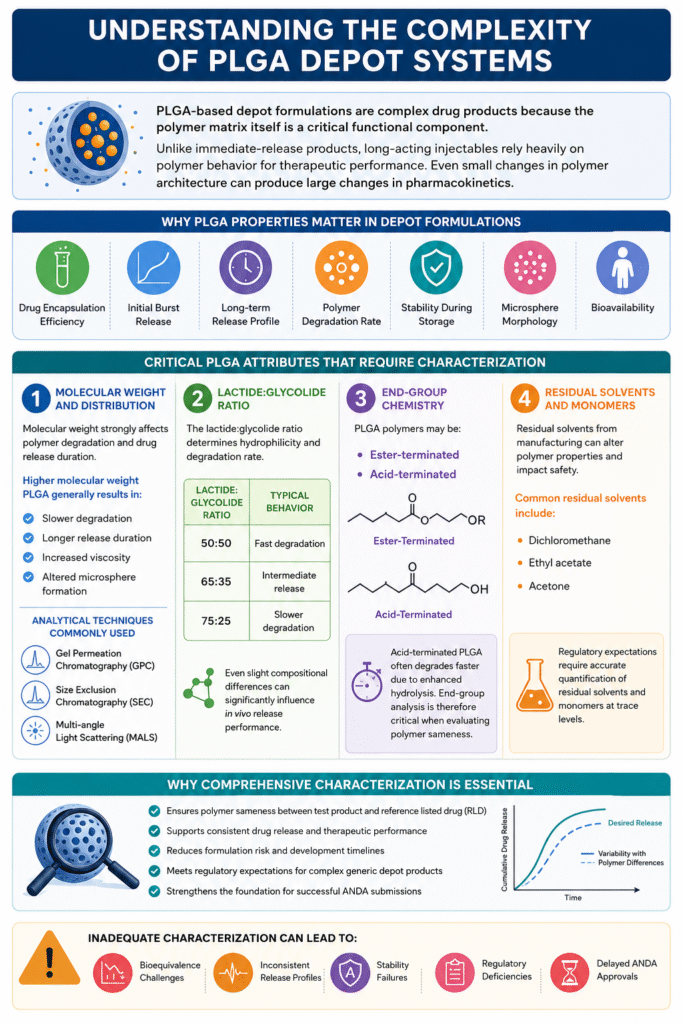
2: Understanding the Complexity of PLGA Depot Systems
PLGA-based depot formulations are classified as complex drug products because the polymer matrix itself directly controls drug release, degradation behavior, and therapeutic performance. Unlike conventional immediate-release formulations, long-acting injectable depots depend heavily on the physicochemical properties of the polymer system.
Even minor variations in polymer architecture can significantly alter pharmacokinetics, release kinetics, stability, and bioavailability. As a result, comprehensive polymer characterization is essential for demonstrating equivalence in generic depot formulations.
Additional scientific background regarding PLGA for oncology implant applications and long-acting injectable drug delivery technologies further highlights the complexity of sustained-release polymer systems.
Critical PLGA Attributes That Require Characterization:
1. Molecular Weight and Distribution
Molecular weight is one of the most important parameters influencing PLGA degradation and sustained drug release performance. Variations in molecular weight can directly affect erosion rate, microsphere integrity, and release duration.
Higher molecular weight PLGA generally results in:
- Slower polymer degradation
- Longer drug release duration
- Increased polymer viscosity
- Altered microsphere formation characteristics
Because PLGA polymers exhibit molecular weight distributions rather than single discrete values, both average molecular weight and polydispersity are important characterization parameters.
Common Analytical Techniques
The following analytical methods are widely used for molecular weight characterization:
- Gel Permeation Chromatography (GPC)
- Size Exclusion Chromatography (SEC)
- Multi-angle light scattering (MALS)
These techniques help establish polymer consistency and support comparative analysis between the test product and the reference listed drug (RLD).
2. Lactide:Glycolide Ratio
The lactide:glycolide ratio significantly influences polymer hydrophilicity, degradation rate, and drug release behavior. This ratio is one of the primary determinants of depot performance.
