
Introduction:
A Bioanalytical CRO Partnership is often the backbone of successful drug development programs. Pharmaceutical and biotechnology companies rely on expert bioanalytical partners and bioanalytical laboratory services to generate accurate, regulatory-compliant data that guides critical development decisions.
From early discovery and preclinical evaluation to clinical trials and regulatory submissions, bioanalytical data generated through bioanalytical services in drug development drives major value inflection points that determine whether a drug candidate advances or stops.
A strong partnership with an experienced bioanalytical CRO enables companies to reduce risk, accelerate timelines, and generate high-quality analytical data that regulators and investors trust.
This article explores how Bioanalytical CRO Partnerships support key value inflection points, and why choosing the right CRO partner can significantly impact the success of your drug development program.
Share via:
Summary:
- Bioanalytical CRO Partnership plays a critical role in supporting drug development milestones and accelerating clinical progress.
- Strategic collaboration with a specialized bioanalytical CRO helps companies generate reliable pharmacokinetic (PK), pharmacodynamic (PD), and biomarker data required for regulatory decisions through services like PK/PD bioanalysis and biomarker bioanalytical services.
- Strong CRO partnerships enable faster proof-of-concept, IND-enabling bioanalytical studies, and clinical advancement.
- High-quality LC-MS/MS bioanalysis and high-sensitivity bioanalysis ensure data integrity and regulatory compliance.
- Early collaboration with experienced CROs helps reduce development risks and optimize study design using bioanalytical method development and rapid bioanalytical method development.
- A trusted partner like ResolveMass Laboratories Inc. supports pharmaceutical and biotech companies through key value inflection points using regulated bioanalytical services and clinical bioanalytical services.
1: Understanding Value Inflection Points in Drug Development
Value inflection points are milestones where new data significantly increases the value of a drug development program.
These milestones demonstrate scientific proof, reduce uncertainty, and attract investment or regulatory confidence. High-quality data generated through robust bioanalytical data and quality bioanalysis plays a key role in validating these milestones.
Key Drug Development Inflection Points
| Development Stage | Key Value Inflection Point | Role of Bioanalysis |
|---|---|---|
| Discovery | Lead candidate selection | Quantification of drug exposure |
| Preclinical | IND-enabling studies | PK/PD and toxicokinetic analysis |
| Early Clinical | Proof-of-concept | Biomarker and drug concentration analysis |
| Clinical Development | Dose optimization | PK modeling and therapeutic monitoring |
| Regulatory Submission | NDA/ANDA filing | Validated bioanalytical data |
At each stage, bioanalytical quantification using technologies like LC-MS/MS bioanalytical services ensures reliable measurement of drug concentrations, metabolites, and biomarkers.
2: Why Bioanalytical CRO Partnerships Are Critical for Drug Development
A Bioanalytical CRO Partnership provides specialized expertise, advanced instrumentation, and regulatory-compliant workflows that many pharmaceutical and biotechnology companies cannot maintain internally. By collaborating with an experienced bioanalytical partner, companies gain access to validated analytical methods, high-quality data generation, and scientific expertise that support critical development decisions.
Many biotech companies increasingly rely on bioanalytical outsourcing and outsourced bioanalysis services to reduce infrastructure costs while accessing expert scientific capabilities. This approach allows organizations to focus on drug discovery and clinical strategy while ensuring reliable bioanalytical data generation.
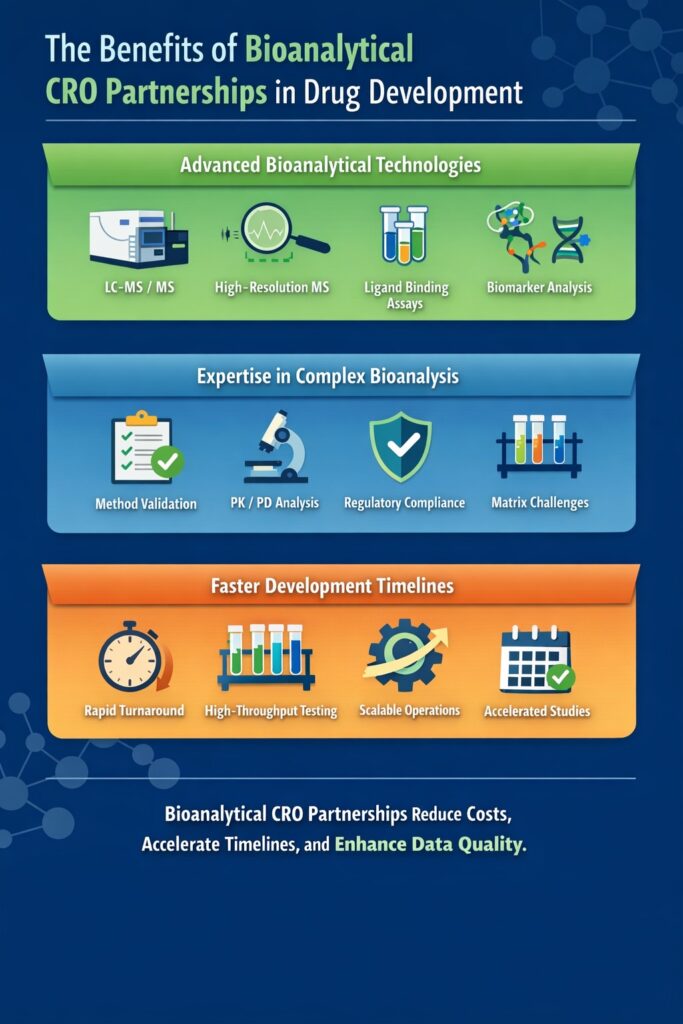
Key Benefits of Bioanalytical CRO Partnerships
1. Access to Advanced Bioanalytical Technologies
Modern drug development requires highly sophisticated analytical technologies to detect and quantify drugs, metabolites, and biomarkers with high sensitivity and accuracy.