Typical PLGA compositions demonstrate different degradation profiles:
| Lactide:Glycolide Ratio | Typical Behavior |
|---|---|
| 50:50 | Fast degradation |
| 65:35 | Intermediate release |
| 75:25 | Slower degradation |
Even slight differences in copolymer composition can produce substantial changes in:
- Water uptake
- Polymer erosion
- Drug diffusion
- In vivo release kinetics
For ANDA submissions, accurate determination of monomer composition is therefore a critical regulatory expectation.
3. End-Group Chemistry
PLGA end-group chemistry can strongly influence hydrolysis rate, degradation behavior, and formulation stability.
PLGA polymers are commonly classified as:
- Ester-terminated
- Acid-terminated
Acid-terminated PLGA typically degrades faster because free carboxylic acid groups promote hydrolysis within the polymer matrix. This can accelerate polymer erosion and modify drug release characteristics.
End-group analysis is therefore essential when evaluating polymer sameness between generic and reference products.
Analytical methods commonly used for end-group characterization include:
- Mass spectrometry-based techniques
- Nuclear Magnetic Resonance (NMR) spectroscopy
- FTIR spectroscopy
4. Residual Solvents and Monomers
Residual solvents originating from polymer manufacturing or microsphere processing can affect product quality, polymer stability, and patient safety. Trace-level impurities may also alter release performance and degradation kinetics.
Common residual solvents found in PLGA formulations include:
- Dichloromethane
- Ethyl acetate
- Acetone
Organizations involved in GMP PLGA requirements for pharmaceutical applications and PLGA supplier qualification benefits often prioritize residual impurity control during development.
Residual monomers and processing impurities may additionally contribute to:
- Polymer instability
- Toxicological concerns
- Variability in release behavior
Regulatory agencies require highly sensitive and validated analytical methods capable of quantifying these compounds at trace levels according to ICH guidelines.
Common Analytical Techniques for Residual Solvent Analysis
Typical analytical platforms include:
- Gas Chromatography-Mass Spectrometry (GC-MS)
- Headspace GC
- LC-MS for impurity profiling
Accurate residual solvent characterization is an important component of comprehensive Octreotide PLGA Characterization strategies supporting successful ANDA submissions.
3: Case Study: Octreotide PLGA Characterization Strategy
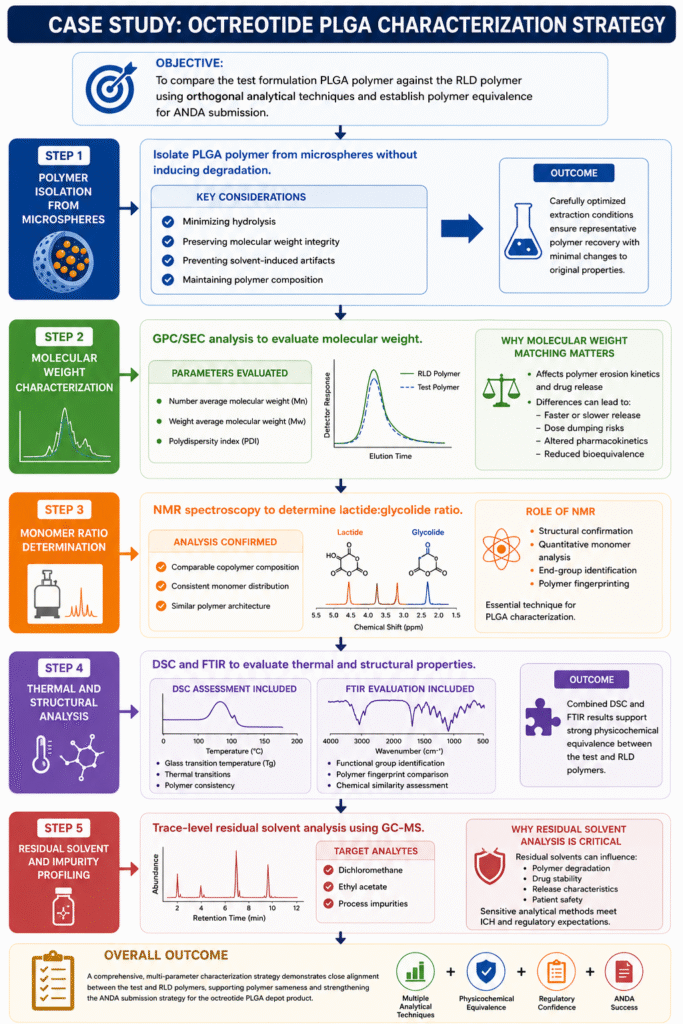
In this case study, a generic pharmaceutical developer sought to establish polymer equivalence for an octreotide PLGA depot product intended for ANDA submission.
The primary objective was to compare the test formulation polymer against the RLD polymer using orthogonal analytical techniques.
Similar analytical challenges have also been observed in dexamethasone implant PLGA characterization case studies and Exenatide PLGA microsphere characterization case studies.
Step 1: Polymer Isolation from Microspheres
The first challenge involved isolating PLGA polymer from the microsphere matrix without inducing degradation.
Key considerations included:
- Minimizing hydrolysis
- Preserving molecular weight integrity
- Preventing solvent-induced artifacts
- Maintaining polymer composition
Carefully optimized extraction conditions were used to ensure representative polymer recovery.
Step 2: Molecular Weight Characterization
GPC/SEC analysis was performed to evaluate:
- Number average molecular weight (Mn)
- Weight average molecular weight (Mw)
- Polydispersity index (PDI)
The analysis demonstrated close alignment between the test and reference polymers.
Why Molecular Weight Matching Matters
Drug release from PLGA depots depends heavily on polymer erosion kinetics.
Differences in molecular weight can lead to:
- Faster or slower release
- Dose dumping risks
- Altered pharmacokinetics
- Reduced bioequivalence
For this reason, regulators expect detailed comparative data during ANDA review.
Step 3: Monomer Ratio Determination
Nuclear Magnetic Resonance (NMR) spectroscopy was used to determine the lactide:glycolide ratio.
The analysis confirmed:
- Comparable copolymer composition
- Consistent monomer distribution
- Similar polymer architecture
Role of NMR in Octreotide PLGA Characterization
NMR is particularly valuable because it provides:
- Structural confirmation
- Quantitative monomer analysis
- End-group identification
- Polymer fingerprinting
This technique is widely considered essential for PLGA characterization studies.
Step 4: Thermal and Structural Analysis
Differential Scanning Calorimetry (DSC) and FTIR spectroscopy were used to evaluate thermal and structural properties.
DSC Assessment Included:
- Glass transition temperature (Tg)
- Thermal transitions
- Polymer consistency
FTIR Evaluation Included:
- Functional group identification
- Polymer fingerprint comparison
- Chemical similarity assessment
The combined results supported strong physicochemical equivalence between polymers.
Step 5: Residual Solvent and Impurity Profiling
Trace-level residual solvent analysis was performed using GC-MS techniques.
Target analytes included:
- Dichloromethane
- Ethyl acetate
- Process impurities
Why Residual Solvent Analysis Is Critical
Residual solvents can influence:
- Polymer degradation
- Drug stability
- Release characteristics
- Patient safety
Sensitive analytical methods are therefore required to meet ICH and regulatory expectations.
4: Analytical Techniques Used in Octreotide PLGA Characterization
A successful characterization strategy typically requires multiple orthogonal techniques.
| Technique | Purpose |
|---|---|
| GPC/SEC | Molecular weight analysis |
| NMR | Monomer ratio and structure |
| LC-MS | Impurity identification |
| GC-MS | Residual solvent analysis |
| DSC | Thermal characterization |
| FTIR | Functional group analysis |
| SEM | Microsphere morphology |
| ICP-MS | Elemental impurity testing |
Combining these techniques provides a comprehensive understanding of polymer equivalence.
5: Challenges in Demonstrating Polymer Sameness
Demonstrating polymer sameness for PLGA depot formulations is highly complex because PLGA polymers are inherently variable, sensitive to processing conditions, and difficult to fully characterize. For generic long-acting injectable products, even small polymer differences can influence drug release, degradation behavior, and therapeutic performance.
As a result, regulatory agencies expect detailed comparative characterization using multiple orthogonal analytical techniques to establish equivalence between the test product and the reference listed drug (RLD).
Common Challenges Include:
1. Polymer Heterogeneity
PLGA polymers are inherently heterogeneous materials rather than single uniform molecular structures. Unlike small-molecule excipients, PLGA consists of polymer chains with varying molecular weights and compositional distributions.