Common technologies used in a Bioanalytical CRO Partnership include:
- LC-MS/MS
- High-resolution mass spectrometry
- Ligand-binding assays
- Biomarker analysis platforms
A trusted Bioanalytical CRO Partnership ensures access to validated instruments, optimized analytical workflows, and experienced scientists capable of handling complex drug molecules.
Advanced technologies like proteomics bioanalytical services and LC-MS/MS bioanalysis of xenobiotics enable sensitive detection of drug molecules and metabolites in biological samples, supporting accurate pharmacokinetic and pharmacodynamic analysis.
2. Expertise in Complex Bioanalysis
Drug development programs often involve complex analytical challenges that require specialized scientific expertise.
Experienced CRO scientists provide expertise in:
- bioanalytical method validation
- bioanalytical quantification
- PK/PD interpretation
- regulatory compliance
They also help address challenges such as bioanalytical matrix effects and challenges in bioanalytical method development, ensuring that analytical methods remain robust, reproducible, and regulatory compliant.
3. Faster Development Timelines
Drug development timelines are highly competitive, and delays in bioanalytical testing can significantly slow down program progress.
Strategic Bioanalytical CRO Partnerships help accelerate timelines by:
- high-throughput bioanalysis
- scalable bioanalysis infrastructure
- experienced project management
- rapid analytical turnaround
For emerging biotechnology companies, solutions such as affordable bioanalytical services for biotech startups and cost-effective bioanalytical services provide access to high-quality analytical support without requiring major infrastructure investments.
For startups, affordable bioanalytical services for biotech startups and cost-effective bioanalytical services are especially valuable.
3: Bioanalytical CRO Partnerships in Early Drug Discovery
Early-stage Bioanalytical CRO Partnerships help identify promising drug candidates and eliminate weak compounds quickly.
These programs often rely on bioanalytical CRO for drug discovery and integrated chemistry workflows such as integrated chemistry and bioanalytical CRO.
During discovery and preclinical research, bioanalysis provides:
- Drug concentration measurement
- Metabolic stability data
- Tissue distribution studies
- PK screening
Key Contributions at This Stage
- Rapid screening of candidate molecules
- Early pharmacokinetic profiling
- Drug exposure evaluation
- Metabolite identification
Researchers often utilize virtual bioanalytical strategy to optimize early program design.
These insights enable companies to focus resources on the most promising drug candidates.
4: Bioanalytical CRO Partnerships for IND-Enabling Studies
IND-enabling studies require validated bioanalytical methods and regulatory-grade data.
These studies involve toxicokinetic bioanalysis, PK/TK bioanalysis, and detailed method validation.
Companies preparing regulatory submissions rely on bioanalytical services for IND/NDA submissions.
A strong Bioanalytical CRO Partnership ensures that drug developers generate data required for Investigational New Drug (IND) applications.
Bioanalysis Requirements for IND Submission
Typical IND-supporting bioanalysis includes:
- Toxicokinetic studies
- PK profiling
- Bioanalytical method validation
- GLP-compliant sample analysis
Why CRO Expertise Matters
Regulatory authorities expect:
- Robust validated methods
- Accurate drug quantification
- Reproducible analytical workflows
Accurate bioanalytical stability testing is also critical for regulatory acceptance.
Experienced CROs like ResolveMass Laboratories Inc. implement strict quality control procedures and validated LC-MS workflows to ensure data reliability.