This heterogeneity can affect:
- Drug release kinetics
- Polymer degradation behavior
- Microsphere formation
- Stability profiles
Because PLGA exists as a molecular weight distribution rather than a discrete compound, characterization requires advanced analytical approaches capable of evaluating polymer populations rather than individual molecules.
Analytical Considerations:
Common techniques used to assess polymer heterogeneity include:
- Gel Permeation Chromatography (GPC)
- Size Exclusion Chromatography (SEC)
- Multi-angle light scattering (MALS)
These methods help evaluate:
- Polymer consistency
- Molecular weight distribution
- Polydispersity index (PDI)
2. Extraction-Induced Changes
Polymer isolation from microsphere formulations can unintentionally alter the original polymer properties if extraction conditions are not carefully controlled.
Improper extraction procedures may lead to:
- Molecular weight reduction
- End-group modification
- Polymer degradation
- Loss of polymer integrity
PLGA polymers are highly sensitive to:
- Moisture
- Heat
- Harsh solvents
- Extended processing times
Even minor extraction-induced artifacts can compromise comparative characterization data and create misleading conclusions regarding polymer sameness.
Importance of Optimized Extraction Methods
Carefully validated extraction procedures are therefore essential to:
- Preserve native polymer structure
- Minimize hydrolysis
- Maintain compositional integrity
- Ensure representative analytical results
3. Limited Reference Information
One of the major regulatory and technical challenges is the limited availability of detailed information about the reference listed drug (RLD) polymer system.
In many cases:
- Polymer specifications are proprietary
- Manufacturing processes are undisclosed
- Exact polymer sourcing information is unavailable
- Critical formulation parameters are only partially described
As a result, generic developers must reverse-engineer the polymer system using advanced analytical characterization strategies.
Organizations conducting reverse engineering of PLGA polymer in Lupron Depot and evaluating leuprolide depot formulation challenges encounter similar analytical complexities.
Reverse Engineering Challenges
Analytical teams often need to determine:
- Molecular weight characteristics
- Lactide:glycolide ratio
- End-group chemistry
- Residual solvent levels
- Thermal behavior
- Polymer architecture
This requires highly sensitive and orthogonal analytical methodologies capable of generating comprehensive polymer fingerprints.
4. Batch-to-Batch Variability
Even commercially approved PLGA depot products may exhibit controlled variability between manufacturing lots. This variability can complicate direct comparisons between the generic product and the RLD.
Potential sources of variability include:
- Polymer manufacturing differences
- Process conditions
- Residual solvent content
- Microsphere preparation methods
- Storage conditions
Small variations may influence:
- Drug release profiles
- Particle morphology
- Polymer degradation kinetics
- Stability performance
For this reason, comparative studies often require evaluation of multiple RLD batches to establish realistic variability ranges.
6: Regulatory Expectations for Complex Generic Depots
The FDA and other regulatory agencies continue to increase expectations for complex injectable generics.
For PLGA depot products, agencies typically expect:
- Extensive physicochemical characterization
- Orthogonal analytical methods
- Comparative release studies
- Impurity profiling
- Stability assessment
- In vitro and potentially in vivo correlation data
Importance of Risk-Based Analytical Strategies
A risk-based approach helps prioritize critical quality attributes (CQAs) most likely to affect clinical performance.
This includes evaluation of:
- Polymer composition
- Release kinetics
- Degradation pathways
- Particle morphology
- Stability behavior
Comprehensive characterization reduces regulatory uncertainty and strengthens ANDA submissions.
7: How Advanced Analytical Expertise Supports ANDA Success
Complex polymeric depots require multidisciplinary analytical capabilities.
Successful characterization programs often combine expertise in:
- Polymer science
- Mass spectrometry
- Chromatography
- Pharmaceutical formulation
- Regulatory science
At ResolveMass Laboratories Inc., advanced analytical platforms support detailed characterization of complex injectable products, including PLGA-based depot formulations.
Capabilities may include:
- High-resolution mass spectrometry
- Polymer molecular weight analysis
- Extractables and leachables testing
- Residual solvent analysis
- Structural elucidation
- Stability-indicating methods
These analytical approaches help pharmaceutical developers generate robust datasets for regulatory submissions.
Additional resources covering PLGA characterization of Lupron Depot and formulating highly potent APIs using PLGA microspheres further illustrate the growing complexity of PLGA-based drug delivery systems.
8: Future Trends in PLGA Depot Characterization
The future of complex generic development will likely involve increasingly sophisticated characterization technologies.
Emerging trends include:
- Advanced multidimensional chromatography
- High-resolution polymer mapping
- AI-assisted analytical interpretation
- Enhanced in vitro–in vivo correlation (IVIVC)
- Improved particle imaging techniques
As regulatory standards evolve, deeper polymer understanding will become even more important for ANDA success.
Further scientific discussions regarding PLGA for parenteral use continue to support innovation in long-acting injectable development.
Conclusion:
Octreotide PLGA Characterization is a critical component of developing successful generic long-acting injectable depot products. Demonstrating polymer sameness requires detailed evaluation of molecular weight, monomer ratio, end-group chemistry, thermal behavior, residual solvents, and impurity profiles.
Because PLGA polymer properties directly influence drug release and therapeutic performance, regulatory agencies expect robust analytical evidence supporting equivalence between the generic product and the reference listed drug.
Using orthogonal analytical techniques and experienced scientific interpretation, pharmaceutical companies can reduce development risk, strengthen ANDA submissions, and improve the likelihood of regulatory approval for complex depot formulations.
For organizations developing PLGA-based injectables, comprehensive analytical characterization is no longer optional—it is essential for success.
Frequently Asked Questions:
Molecular weight significantly impacts polymer degradation and drug release kinetics. Higher molecular weight PLGA typically degrades more slowly and provides extended drug release, while lower molecular weight polymers may result in faster release and altered pharmacokinetics. Accurate molecular weight matching is therefore essential for ANDA success.
The lactide:glycolide ratio controls polymer hydrophilicity, degradation behavior, and release profile. Different ratios produce different release durations and erosion rates. Even small changes in monomer composition can affect in vivo performance, making precise ratio determination critical during characterization studies.
Residual solvents can affect polymer stability, degradation, drug release behavior, and patient safety. Regulatory agencies require trace-level quantification of solvents such as dichloromethane, ethyl acetate, and acetone using validated analytical methods like GC-MS and headspace GC.
Major challenges include:
-Polymer heterogeneity
-Extraction-induced polymer changes
-Limited RLD formulation information
-Batch-to-batch variability
-Complex degradation behavior
Because PLGA polymers are highly sensitive systems, demonstrating sameness requires extensive analytical expertise and orthogonal characterization methods.
Orthogonal analytical techniques provide complementary information about the polymer system. Using multiple techniques improves confidence in characterization results and helps establish polymer equivalence more accurately. Regulatory agencies often expect this comprehensive approach for complex depot products.
NMR spectroscopy is widely used to determine lactide:glycolide ratio, polymer structure, and end-group chemistry. It also supports polymer fingerprinting and compositional analysis, making it one of the most important techniques for PLGA characterization studies.
Reference
- Beig A, Feng L, Walker J, Ackermann R, Hong JK, Li T, Wang Y, Qin B, Schwendeman SP. Physical–chemical characterization of octreotide encapsulated in commercial glucose-star PLGA microspheres. Molecular Pharmaceutics. 2020 Sep 2;17(11):4141-51.https://pubs.acs.org/doi/abs/10.1021/acs.molpharmaceut.0c00619
- Murty SB. Drug stability and release kinetics of a somatostatin analogue formulated in polymeric biodegradable microspheres. University of Kentucky; 2003.https://search.proquest.com/openview/3501126df23a8549ffe363e57a5a89cf/1?pq-origsite=gscholar&cbl=18750&diss=y
- Rhee YS, Sohn M, Woo BH, Thanoo BC, DeLuca PP, Mansour HM. Sustained-release delivery of octreotide from biodegradable polymeric microspheres. Aaps Pharmscitech. 2011 Dec;12(4):1293-301.https://link.springer.com/article/10.1208/s12249-011-9693-z
- Muddineti OS, Omri A. Current trends in PLGA based long-acting injectable products: The industry perspective. Expert opinion on drug delivery. 2022 May 4;19(5):559-76.https://www.tandfonline.com/doi/abs/10.1080/17425247.2022.2075845