5: Bioanalytical CRO Partnerships Supporting Proof-of-Concept
Proof-of-concept studies are one of the most important value inflection points in drug development.
These studies rely on bioanalytical services for rapid proof-of-concept to generate early clinical data.
Positive proof-of-concept results demonstrate that a drug candidate:
- Reaches its target
- Produces measurable biological effects
- Shows acceptable pharmacokinetics
A reliable Bioanalytical CRO Partnership helps generate the data needed to demonstrate these outcomes.
Bioanalytical Data Required
Proof-of-concept studies often involve:
- PK/PD correlation
- Biomarker measurement
- Target engagement analysis
- Dose-response evaluation
These studies often require biomarker bioanalytical services and high sensitivity bioanalysis.
High-quality bioanalysis ensures confidence in early clinical data, which is critical for attracting investors and advancing clinical trials.
6: Bioanalytical CRO Partnerships in Clinical Development
During clinical trials, Bioanalytical CRO Partnerships support accurate measurement of drug exposure across patient populations.
Clinical bioanalysis plays a vital role in:
- Dose optimization
- Safety monitoring
- Therapeutic window identification
- Regulatory reporting
Clinical Bioanalysis Workflow
Typical CRO-supported workflow includes:
- Sample collection and handling
- Bioanalytical method validation
- Clinical sample analysis
- Data quality assurance
- PK/PD modeling
Maintaining data integrity in bioanalytical studies is essential for regulatory approval.
A reliable Bioanalytical CRO Partnership ensures regulatory compliance and consistent data quality across large clinical datasets.
7: Key Features of a High-Quality Bioanalytical CRO Partnership
A successful Bioanalytical CRO Partnership depends on selecting a partner with strong scientific capabilities, advanced analytical infrastructure, and deep regulatory expertise. The right CRO partner not only generates reliable data but also supports critical decision-making across the drug development lifecycle.
When evaluating a Bioanalytical CRO Partnership, companies should assess expertise in complex drug modalities, regulatory compliance frameworks, data integrity systems, and scalable bioanalytical capacity.
Scientific Expertise
Scientific expertise is one of the most important factors in a Bioanalytical CRO Partnership. Drug development programs increasingly involve complex therapeutic modalities that require specialized analytical approaches.
Key areas of expertise to look for include:
- LC-MS bioanalysis for oligonucleotides
- antibody-drug conjugate bioanalytical services
- cell and gene therapy bioanalysis
- biosimilar bioanalysis
Advanced modalities also require advanced bioanalytical strategies for complex drug modalities.
In addition, complex drug molecules often require advanced bioanalytical strategies for complex drug modalities, including specialized extraction methods, ultra-sensitive detection techniques, and robust validation protocols.
Regulatory Compliance
Regulatory compliance is another critical component of a reliable Bioanalytical CRO Partnership. Bioanalytical data generated during drug development must meet strict regulatory requirements to support clinical trials and regulatory submissions.
A qualified CRO should operate under internationally recognized regulatory frameworks such as:
- GLP standards
- FDA bioanalytical validation guidance
- ICH M10 regulations
Additionally, Understanding discovery vs regulated bioanalysis is essential when transitioning from early discovery to clinical development. This ensures that analytical workflows evolve appropriately as drug candidates move through the development pipeline.
Data Integrity
High-quality bioanalytical data is essential for regulatory acceptance and scientific credibility. A reliable Bioanalytical CRO Partnership must maintain rigorous data integrity practices to ensure the accuracy and traceability of analytical results.
Key elements of strong data integrity include:
- strict data traceability
- audit-ready documentation
- robust quality management systems
Strong data governance practices help maintain robust bioanalytical data that regulators, sponsors, and investors can trust.
Scalable Infrastructure
Drug development programs often expand rapidly as candidates progress from early studies to large clinical trials. A strong Bioanalytical CRO Partnership must therefore provide scalable infrastructure capable of handling increasing analytical demands.
Key infrastructure capabilities include:
- support for large clinical bioanalytical studies
- high-throughput bioanalysis platforms
- rapid sample processing and turnaround times
Scalable laboratory infrastructure ensures that bioanalytical workflows remain efficient and consistent as study sizes grow and timelines become more demanding.

8: How ResolveMass Supports Strategic Bioanalytical CRO Partnerships
ResolveMass Laboratories Inc. provides specialized bioanalytical services designed to support key drug development milestones.
ResolveMass Laboratories Inc. provides specialized bioanalytical services designed to support key development milestones.
Organizations worldwide rely on ResolveMass bioanalytical services overview to understand the full scope of capabilities available.
With deep expertise in LC-MS bioanalysis, ResolveMass helps pharmaceutical and biotech companies generate high-quality analytical data that supports regulatory and strategic decisions.
Core Capabilities
ResolveMass provides expertise in:
- Pharmacokinetic (PK) bioanalysis
- Pharmacodynamic (PD) studies
- Biomarker bioanalysis
- Oligonucleotide bioanalysis
- Peptide characterization
- Method development and validation
Advanced capabilities also include AI in bioanalysis and bioanalytical CRO for AI drug discovery.
Advantages of Partnering with ResolveMass
- Experienced bioanalytical scientists
- Advanced LC-MS instrumentation
- Regulatory-compliant workflows
- High data reliability
- Flexible project collaboration
Companies can also benefit from outsourced bioanalysis for drug development and bioanalytical services outsourcing for pharma.
These strengths make ResolveMass a trusted Bioanalytical CRO Partnership partner for companies seeking to accelerate development timelines and achieve critical milestones.
9: How Early Bioanalytical CRO Partnerships Reduce Development Risk
Engaging a Bioanalytical CRO Partnership early in development significantly reduces scientific and regulatory risks.
Startups especially benefit from outsource bioanalysis for biotech startups and transparent insights into bioanalytical testing services cost.
Early collaboration helps avoid common bioanalytical mistakes and improves study design.
Early collaboration enables:
- optimized study design
- validated analytical methods
- better PK/PD interpretation
- improved clinical planning
Strategic Benefits
Companies that establish strong CRO partnerships early often experience:
- fewer regulatory delays
- faster development timelines
- higher-quality data packages
- better investor confidence
Ultimately, this leads to stronger value inflection points throughout the development process.
Conclusion:
A Bioanalytical CRO Partnership is essential for generating reliable analytical data that drives drug development decisions.
From discovery and preclinical research to clinical trials and regulatory submissions, bioanalysis supports key value inflection points that determine the success of pharmaceutical programs.
Strategic collaboration with an experienced CRO ensures:
- accurate pharmacokinetic data
- regulatory-compliant bioanalysis
- faster development timelines
- reduced program risk
By partnering with specialized experts like ResolveMass Laboratories Inc., pharmaceutical and biotech companies gain access to advanced bioanalytical capabilities that accelerate drug development and strengthen regulatory confidence.
Frequently Asked Questions:
When selecting a Bioanalytical CRO Partnership, companies should evaluate:
-Scientific expertise in LC-MS/MS bioanalysis
-Experience with complex drug modalities
-Regulatory compliance (FDA, EMA, ICH guidelines)
-Data integrity and quality management systems
-Scalability for clinical studies
-Proven track record supporting IND and NDA submissions
These factors ensure reliable bioanalytical support throughout the drug development lifecycle.
Biotech startups often rely on Bioanalytical CRO Partnerships because building in-house bioanalytical laboratories requires significant infrastructure investment, specialized expertise, and regulatory compliance systems. Outsourcing allows startups to access advanced bioanalytical capabilities while focusing their resources on drug discovery and development.
Companies should ideally establish a Bioanalytical CRO Partnership early in drug discovery or preclinical development. Early collaboration allows CRO scientists to help design studies, develop analytical methods, and optimize pharmacokinetic strategies, reducing development risks and improving the quality of regulatory data packages.
A Bioanalytical CRO Partnership supports IND submissions by generating validated bioanalytical data required by regulatory authorities. This includes pharmacokinetic studies, toxicokinetic bioanalysis, biomarker measurements, and validated analytical methods that demonstrate the safety and exposure profile of a drug candidate before first-in-human trials.
Reference
- Xiaojing Yu & Arkady I Gusev. CRO Benchmarking for Clinical Biomarker Analysis Outsourcing.https://www.tandfonline.com/doi/full/10.4155/bio-2019-0123
- Scott Summerfield. The Business of Bioanalysis: New Technology Integration into Bioanalytical Workflows.https://www.tandfonline.com/doi/full/10.4155/bio-2018-0269
- Stephen Lowes. Outsourcing in Bioanalysis: A CRO Perspective.https://www.tandfonline.com/doi/full/10.4155/bio-2017-4994
- Philip Timmerman. Beyond contracts: principles for Pharma–CRO collaboration from the European Bioanalysis Forum.https://www.tandfonline.com/doi/full/10.1080/17576180.2025.2582422
- Andrew Dolman, Xiaojing Yu. Data Driven CRO Benchmarking for Biomarker Analysis.https://www.tandfonline.com/doi/abs/10.4155/bio-2020-0164
- Makoto Niwa. Pitfalls in bioanalytical work with a Japanese CRO or sponsor: from a project risk management perspective.https://www.tandfonline.com/doi/full/10.4155/bio-2022-0224


